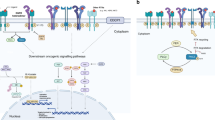
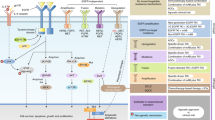
Key Points
-
Epidermal growth factor receptor (EGFR)-mutant tumours define a unique subset of non-small-cell lung cancer (NSCLC). These tumours display sensitivity to small-molecule EGFR tyrosine kinase inhibitors (TKIs).
-
Prospective clinical trials have now validated the correlation between EGFR mutation and TKI sensitivity.
-
Multiple mechanisms of primary and secondary resistance of lung tumours to EGFR TKIs have been elucidated.
-
Unfortunately, all patients eventually develop acquired resistance. Secondary mutations in EGFR and/or amplification of the gene encoding the MET receptor tyrosine kinase account for most cases of resistance.
-
On the basis of promising preclinical data, many new rationally designed strategies are now being pursued in clinical trials to overcome acquired resistance.
-
EGFR-mutant lung cancer can serve as a model for other cancers driven by oncogenic kinases.
Abstract
Epidermal growth factor receptor (EGFR)-mutant non-small-cell lung cancer (NSCLC) was first recognized in 2004 as a distinct, clinically relevant molecular subset of lung cancer. The disease has been the subject of intensive research at both the basic scientific and clinical levels, becoming a paradigm for how to understand and treat oncogene-driven carcinomas. Although patients with EGFR-mutant tumours have increased sensitivity to tyrosine kinase inhibitors (TKIs), primary and acquired resistance to these agents remains a major clinical problem. This Review summarizes recent developments aimed at treating and ultimately curing the disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jemal, A. et al. Cancer statistics, 2009. CA Cancer J. Clin. 59, 225–249 (2009).
Goldstraw, P. et al. The IASLC Lung Cancer Staging Project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J. Thorac. Oncol. 2, 706–714 (2007).
Hansen, H. H. et al. Combination chemotherapy of advanced lung cancer: a randomized trial. Cancer 38, 2201–2207 (1976).
Weinstein, I. B. Cancer. Addiction to oncogenes — the Achilles heal of cancer. Science 297, 63–64 (2002).
Hynes, N. E. & Lane, H. A. ERBB receptors and cancer: the complexity of targeted inhibitors. Nature Rev. Cancer 5, 341–354 (2005).
Marks, J. L. et al. Prognostic and therapeutic implications of EGFR and KRAS mutations in resected lung adenocarcinoma. J. Thorac. Oncol. 3, 111–116 (2008).
Herbst, R. S. et al. Selective oral epidermal growth factor receptor tyrosine kinase inhibitor ZD1839 is generally well-tolerated and has activity in non-small-cell lung cancer and other solid tumors: results of a Phase I trial. J. Clin. Oncol. 20, 3815–3825 (2002).
Albanell, J. et al. Pharmacodynamic studies of the epidermal growth factor receptor inhibitor ZD1839 in skin from cancer patients: histopathologic and molecular consequences of receptor inhibition. J. Clin. Oncol. 20, 110–124 (2002).
Ranson, M. et al. ZD1839, a selective oral epidermal growth factor receptor-tyrosine kinase inhibitor, is well tolerated and active in patients with solid, malignant tumors: results of a phase I trial. J. Clin. Oncol. 20, 2240–2250 (2002).
Nakagawa, K. et al. Phase I pharmacokinetic trial of the selective oral epidermal growth factor receptor tyrosine kinase inhibitor gefitinib ('Iressa', ZD1839) in Japanese patients with solid malignant tumors. Ann. Oncol. 14, 922–930 (2003).
Kris, M. G. et al. Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer: a randomized trial. JAMA 290, 2149–2158 (2003).
Fukuoka, M. et al. Multi-institutional randomized Phase II trial of gefitinib for previously treated patients with advanced non-small-cell lung cancer (The IDEAL 1 Trial). J. Clin. Oncol. 21, 2237–2246 (2003).
Miller, V. A. et al. Bronchioloalveolar pathologic subtype and smoking history predict sensitivity to gefitinib in advanced non-small-cell lung cancer. J. Clin. Oncol. 22, 1103–1109 (2004).
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–2139 (2004).
Paez, J. G. et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304, 1497–1500 (2004).
Pao, W. et al. EGF receptor gene mutations are common in lung cancers from 'never smokers' and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc. Natl Acad. Sci. USA 101, 13306–13311 (2004).
Nguyen, K. S., Kobayashi, S. & Costa, D. B. Acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small-cell lung cancers dependent on the epidermal growth factor receptor pathway. Clin. Lung Cancer 10, 281–289 (2009).
Shepherd, F. A. et al. Erlotinib in previously treated non-small-cell lung cancer. N. Engl. J. Med. 353, 123–132 (2005).
Thatcher, N. et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). Lancet 366, 1527–1537 (2005).
Mok, T. S. et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med. 361, 947–957 (2009). This paper presents results from IPASS (a randomized prospective Phase III clinical trial), which demonstrated the superiority of gefitinib over chemotherapy for the treatment of East Asian patients with chemotherapy-naive metastatic pulmonary adenocarcinoma harbouring EGFR mutations.
Mitsudomi, T. et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised Phase 3 trial. Lancet Oncol. 11, 121–128 (2009). This paper presents results from the WJTOG3405 trial (a randomized prospective Phase III clinical trial), which demonstrated the superiority of gefitinib over chemotherapy in prospectively genotyped Japanese patients with EGFR -mutant NSCLC.
Miller, V. A. et al. Molecular characteristics of bronchioloalveolar carcinoma and adenocarcinoma, bronchioloalveolar carcinoma subtype, predict response to erlotinib. J. Clin. Oncol. 26, 1472–1478 (2008).
Rosell, R. et al. Screening for epidermal growth factor receptor mutations in lung cancer. N. Engl. J. Med. 361, 958–967 (2009). This paper shows the effectiveness of erlotinib in Caucasian (Spanish) patients with EGFR -mutant NSCLC.
Maemondo, M. et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N. Engl. J. Med. 362, 2380–2388 (2010). References 20–24 definitively establish the clinical role of EGFR mutations and EGFR TKIs in lung cancer.
Yang, C. H. et al. Specific EGFR mutations predict treatment outcome of stage IIIB/IV patients with chemotherapy-naive non-small-cell lung cancer receiving first-line gefitinib monotherapy. J. Clin. Oncol. 26, 2745–2753 (2008).
Han, S. W. et al. Predictive and prognostic impact of epidermal growth factor receptor mutation in non-small-cell lung cancer patients treated with gefitinib. J. Clin. Oncol. 23, 2493–2501 (2005).
Pao, W. & Ladanyi, M. Epidermal growth factor receptor mutation testing in lung cancer: searching for the ideal method. Clin. Cancer Res. 13, 4954–4955 (2007).
Han, S. W. et al. Mucoepidermoid carcinoma of lung: potential target of EGFR-directed treatment. Lung Cancer 61, 30–34 (2008).
O'Neill, I. D. Gefitinib as targeted therapy for mucoepidermoid carcinoma of the lung: possible significance of CRTC1–MAML2 oncogene. Lung Cancer 64, 129–130 (2009).
Coxon, A. et al. Mect1–Maml2 fusion oncogene linked to the aberrant activation of cyclic AMP/CREB regulated genes. Cancer Res. 65, 7137–7144 (2005).
Behboudi, A. et al. Molecular classification of mucoepidermoid carcinomas-prognostic significance of the MECT1–MAML2 fusion oncogene. Genes Chromosomes Cancer 45, 470–481 (2006).
Hirsch, F. R. et al. Increased epidermal growth factor receptor gene copy number detected by fluorescence in situ hybridization associates with increased sensitivity to gefitinib in patients with bronchioloalveolar carcinoma subtypes: a Southwest Oncology Group Study. J. Clin. Oncol. 23, 6838–6845 (2005).
Cappuzzo, F. et al. Epidermal growth factor receptor gene and protein and gefitinib sensitivity in non-small-cell lung cancer. J. Natl Cancer Inst. 97, 643–655 (2005).
Hirsch, F. R. et al. Molecular predictors of outcome with gefitinib in a Phase III placebo-controlled study in advanced non-small-cell lung cancer. J. Clin. Oncol. 24, 5034–5042 (2006).
Tsao, M. S. et al. Erlotinib in lung cancer — molecular and clinical predictors of outcome. N. Engl. J. Med. 353, 133–144 (2005).
Hirsch, F. R. et al. Combination of EGFR gene copy number and protein expression predicts outcome for advanced non-small-cell lung cancer patients treated with gefitinib. Ann. Oncol. 18, 752–760 (2007).
Parra, H. S. et al. Analysis of epidermal growth factor receptor expression as a predictive factor for response to gefitinib ('Iressa', ZD1839) in non-small-cell lung cancer. Br. J. Cancer 91, 208–212 (2004).
Yeo, W.-L. et al. Erlotinib at a dose of 25 mg daily for non-small-cell lung cancers with EGFR mutations. J. Thorac. Oncol. 5, 1048–1053 (2010).
Gandhi, J. et al. Alterations in genes of the EGFR signaling pathway and their relationship to EGFR tyrosine kinase inhibitor sensitivity in lung cancer cell lines. PLoS ONE 4, e4576 (2009).
Bean, J. et al. MET amplification occurs with or without T790M mutations in EGFR mutant lung tumors with acquired resistance to gefitinib or erlotinib. Proc. Natl Acad. Sci. USA 104, 20932–20937 (2007). This work confirms MET amplification as a mechanism of acquired resistance to gefitinib, extends the finding to erlotinib and shows that resistance can occur with or without the T790M change.
Engelman, J. A. et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 316, 1039–1043 (2007). This paper reports the discovery of MET amplification as a mechanism of acquired resistance to gefitinib in cell line models and patient samples in mediating resistance. References 40 and 41 establish MET amplification as a mechanism of acquired resistance to EGFR TKIs in lung cancer.
Rosell, R., Viteri, S., Molina, M. A., Benlloch, S. & Taron, M. Epidermal growth factor receptor tyrosine kinase inhibitors as first-line treatment in advanced nonsmall-cell lung cancer. Curr. Opin. Oncol. 22, 112–120 (2010).
Mendelsohn, J. & Baselga, J. Epidermal growth factor receptor targeting in cancer. Semin. Oncol. 33, 369–385 (2006).
Bonner, J. A. et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N. Engl. J. Med. 354, 567–578 (2006).
Jonker, D. J. et al. Cetuximab for the treatment of colorectal cancer. N. Engl. J. Med. 357, 2040–2048 (2007).
Hanna, N. et al. Phase II trial of cetuximab in patients with previously treated non-small-cell lung cancer. J. Clin. Oncol. 24, 5253–5258 (2006).
Rosell, R. et al. Randomized Phase II study of cetuximab plus cisplatin/vinorelbine compared with cisplatin/vinorelbine alone as first-line therapy in EGFR-expressing advanced non-small-cell lung cancer. Ann. Oncol. 19, 362–369 (2008).
Pirker, R. & Minar, W. Chemotherapy of advanced non-small cell lung cancer. Front. Radiat. Ther. Oncol. 42, 157–163 (2010).
Lynch, T. J. et al. Cetuximab and first-line taxane/carboplatin chemotherapy in advanced non-small-cell lung cancer: results of the randomized multicenter Phase III trial BMS099. J. Clin. Oncol. 28, 911–917 (2010).
Mukohara, T. et al. Differential effects of gefitinib and cetuximab on non-small-cell lung cancers bearing epidermal growth factor receptor mutations. J. Natl Cancer Inst. 97, 1185–1194 (2005).
Khambata-Ford, S. et al. Analysis of potential predictive markers of cetuximab benefit in BMS099, a phase III study of cetuximab and first-line taxane/carboplatin in advanced non-small-cell lung cancer. J. Clin. Oncol. 28, 918–927 (2010).
Li, S. et al. Structural basis for inhibition of the epidermal growth factor receptor by cetuximab. Cancer Cell 7, 301–311 (2005).
Ji, H. et al. The impact of human EGFR kinase domain mutations on lung tumorigenesis and in vivo sensitivity to EGFR-targeted therapies. Cancer Cell 9, 485–495 (2006).
Regales, L. et al. Dual targeting of EGFR can overcome a major drug resistance mutation in mouse models of EGFR mutant lung cancer. J. Clin. Invest. 119, 3000–3010 (2009). This paper identifies a potential new strategy to overcome T790M-mediated resistance using a small-molecule inhibitor (BIBW2992) combined with an EGFR-specific antibody (cetuximab) in an EGFR-L858R and T790M transgenic mouse model.
Soh, J. et al. Oncogene mutations, copy number gains and mutant allele specific imbalance (MASI) frequently occur together in tumor cells. PLoS ONE 4, e7464 (2009).
Balak, M. N. et al. Novel D761Y and common secondary T790M mutations in epidermal growth factor receptor-mutant lung adenocarcinomas with acquired resistance to kinase inhibitors. Clin. Cancer Res. 12, 6494–6501 (2006).
Weir, B. A. et al. Characterizing the cancer genome in lung adenocarcinoma. Nature 450, 893–898 (2007).
Ding, L. et al. Somatic mutations affect key pathways in lung adenocarcinoma. Nature 455, 1069–1075 (2008).
Chitale, D. et al. An integrated genomic analysis of lung cancer reveals loss of DUSP4 in EGFR-mutant tumors. Oncogene 28, 2773–2783 (2009).
Yun, C. H. et al. Structures of lung cancer-derived EGFR mutants and inhibitor complexes: mechanism of activation and insights into differential inhibitor sensitivity. Cancer Cell 11, 217–227 (2007).
Yun, C. H. et al. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc. Natl Acad. Sci. USA 105, 2070–2075 (2008). This paper proposes that the T790M mutation in EGFR causes drug resistance by restoring the affinity for ATP.
Carey, K. D. et al. Kinetic analysis of epidermal growth factor receptor somatic mutant proteins shows increased sensitivity to the epidermal growth factor receptor tyrosine kinase inhibitor, erlotinib. Cancer Res. 66, 8163–8171 (2006).
Red Brewer, M. et al. The juxtamembrane region of the EGF receptor functions as an activation domain. Mol. Cell 34, 641–651 (2009).
Godin-Heymann, N. et al. Oncogenic activity of epidermal growth factor receptor kinase mutant alleles is enhanced by the T790M drug resistance mutation. Cancer Res. 67, 7319–7326 (2007).
Mulloy, R. et al. Epidermal growth factor receptor mutants from human lung cancers exhibit enhanced catalytic activity and increased sensitivity to gefitinib. Cancer Res. 67, 2325–2330 (2007).
Gong, Y. et al. Induction of BIM is essential for apoptosis triggered by EGFR kinase inhibitors in mutant EGFR-dependent lung adenocarcinomas. PLoS Med. 4, e294 (2007).
Costa, D. B. et al. Effects of erlotinib in EGFR mutated non-small cell lung cancers with resistance to gefitinib. Clin. Cancer Res. 14, 7060–7067 (2008).
Cragg, M. S., Kuroda, J., Puthalakath, H., Huang, D. C. & Strasser, A. Gefitinib-induced killing of NSCLC cell lines expressing mutant EGFR requires BIM and can be enhanced by BH3 mimetics. PLoS Med. 4, 1681–1690 (2007).
Deng, J. et al. Proapoptotic BH3-only BCL-2 family prote in BIM connects death signaling from epidermal growth factor receptor inhibition to the mitochondrion. Cancer Res. 67, 11867–11875 (2007). References 66–69 show that the pro-apoptotic BCL-2 family member BIM is required for EGFR TKI-induced apoptosis in EGFR -mutant lung cancer and that the BCL-2 antagonist ABT-737 can enhance TKI-induced cell killing.
Faber, A. C., Wong, K. K. & Engelman, J. A. Differences underlying EGFR and HER2 oncogene addiction. Cell Cycle 9, 851–852 (2010).
Shigematsu, H. et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. J. Natl Cancer Inst. 97, 339–346 (2005).
Politi, K. et al. Lung adenocarcinomas induced in mice by mutant EGF receptors found in human lung cancers respond to a tyrosine kinase inhibitor or to down-regulation of the receptors. Genes Dev. 20, 1496–1510 (2006).
Kosaka, T., Yatabe, Y., Onozato, R., Kuwano, H. & Mitsudomi, T. Prognostic implication of EGFR, KRAS, and TP53 gene mutations in a large cohort of Japanese patients with surgically treated lung adenocarcinoma. J. Thorac. Oncol. 4, 22–29 (2009).
Eisenhauer, E. A. et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur. J. Cancer 45, 228–247 (2009).
Wu, J. Y. et al. Lung cancer with epidermal growth factor receptor exon 20 mutations is associated with poor gefitinib treatment response. Clin. Cancer Res. 14, 4877–4882 (2008).
Greulich, H. et al. Oncogenic transformation by inhibitor-sensitive and -resistant EGFR mutants. PLoS Med. 2, e313 (2005).
Maheswaran, S. et al. Detection of mutations in EGFR in circulating lung-cancer cells. N. Engl. J. Med. 359, 366–377 (2008).
Inukai, M. et al. Presence of epidermal growth factor receptor gene T790M mutation as a minor clone in non-small cell lung cancer. Cancer Res. 66, 7854–7858 (2006).
Prudkin, L., Tang, X. & Wistuba, I. I. Germ-line and somatic presentations of the EGFR T790M mutation in lung cancer. J. Thorac. Oncol. 4, 139–141 (2009).
Tam, I. Y. et al. Distinct epidermal growth factor receptor and KRAS mutation patterns in non-small cell lung cancer patients with different tobacco exposure and clinicopathologic features. Clin. Cancer Res. 12, 1647–1653 (2006).
Tam, I. Y. et al. Double EGFR mutants containing rare EGFR mutant types show reduced in vitro response to gefitinib compared with common activating missense mutations. Mol. Cancer Ther. 8, 2142–2151 (2009).
Kawano, O. et al. PIK3CA mutation status in Japanese lung cancer patients. Lung Cancer 54, 209–215 (2006).
Engelman, J. A. et al. Allelic dilution obscures detection of a biologically significant resistance mutation in EGFR-amplified lung cancer. J. Clin. Invest. 116, 2695–2706 (2006).
Sos, M. L. et al. PTEN loss contributes to erlotinib resistance in EGFR-mutant lung cancer by activation of Akt and EGFR. Cancer Res. 69, 3256–3261 (2009).
Vivanco, I. et al. The phosphatase and tensin homolog regulates epidermal growth factor receptor (EGFR) inhibitor response by targeting EGFR for degradation. Proc. Natl Acad. Sci. USA 107, 6459–6464 (2010).
Sharma, S. V. et al. A chromatin-mediated reversible drug-tolerant state in cancer cell subpopulations. Cell 141, 69–80 (2010).
Gong, Y. et al. High expression levels of total IGF-1R and sensitivity of NSCLC cells in vitro to an anti-IGF-1R antibody (R1507). PLoS ONE 4, e7273 (2009).
Pao, W. et al. KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib. PLoS Med. 2, e17 (2005).
Linardou, H. et al. Assessment of somatic k-RAS mutations as a mechanism associated with resistance to EGFR-targeted agents: a systematic review and meta-analysis of studies in advanced non-small-cell lung cancer and metastatic colorectal cancer. Lancet Oncol. 9, 962–972 (2008).
Brose, M. S. et al. BRAF and RAS mutations in human lung cancer and melanoma. Cancer Res. 62, 6997–7000 (2002).
Davies, H. et al. Mutations of the BRAF gene in human cancer. Nature 417, 949–954 (2002).
Naoki, K., Chen, T. H., Richards, W. G., Sugarbaker, D. J. & Meyerson, M. Missense mutations of the BRAF gene in human lung adenocarcinoma. Cancer Res. 62, 7001–7003 (2002).
Poulikakos, P. I., Zhang, C., Bollag, G., Shokat, K. M. & Rosen, N. RAF inhibitors transactivate RAF dimers and ERK signalling in cells with wild-type BRAF. Nature 464, 427–430 (2010).
Solit, D. B. et al. BRAF mutation predicts sensitivity to MEK inhibition. Nature 439, 358–362 (2006).
Pratilas, C. A. et al. Genetic predictors of MEK dependence in non-small cell lung cancer. Cancer Res. 68, 9375–9383 (2008).
Haura, E. B. et al. A Phase II study of PD-0325901, an oral MEK inhibitor, in previously treated patients with advanced non-small cell lung cancer. Clin. Cancer Res. 16, 2450–2457 (2010).
Soda, M. et al. Identification of the transforming EML4–ALK fusion gene in non-small-cell lung cancer. Nature 448, 561–566 (2007).
Rikova, K. et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 131, 1190–1203 (2007).
Horn, L. & Pao, W. EML4–ALK: honing in on a new target in non-small-cell lung cancer. J. Clin. Oncol. 27, 4232–4235 (2009).
Shaw, A. T. et al. Clinical features and outcome of patients with non-small-cell lung cancer who harbor EML4–ALK. J. Clin. Oncol. 27, 4247–4253 (2009).
Yano, S. et al. Hepatocyte growth factor induces gefitinib resistance of lung adenocarcinoma with epidermal growth factor receptor-activating mutations. Cancer Res. 68, 9479–9487 (2008).
Turke, A. B. et al. Preexistence and clonal selection of MET amplification in EGFR mutant NSCLC. Cancer Cell 17, 77–88 (2010).
Jackman, D. et al. Clinical definition of acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small-cell lung cancer. J. Clin. Oncol. 28, 357–360 (2009).
Tamborini, E. et al. A new mutation in the KIT ATP pocket causes acquired resistance to imatinib in a gastrointestinal stromal tumor patient. Gastroenterology 127, 294–299 (2004).
Gorre, M. E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR–ABL gene mutation or amplification. Science 293, 876–880 (2001).
Kobayashi, S. et al. EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 352, 786–792 (2005).
Pao, W. et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2, e73 (2005).
Clarke, J. L., Pao, W., Wu, N., Miller, V. A. & Lassman, A. B. High dose weekly erlotinib achieves therapeutic concentrations in CSF and is effective in leptomeningeal metastases from epidermal growth factor receptor mutant lung cancer. J. Neurooncol. 99, 283–286 (2010).
Jackman, D. M. et al. Response and resistance in a non-small-cell lung cancer patient with an epidermal growth factor receptor mutation and leptomeningeal metastases treated with high-dose gefitinib. J. Clin. Oncol. 24, 4517–4520 (2006).
Bell, D. W. et al. Inherited susceptibility to lung cancer may be associated with the T790M drug resistance mutation in EGFR. Nature Genet. 37, 1315–1316 (2005).
Girard, N. et al. Analysis of genetic variants in never-smokers with lung cancer facilitated by an internet-based blood collection protocol: a preliminary report. Clin. Cancer Res. 16, 755–763 (2010).
Vikis, H. et al. EGFR-T790M is a rare lung cancer susceptibility allele with enhanced kinase activity. Cancer Res. 67, 4665–4670 (2007).
Sos, M. L. et al. Chemogenomic profiling provides insights into the limited activity of irreversible EGFR inhibitors in tumor cells expressing the T790M EGFR resistance mutation. Cancer Res. 70, 868–874 (2010).
Mok, T. S. Living with imperfection. J. Clin. Oncol. 28, 191–192 (2010).
Riely, G. J. et al. Prospective assessment of discontinuation and reinitiation of erlotinib or gefitinib in patients with acquired resistance to erlotinib or gefitinib followed by the addition of everolimus. Clin. Cancer Res. 13, 5150–5155 (2007).
Milton, D. T. et al. Molecular on/off switch. J. Clin. Oncol. 24, 4940–4942 (2006).
Kurata, T. et al. Effect of re-treatment with gefitinib ('Iressa', ZD1839) after acquisition of resistance. Ann. Oncol. 15, 173–174 (2004).
Oh, I.-J. et al. in AACR–IASLC Joint Conference on Molecular Origins of Lung Cancer: Prospects for Personalized Prevention and Therapy (Coronado, California USA, 2010).
Yano, S. et al. Retreatment of lung adenocarcinoma patients with gefitinib who had experienced favorable results from their initial treatment with this selective epidermal growth factor receptor inhibitor: a report of three cases. Oncol. Res. 15, 107–111 (2005).
Costa, D. B., Schumer, S. T., Tenen, D. G. & Kobayashi, S. Differential responses to erlotinib in epidermal growth factor receptor (EGFR)-mutated lung cancers with acquired resistance to gefitinib carrying the L747S or T790M secondary mutations. J. Clin. Oncol. 26, 1182–1186 (2008).
Bean, J. et al. Acquired resistance to epidermal growth factor receptor kinase inhibitors associated with a novel T854A mutation in a patient with EGFR-mutant lung adenocarcinoma. Clin. Cancer Res. 14, 7519–7525 (2008).
Avizienyte, E., Ward, R. A. & Garner, A. P. Comparison of the EGFR resistance mutation profiles generated by EGFR-targeted tyrosine kinase inhibitors and the impact of drug combinations. Biochem. J. 415, 197–206 (2008).
Costa, D. B. et al. BIM mediates EGFR tyrosine kinase inhibitor-induced apoptosis in lung cancers with oncogenic EGFR mutations. PLoS Med. 4, 1669–1680 (2007).
Kumar, A., Petri, E. T., Halmos, B. & Boggon, T. J. Structure and clinical relevance of the epidermal growth factor receptor in human cancer. J. Clin. Oncol. 26, 1742–1751 (2008).
Politi, K., Fan, P. D., Shen, R., Zakowski, M. & Varmus, H. Erlotinib resistance in mouse models of epidermal growth factor receptor-induced lung adenocarcinoma. Dis. Model. Mech. 3, 111–119 (2010).
Frederick, B. A. et al. Epithelial to mesenchymal transition predicts gefitinib resistance in cell lines of head and neck squamous cell carcinoma and non-small cell lung carcinoma. Mol. Cancer Ther. 6, 1683–1691 (2007).
Uramoto, H. et al. Epithelial-mesenchymal transition in EGFR-TKI acquired resistant lung adenocarcinoma. Anticancer Res. 30, 2513–2517 (2010).
Barr, S. et al. Bypassing cellular EGF receptor dependence through epithelial-to-mesenchymal-like transitions. Clin. Exp. Metastasis 25, 685–693 (2008).
Guix, M. et al. Acquired resistance to EGFR tyrosine kinase inhibitors in cancer cells is mediated by loss of IGF-binding proteins. J. Clin. Invest. 118, 2609–2619 (2008).
Zakowski, M. F., Ladanyi, M. & Kris, M. G. EGFR mutations in small-cell lung cancers in patients who have never smoked. N. Engl. J. Med. 355, 213–215 (2006).
Morinaga, R. et al. Sequential occurrence of non-small cell and small cell lung cancer with the same EGFR mutation. Lung Cancer 58, 411–413 (2007).
Tatematsu, A. et al. Epidermal growth factor receptor mutations in small cell lung cancer. Clin. Cancer Res. 14, 6092–6096 (2008).
Janne, P. A. et al. Preliminary activity and safety results from a phase I clinical trial of PF-00299804, an irreversible pan-HER inhibitor, in patients (pts) with NSCLC. J. Clin. Oncol. Abstr. 8027 (2010).
Dziadziuszko, R., Camidge, D. R. & Hirsch, F. R. The insulin-like growth factor pathway in lung cancer. J. Thorac. Oncol. 3, 815–818 (2008).
Pao, G. & Girard, N. New driver mutations in non-small-cell lung cancer. Lancet Oncol. (in the press)
Kwak, E. L. et al. Irreversible inhibitors of the EGF receptor may circumvent acquired resistance to gefitinib. Proc. Natl Acad. Sci. USA 102, 7665–7670 (2005).
Carter, T. A. et al. Inhibition of drug-resistant mutants of ABL, KIT, and EGF receptor kinases. Proc. Natl Acad. Sci. USA 102, 11011–11016 (2005).
Sharma, S. V., Bell, D. W., Settleman, J. & Haber, D. A. Epidermal growth factor receptor mutations in lung cancer. Nature Rev. Cancer 7, 169–181 (2007).
Wong, K. K. et al. A phase I study with neratinib (HKI-272), an irreversible pan ErbB receptor tyrosine kinase inhibitor, in patients with solid tumors. Clin. Cancer Res. 15, 2552–2558 (2009).
Sequist, L. V. et al. Neratinib, an irreversible pan-ErbB receptor tyrosine kinase inhibitor: results of a Phase II trial in patients with advanced non-small-cell lung cancer. J. Clin. Oncol. 28, 3076–3083 (2010).
Li, D. et al. BIBW2992, an irreversible EGFR/HER2 inhibitor highly effective in preclinical lung cancer models. Oncogene 27, 4702–4711 (2008).
Engelman, J. A. et al. PF00299804, an irreversible pan-ERBB inhibitor, is effective in lung cancer models with EGFR and ERBB2 mutations that are resistant to gefitinib. Cancer Res. 67, 11924–11932 (2007).
Gonzales, A. J. et al. Antitumor activity and pharmacokinetic properties of PF-00299804, a second-generation irreversible pan-erbB receptor tyrosine kinase inhibitor. Mol. Cancer Ther. 7, 1880–1889 (2008).
Ogino, A. et al. Emergence of epidermal growth factor receptor T790M mutation during chronic exposure to gefitinib in a non small cell lung cancer cell line. Cancer Res. 67, 7807–7814 (2007).
Godin-Heymann, N. et al. The T790M 'gatekeeper' mutation in EGFR mediates resistance to low concentrations of an irreversible EGFR inhibitor. Mol. Cancer Ther. 7, 874–879 (2008). This paper shows that despite being more potent than gefitinib, the irreversible EGFR inhibitor HKI-272 actually selects for T790M-mediated resistance in vitro , suggesting that this class of compounds may not be as effective as hoped unless high concentrations can be achieved in patients.
Ercan, D. et al. Amplification of EGFR T790M causes resistance to an irreversible EGFR inhibitor. Oncogene 29, 2346–2356 (2010).
Zhou, W. et al. Novel mutant-selective EGFR kinase inhibitors against EGFR T790M. Nature 462, 1070–1074 (2009). This work reports the identification and characterization of WZ4002, a novel EGFR inhibitor with potent activity against EGFR-T790M-harbouring receptors.
Li, D. et al. Bronchial and peripheral murine lung carcinomas induced by T790M-L858R mutant EGFR respond to HKI-272 and rapamycin combination therapy. Cancer Cell 12, 81–93 (2007).
Comoglio, P. M., Giordano, S. & Trusolino, L. Drug development of MET inhibitors: targeting oncogene addiction and expedience. Nature Rev. Drug Discov. 7, 504–516 (2008).
Foo, J. & Michor, F. Evolution of resistance to targeted anti-cancer therapies during continuous and pulsed administration strategies. PLoS Comput. Biol. 5, e1000557 (2009).
Shah, N. P. et al. Potent, transient inhibition of BCR–ABL with dasatinib 100 mg daily achieves rapid and durable cytogenetic responses and high transformation-free survival rates in chronic phase chronic myeloid leukemia patients with resistance, suboptimal response or intolerance to imatinib. Haematologica 95, 232–240 (2010).
Schiller, J. H. et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N. Engl. J. Med. 346, 92–98 (2002).
Sandler, A. et al. Paclitaxel–carboplatin alone or with bevacizumab for non-small-cell lung cancer. N. Engl. J. Med. 355, 2542–2550 (2006).
Amado, R. G. et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J. Clin. Oncol. 26, 1626–1634 (2008).
Van Cutsem, E. et al. Cetuximab and chemotherapy as initial treatment for metastatic colorectal cancer. N. Engl. J. Med. 360, 1408–1417 (2009).
Eberhard, D. A. et al. Mutations in the epidermal growth factor receptor and in KRAS are predictive and prognostic indicators in patients with non-small-cell lung cancer treated with chemotherapy alone and in combination with erlotinib. J. Clin. Oncol. 23, 5900–5909 (2005).
Kim, E. S. et al. Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): a randomised Phase III trial. Lancet 372, 1809–1818 (2008).
Gatzemeier, U. et al. Phase III study of erlotinib in combination with cisplatin and gemcitabine in advanced non-small-cell lung cancer: the Tarceva Lung Cancer Investigation Trial. J. Clin. Oncol. 25, 1545–1552 (2007).
Zhu, C. Q. et al. Role of KRAS and EGFR as biomarkers of response to erlotinib in National Cancer Institute of Canada Clinical Trials Group Study BR.21. J. Clin. Oncol. 26, 4268–4275 (2008).
Bell, D. W. et al. Epidermal growth factor receptor mutations and gene amplification in non-small-cell lung cancer: molecular analysis of the IDEAL/INTACT gefitinib trials. J. Clin. Oncol. 23, 8081–8092 (2005).
Shah, N. P. et al. Multiple BCR–ABL kinase domain mutations confer polyclonal resistance to the tyrosine kinase inhibitor imatinib (STI571) in chronic phase and blast crisis chronic myeloid leukemia. Cancer Cell 2, 117–125 (2002).
Branford, S. et al. Detection of BCR–ABL mutations in patients with CML treated with imatinib is virtually always accompanied by clinical resistance, and mutations in the ATP phosphate-binding loop (P-loop) are associated with a poor prognosis. Blood 102, 276–283 (2003).
Antonescu, C. R. et al. Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin. Cancer Res. 11, 4182–4190 (2005).
Wardelmann, E. et al. Polyclonal evolution of multiple secondary KIT mutations in gastrointestinal stromal tumors under treatment with imatinib mesylate. Clin. Cancer Res. 12, 1743–1749 (2006).
Cowan-Jacob, S. W. et al. Structural biology contributions to the discovery of drugs to treat chronic myelogenous leukaemia. Acta Crystallogr. D Biol. Crystallogr. 63, 80–93 (2007).
Mol., C. D. et al. Structural basis for the autoinhibition and STI-571 inhibition of c-Kit tyrosine kinase. J. Biol. Chem. 279, 31655–31663 (2004).
Pettersen, E. F. et al. UCSF Chimera — a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Giaccone, G. et al. Gefitinib in combination with gemcitabine and cisplatin in advanced non-small-cell lung cancer: a Phase III trial — INTACT 1. J. Clin. Oncol. 22, 777–784 (2004).
Herbst, R. S. et al. Gefitinib in combination with paclitaxel and carboplatin in advanced non-small-cell lung cancer: a Phase III trial —I NTACT 2. J. Clin. Oncol. 22, 785–794 (2004).
Herbst, R. S. et al. TRIBUTE: a Phase III trial of erlotinib hydrochloride (OSI-774) combined with carboplatin and paclitaxel chemotherapy in advanced non-small-cell lung cancer. J. Clin. Oncol. 23, 5892–5899 (2005).
Pirker, R. et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an open-label randomised Phase III trial. Lancet 373, 1525–1531 (2009).
Wakeling, A. E. et al. ZD1839 (Iressa): an orally active inhibitor of epidermal growth factor signaling with potential for cancer therapy. Cancer Res. 62, 5749–5754 (2002).
Hidalgo, M. et al. Phase I and pharmacologic study of OSI-774, an epidermal growth factor receptor tyrosine kinase inhibitor, in patients with advanced solid malignancies. J. Clin. Oncol. 19, 3267–3279 (2001).
Gendreau, S. B. et al. Inhibition of the T790M gatekeeper mutant of the epidermal growth factor receptor by EXEL-7647. Clin. Cancer Res. 13, 3713–3723 (2007).
Ryan, A. J. & Wedge, S. R. ZD6474 — a novel inhibitor of VEGFR and EGFR tyrosine kinase activity. Br. J. Cancer 92 (Suppl. 1), 6–13 (2005).
Xia, W. et al. Anti-tumor activity of GW572016: a dual tyrosine kinase inhibitor blocks EGF activation of EGFR/erbB2 and downstream Erk1/2 and AKT pathways. Oncogene 21, 6255–6263 (2002).
Erlichman, C. et al. Phase I study of EKB-569, an irreversible inhibitor of the epidermal growth factor receptor, in patients with advanced solid tumors. J. Clin. Oncol. 24, 2252–2260 (2006).
Rabindran, S. K. et al. Antitumor activity of HKI-272, an orally active, irreversible inhibitor of the HER-2 tyrosine kinase. Cancer Res. 64, 3958–3965 (2004).
Allen, L. F., Eiseman, I. A., Fry, D. W. & Lenehan, P. F. CI-1033, an irreversible pan-erbB receptor inhibitor and its potential application for the treatment of breast cancer. Semin. Oncol. 30, 65–78 (2003).
Acknowledgements
The authors would like to apologize to those whose work was not included owing to space constraints. They thank C. Lovly, K. Politi and M. R. Brewer for their insightful feedback on the manuscript, and M. R. Brewer for assistance with the crystal structure diagrams. This work was supported by US National Institutes of Health (NIH) National Cancer Institute (NCI) grants R01-CA121210, P01-CA129243 and U54-CA143798. W.P. received additional support from Vanderbilt's Specialized Program of Research Excellence in Lung Cancer grant (CA90949) and the Vanderbilt-Ingram Cancer Center (VICC) core grant (P30-CA68485).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Rights to EGFR T790M testing were licensed on behalf of William Pao and others to Molecular MD. William Pao has received research funding from Exelixis and has consulted for Molecular MD, AstraZeneca, Bristol-Myers Squibb and Symphony Evolution.
Related links
Glossary
- Driver mutation
-
An oncogenic mutation that induces and sustains tumorigenesis.
- Oncogene addiction
-
The phenomenon in which cancer cells become dependent on or addicted to signalling from oncogenic mutants for survival.
- Primary resistance
-
The initial resistance to therapy.
- Gefitinib
-
The first quinazoline-based reversible small-molecule EGFR tyrosine kinase inhibitor.
- Erlotinib
-
An FDA-approved quinazoline-based EGFR inhibitor.
- Prospective single-arm study
-
A clinical trial in which a drug is administered in a prospective manner to a single group of patients (defined by certain characteristics) to see whether their condition improves. Single-arm studies are distinct from two-arm studies, in which a group of patients is randomly administered one of two possible treatments (for example, an experimental treatment versus standard treatment) to determine which treatment is better.
- Response rate
-
RR. The proportion of patients undergoing a documented radiographic response as determined by response evaluation criteria in solid tumours.
- Progression-free survival
-
PFS. The length of time during and after treatment in which a disease does not progress.
- Time to progression
-
TTP. Time from the beginning of treatment until treatment failure.
- Never smoker
-
An individual who has smoked <100 cigarettes in their lifetime.
- Former light smoker
-
An individual who has stopped smoking for at least 15 years previously and has a total of ≤10 pack-years of smoking.
- Carboplatin–paclitaxel
-
An example of a platinum doublet for first-line treatment of NSCLC.
- Hazard ratio
-
HR. A measure of how often an event happens in one group compared with how often it happens in another group.
- Confidence interval
-
CI. A calculated value that shows the range in which a particular treatment effect is likely to be observed.
- Chimeric IgG monoclonal antibody
-
A recombinant antibody made from two species (in the case of cetuximab, the fusion contains human and mouse sequences).
- Acquired resistance
-
Resistance that develops after the initial response to therapy.
- Gatekeeper residue
-
A conserved residue that lies at the opening of the ATP-binding pocket in several kinases.
- Disease flare
-
Rapid tumour growth following withdrawal of therapy.
- Resistance mutation screen
-
A comprehensive cell-based screen to identify all potential mutations in a target gene that confer resistance to a given agent.
- Irreversible EGFR inhibitor
-
A small-molecule inhibitor that binds permanently in the ATP-binding pocket of EGFR through a covalent bond at C797.
- Chemogenomic profiling
-
The technique of coupling chemical compound sensitivity to genomic signatures.
- Quinazoline core
-
A scaffold built on the fusion of a benzene ring and a pyrimidine ring.
- Anilinopyrimidine core
-
A scaffold built on an anilino group and pyrimidine ring.
Rights and permissions
About this article
Cite this article
Pao, W., Chmielecki, J. Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer. Nat Rev Cancer 10, 760–774 (2010). https://doi.org/10.1038/nrc2947
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc2947
This article is cited by
-
The impact of previous extrapulmonary malignancies on survival of surgically treated primary lung cancer: a comprehensive systematic review and meta-analysis
Updates in Surgery (2023)
-
Real-life comparison of afatinib and erlotinib in non-small cell lung cancer with rare EGFR exon 18 and exon 20 mutations: a Turkish Oncology Group (TOG) study
Journal of Cancer Research and Clinical Oncology (2023)
-
Clinical efficacy of osimertinib in EGFR-mutant non-small cell lung cancer with distant metastasis
BMC Cancer (2022)
-
Itraconazole and rifampicin, as CYP3A modulators but not P-gp modulators, affect the pharmacokinetics of almonertinib and active metabolite HAS-719 in healthy volunteers
Acta Pharmacologica Sinica (2022)
-
CRL2KLHDC3 mediates p14ARF N-terminal ubiquitylation degradation to promote non-small cell lung carcinoma progression
Oncogene (2022)