Key Points
-
Angiogenesis and lymphangiogenesis have important roles in cancer progression: angiogenesis, the growth of new blood vessels, promotes tumour growth and tumour metastasis, and lymphangiogenesis, the growth of new lymphatic vessels, promotes tumour metastasis. Angiogenesis and lymphangiogenesis are regulated by integrins, which are members of a family of cell surface adhesion receptors.
-
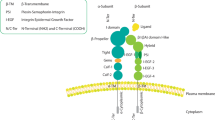
The integrin family is an extensive group of structurally related receptors for extracellular matrix proteins and immunoglobulin superfamily molecules. Integrin ligation promotes intracellular signal transduction, cell migration and survival in angiogenesis and lymphangiogenesis.
-
A number of endothelial cell integrins regulate angiogenesis in diverse manners, including integrins α1β1, α2β1, α4β1, α5β1, α9β1 and α6β4. αv integrins are also important in angiogenesis, although the exact nature of these roles is hotly disputed. Expression and function analysis of αv integrins in wild-type animals using integrin antagonists as well as analysis of knock-in mutant mice indicate that αv integrins promote angiogenesis, whereas genetic deletion studies suggest that αv integrins are not required for angiogenesis.
-
Although less is known about the integrins that regulate lymphangiogenesis, integrin α9 is required for normal developmental lymphangiogenesis. Integrins α4β1, α2β1 and α1β1 have also been implicated in the regulation of tumour lymphangiogenesis.
-
Integrins on bone marrow-derived myeloid cells can also promote angiogenesis. Circulating bone marrow-derived cells migrate into tumours in response to tumour-secreted chemokines and cytokines and integrins α4β1 and αMβ2 (CD11b) have key roles in this process, indirectly influencing tumour angiogenesis.
-
Antagonists of several integrins, including αvβ3, αvβ5 and α5β1, are currently under investigation as clinical agents to suppress tumour angiogenesis and growth either alone or in combination with current cancer therapeutics.
Abstract
Blood vessels promote tumour growth, and both blood and lymphatic vessels facilitate tumour metastasis by serving as conduits for the transport of tumour cells to new sites. Angiogenesis and lymphangiogenesis are regulated by integrins, which are members of a family of cell surface receptors whose ligands are extracellular matrix proteins and immunoglobulin superfamily molecules. Select integrins promote endothelial cell migration and survival during angiogenesis and lymphangiogenesis, whereas other integrins promote pro-angiogenic macrophage trafficking to tumours. Several integrin-targeted therapeutic agents are currently in clinical trials for cancer therapy. Here, we review the evidence implicating integrins as a family of fundamental regulators of angiogenesis and lymphangiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Carmeliet, P. Angiogenesis in life, disease and medicine, Nature 42, 932–936 (2005).
Adams, R. H. & Alitalo, K. Molecular regulation of angiogenesis and lymphangiogenesis. Nature Rev. Mol. Cell Biol. 8, 464–478 (2007).
Lyden, D. et al. Impaired recruitment of bone-marrow-derived endothelial and hematopoietic precursor cells blocks tumor angiogenesis and growth. Nature Med. 11, 1194–1201 (2001). This article established the concept that bone marrow-derived cells in lung and other tissues could help create an environment that attracts metastatic tumour cells.
Schmid, M. C. & Varner, J. A. Myeloid cell trafficking and tumor angiogenesis. Cancer Lett. 250, 1–8 (2007).
Lin, E. Y. & Pollard, J. W. Tumor-associated macrophages press the angiogenic switch in breast cancer. Cancer Res. 67, 5064–5066 (2007). This article shows that macrophages have crucial roles in altering the fate of tumours by secreting pro-angiogenic growth factors.
Ferrara, N. & Kerbel, R. S. Angiogenesis as a therapeutic target. Nature 438, 967–974 (2005).
Roma, A. A. et. al. Peritumoral lymphatic invasion is associated with regional lymph node metastases in prostate adenocarcinoma. Mod. Pathol. 19, 392–398 (2006).
Dadras, S. S. et al. Tumor lymphangiogenesis predicts melanoma metastasis to sentinel lymph nodes. Mod. Pathol. 18, 1232–1242 (2005).
Hirakawa, S. et al. VEGF-A induces tumor and sentinel lymph node lymphangiogenesis and promotes lymphatic metastasis. J. Exp. Med. 201, 1089–1099 (2005). This article showed for the first time that tumors induce lymphangiogenesis not only in the peritumoural space but also in draining lymph nodes.
Hynes, R. O. Integrins: bidirectional, allosteric signaling machines. Cell 110, 673–687 (2002).
Plow, E. F. et al. Ligand binding to integrins. J. Biol. Chem. 275, 21785–21788 (2000).
Haas, T. A. & Plow, E. F. Integrin–ligand interactions: a year in review. Curr Opin Cell Biol. 6, 656–662 (1994).
Komoriya, A. et al. The minimal essential sequence for a major cell type-specific adhesion site (CS1) within the alternatively spliced type III connecting segment domain of fibronectin is leucine–aspartic acid–valine. J. Biol. Chem. 266, 15075–15079 (1991).
Jin, H. & Varner, J. Integrins: roles in cancer development and as treatment targets. Br. J. Cancer 90, 561–565 (2004).
Mitra, S. K., Hanson, D. A. & Schlaepfer, D. D. Focal adhesion kinase: in command and control of cell motility. Nature Rev. Mol. Cell. Biol. 6, 56–68 (2005).
Mitra, S. K & Schlaepfer, D. D. Integrin-regulated FAK–Src signaling in normal and cancer cells. Curr. Opin. Cell Biol. 18, 516–523 (2006).
Stupack, D. G. Integrins as a distinct subtype of dependence receptors. Cell Death Differ. 12, 1021–1030 (2005).
Zhu, J. et al. β8 integrins are required for vascular morphogenesis in mouse embryos. Development 129, 2891–2903 (2002). This article shows that integrin αvb8 is required for the formation of normal brain blood vessels.
Cheresh, D. A. Human endothelial cells synthesize and express an Arg–Gly–Asp-directed adhesion receptor involved in attachment to fibrinogen and von Willebrand factor. Proc. Natl Acad. Sci. USA 84, 6471–6475 (1987).
Brooks, P. C., Clark, R. A. & Cheresh, D. A. Requirement of vascular integrin αvβ3 for angiogenesis. Science 264, 569–571 (1994). This is the first article demonstrating a role for an integrin in angiogenesis.
Brooks, P. C. et al. Integrin αvβ3 antagonists promote tumor regression by inducing apoptosis of angiogenic blood vessels. Cell 79, 1157–1164 (1994).
Brooks, P. C. et al. Antiintegrin αvβ3 blocks human breast cancer growth and angiogenesis in human skin. J. Clin. Invest. 96, 1815–1822 (1995).
Friedlander, M. et al. Definition of two angiogenic pathways by distinct αv integrins. Science 270, 1500–1502 (1995). This article established that two unique pathways of angiogenesis are regulated by two distinct αv integrins.
Friedlander, M. et al. Involvement of integrins αvβ3 and αvβ5 in ocular neovascular diseases. Proc. Natl Acad. Sci. USA 93, 9764–9769 (1996).
Friedlander, M. et al. Angiogenesis inhibition and choroidal neovascularization suppression by sustained delivery of an integrin antagonist, EMD478761. Invest. Ophthalmol. Vis. Sci. 48, 5184–5190 (2007).
Drake, C. J., Cheresh, D. A. & Little, C. D. An antagonist of integrin αvβ3 prevents maturation of blood vessels during embryonic neovascularization. J. Cell Sci. 108, 2655–2661 (1995).
Strömblad, S., Becker, J. C., Yebra, M., Brooks, P. C. & Cheresh. D. A. Suppression of p53 activity and p21WAF1/CIP1 expression by vascular cell integrin αvβ3 during angiogenesis. J. Clin. Invest. 98, 426–433 (1996).
Stupack, D. G., Puente, X. S., Boutsaboualoy, S., Storgard, C. M. & Cheresh, D. A. Apoptosis of adherent cells by recruitment of caspase-8 to unligated integrins. J. Cell Biol. 155, 459–470 (2001). This article established the concept of integrin-mediated death by showing that unligated integrins promote cell death.
Eliceiri, B. P, Klemke, R., Strömblad, S. & Cheresh, D. A. Integrin αvβ3 requirement for sustained mitogen-activated protein kinase activity during angiogenesis. J. Cell Biol. 140, 1255–1263 (1998).
Eliceiri, B. P. et al. Src-mediated coupling of focal adhesion kinase to integrin αvβ5 in vascular endothelial growth factor signaling. J. Cell Biol. 157, 149–160 (2002).
Seker, A. et al. Expression of integrins in cerebral arteriovenous and cavernous malformations. Neurosurgery 58, 159–168 (2006).
Alon, T. et al. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nature Med. 1, 1024–1028 (1995).
Weis, S., Cui, J., Barnes, L. & Cheresh, D. Endothelial barrier disruption by VEGF-mediated Src activity potentiates tumor cell extravasation and metastasis. J. Cell Biol. 167, 223–232 (2004).
Criscuoli, M. L., Nguyen, M. & Eliceiri, B. P. Tumor metastasis but not tumor growth is dependent on Src-mediated vascular permeability. Blood 105, 1508–1514 (2005).
Hodivala-Dilke, K. M. et al. β3-integrin-deficient mice are a model for Glanzmann thrombasthenia showing placental defects and reduced survival. J. Clin. Invest. 103, 229–238 (1999).
Weis, S. M. et al. Cooperation between VEGF and β3 integrin during cardiac vascular development. Blood 109, 1962–1970 (2007).
Reynolds, L. E. et al. Enhanced pathological angiogenesis in mice lacking β3 integrin or β3 and β5 integrins. Nature Med. 8, 27–34 (2002).
Reynolds, A. R. et al. Elevated Flk1 (vascular endothelial growth factor receptor 2) signaling mediates enhanced angiogenesis in β3-integrin-deficient mice. Cancer Res. 64, 8643–8650 (2004). This study established the concept that developmental loss of an integrin could lead to enhanced angiogenesis through compensatory mechanisms.
Huang, X., Griffiths, M., Wu, J., Farese, R. V. Jr & Sheppard, D. Normal development, wound healing, and adenovirus susceptibility in β5-deficient mice. Mol. Cell Biol. 20, 755–759 (2000). This article showed that loss of αvb5 during development has no significant effect on angiogenesis.
Bader, B. L., Rayburn, H., Crowley, D. & Hynes, R. O. Extensive vasculogenesis, angiogenesis, and organogenesis precede lethality in mice lacking all αv integrins. Cell 95, 507–519 (1998). This study shows that αv integrins are essential for development in most animals but that some animals can survive in ovo loss of αv integrins until the early post-natal period.
McCarty, J. H. et al. Selective ablation of α v integrins in the central nervous system leads to cerebral hemorrhage, seizures, axonal degeneration and premature death. Development 132, 165–176 (2005).
McCarty, J. H. et al. Defective associations between blood vessels and brain parenchyma lead to cerebral hemorrhage in mice lacking αv integrins. Mol. Cell. Biol. 22, 7667–7677 (2002).
Mahabeleshwar, G. H., Feng, W., Phillips, D. R. & Byzova, T. V. Integrin signaling is critical for pathological angiogenesis. J. Exp. Med. 203, 2495–2507 (2006). This study shows that animals with an intact but non-functional β3 integrin exhibit defective angiogenesis.
Montgomery, A. M., Reisfeld, R. A. & Cheresh, D. A. Integrin αvβ3 rescues melanoma cells from apoptosis in three-dimensional dermal collagen. Proc. Natl Acad. Sci. USA 91, 8856–8860 (1994).
Clark, R. A. et al. Blood vessel fibronectin increases in conjunction with endothelial cell proliferation and capillary ingrowth during wound healing. J. Invest. Dermatol. 79, 269–276 (1982).
Kim, S., Bell, K., Mousa, S. A. & Varner, J. A. Regulation of angiogenesis in vivo by ligation of integrin α5β1 with the central cell-binding domain of fibronectin. Am. J. Pathol. 156, 1345–1362 (2000). This is the first article showing a role for integrin α5b1 in angiogenesis.
Liao, Y. F., Gotwals, P. J., Koteliansky, V. E., Sheppard, D. & Van De Water, L. The EIIIA segment of fibronectin is a ligand for integrins α9β1 and α4β1 providing a novel mechanism for regulating cell adhesion by alternative splicing. J. Biol. Chem. 277, 14467–14474 (2002).
George, E. L., Baldwin, H. S. & Hynes, R. O. Fibronectins are essential for heart and blood vessel morphogenesis but are dispensable for initial specification of precursor cells. Blood 90, 3073–3081 (1997).
Astrof, S., Crowley, D. & Hynes, R. O. Multiple cardiovascular defects caused by the absence of alternatively spliced segments of fibronectin. Dev. Biol. 311, 11–24 (2007).
Fassler, R. & Meyer, M. Consequences of lack of β1 integrin gene expression in mice. Genes Dev. 9, 1896–1908 (1995).
Stephens, L. E. et al. Deletion of β1 integrins in mice results in inner cell mass failure and peri-implantation lethality. Genes Dev. 9, 1883–1895 (1995).
Tanjore, H., Zeisberg, E. M., Gerami-Naini, B. & Kalluri, R. β1 integrin expression on endothelial cells is required for angiogenesis but not for vasculogenesis. Dev. Dyn. 237, 75–82 (2007).
Bloch, W. et al. β1 integrin is essential for teratoma growth and angiogenesis. J. Cell Biol. 139, 265–278 (1997).
Muether, P. S. et al. The role of integrin α5β1 in the regulation of corneal neovascularization. Exp. Eye Res. 85, 356–365 (2007).
Boudreau, N. J. & Varner, J. A. The homeobox transcription factor Hox D3 promotes integrin α5β1 expression and function during angiogenesis. J. Biol. Chem. 279, 4862–4868 (2004).
Umeda, N. et al. Suppression and regression of choroidal neovascularization by systemic administration of an α5β1 integrin antagonist. Mol. Pharmacol. 69, 1820–1828 (2006).
Kim, S., Harris, M. & Varner, J. A. Regulation of integrin αvβ3-mediated endothelial cell migration and angiogenesis by integrin α5β1 and protein kinase A. J. Biol. Chem. 275, 33920–33928 (2000).
Kim, S., Bakre, M., Yin, H. & Varner, J. A. Inhibition of endothelial cell survival and angiogenesis by protein kinase, A. J. Clin. Invest. 110, 933–941 (2002).
Yang, J. T., Rayburn, H. & Hynes, R. O. Embryonic mesodermal defects in α5 integrin-deficient mice. Development 119, 1093–1105 (1993). This article shows that integrin α5 is required during embryonic development of early blood vessels and other tissues.
Taverna, D. & Hynes, R. O. Reduced blood vessel formation and tumor growth in α5-integrin-negative teratocarcinomas and embryoid bodies. Cancer Res. 61, 5255–5261 (2001).
Francis, S. E. et al. Central roles of α5β1 integrin and fibronectin in vascular development in mouse embryos and embryoid bodies. Arterioscler. Thromb. Vasc. Biol. 22, 927–933 (2002).
Yang, J. T., Rayburn, H. & Hynes, R. O. Cell adhesion events mediated by α 4 integrins are essential in placental and cardiac development. Development 121, 549–560 (1995).
Garmy-Susini, B. et al. Integrin α4β1-VCAM-1-mediated adhesion between endothelial and mural cells is required for blood vessel maturation. J. Clin. Invest. 115, 1542–1551 (2005). This article demonstrates that integrin α4β1 on endothelium promotes endothelial cell motility and angiogenesis as well as a transient association of pericytes with endothelium.
Vlahakis, N. E. et al. Integrin α9β1 directly binds to vascular endothelial growth factor (VEGF)-A and contributes to VEGF-A-induced angiogenesis. J. Biol. Chem. 282, 15187–15196 (2007).
Staniszewska, I. et al. Interaction of α9β1 integrin with thrombospondin-1 promotes angiogenesis. Circ. Res. 100, 1308–1316 (2007).
Liao, Y. F. et al. The EIIIA segment of fibronectin is a ligand for integrins α9β1 and α4β1 providing a novel mechanism for regulating cell adhesion by alternative splicing. J. Biol. Chem. 277, 14467–14474 (2002).
Marcinkiewicz, C. et al. Inhibitory effects of MLDG-containing heterodimeric disintegrins reveal distinct structural requirements for interaction of the integrin α9β1 with VCAM-1, tenascin-C, and osteopontin. J. Biol. Chem. 275, 31930–31937 (2000).
Huang, X. Z. et al. Fatal bilateral chylothorax in mice lacking the integrin α9β1. Mol. Cell. Biol. 20, 5208–5215 (2000). This study demonstrates that integrin α9b1 is required for proper development of the lymphatic system.
Senger, D. R. et al. Angiogenesis promoted by vascular endothelial growth factor: regulation through α1β1 and α2β1 integrins. Proc. Natl Acad. Sci. USA 94, 13612–13617 (1997).
Pozzi, A. et al. Elevated matrix metalloprotease and angiostatin levels in integrin α1-knockout mice cause reduced tumor vascularization. Proc. Natl Acad. Sci. USA 97, 2202–2207 (2000).
Zhang, Z. et al. α2β1 integrin expression in the tumor microenvironment enhances tumor angiogenesis in a tumor-cell specific manner. Blood 111, 1980–1988 (2008). This paper shows that integrin α2b1-null mice exhibit distinct tumour growth patterns that are dependent upon the growth factors that are intrinsically expressed by individual tumour cells.
Lee, T. H. et al. Integrin regulation by vascular endothelial growth factor in human brain microvascular endothelial cells: role of α6β1 integrin in angiogenesis. J. Biol. Chem. 281, 40450–40460 (2006).
Nikolopoulos, S. N., Blaikie, P., Yoshioka, T., Guo, W. & Giancotti, F. G. Integrin β4 signaling promotes tumor angiogenesis. Cancer Cell 6, 471–483 (2004). This paper shows the important role of integrin α6b4 in angiogenesis.
van der Neut, R., Krimpenfort, P., Calafat, J., Niessen, C. M. & Sonnenberg, A. Epithelial detachment due to absence of hemidesmosomes in integrin β4-null mice. Nature Genet. 13, 366–369 (1996).
Georges-Labouesse, E. et al. Absence of integrin α6 leads to epidermolysis bullosa and neonatal death in mice. Nature Genet. 13, 370–373 (1996).
Dowling, J., Yu, Q. C. & Fuchs, E. β4 integrin is required for hemidesmosome formation, cell adhesion and cell survival. J. Cell Biol. 134, 559–572 (1996).
Dans, M., et al. Tyrosine phosphorylation of the β4 integrin cytoplasmic domain mediates Shc signaling to extracellular signal-regulated kinase and antagonizes formation of hemidesmosomes. J. Biol. Chem. 276, 1494–1502 (2001).
Leu, S. J. et al. Identification of a novel integrin α6β1 binding site in the angiogenic inducer CCN1 (CYR61). J. Biol. Chem. 278, 33801–33808 (2003).
Jin, H. et al. A homing mechanism for bone marrow-derived progenitor cell recruitment to the neovasculature. J. Clin. Invest. 116, 652–662 (2006). This paper shows that integrin α4b1 on bone marrow-derived cells promotes monocyte and endothelial precursor cell homing to tumours.
Jin, H., Su, J., Garmy-Susini, B., Kleeman, J. & Varner, J. Integrin α4β1 promotes monocyte trafficking and angiogenesis in tumors. Cancer Res. 66, 2146–2152 (2006).
Wu, Y. et al. Essential role of ICAM-1/CD18 in mediating EPC recruitment, angiogenesis, and repair to the infarcted myocardium. Circ. Res. 99, 315–322 (2006).
Chavakis, E. et al. Role of β2-integrins for homing and neovascularization capacity of endothelial progenitor cells. J. Exp. Med. 1, 63–72 (2005).
Varner, J. A. The sticky truth about angiogenesis and thrombospondins. J. Clin. Invest. 116, 3111–3113 (2006).
Kopp, H. G. & Rafii, S. Thrombopoietic cells and the bone marrow vascular niche. Ann. NY Acad. Sci. 1106, 175–179 (2007).
Banerji, S. et al. LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J. Cell Biol. 144, 789–801 (1999).
Wigle, J. T. & Oliver, G. Prox1 function is required for the development of the murine lymphatic system. Cell 98, 769–778 (1999).
Breiteneder-Geleff, S. et al. Angiosarcomas express mixed endothelial phenotypes of blood and lymphatic capillaries: podoplanin as a specific marker for lymphatic endothelium. Am. J. Pathol. 154, 385–394 (1999).
Mishima, K. et al. Prox1 induces lymphatic endothelial differentiation via integrin α9 and other signaling cascades. Mol. Biol. Cell. 18, 1421–1429 (2007).
Vlahakis, N. E., Young, B. A., Atakilit, A. & Sheppard, D. The lymphangiogenic vascular endothelial growth factors VEGF-C and -D are ligands for the integrin α9β1. J. Biol. Chem. 280, 4544–4552 (2005).
Hong, Y. K. et al. VEGF-A promotes tissue repair-associated lymphatic vessel formation via VEGFR-2 and the α1β1 and α2β1 integrins. FASEB J. 18, 1111–1113 (2004). This article established the role of integrins α1b1 and α2b1 in lymphangiogenesis.
Dietrich, T. et al. Inhibition of inflammatory lymphangiogenesis by integrin α5 blockade. Am. J. Pathol. 171, 361–372 (2007).
Garmy-Susini, B., Makale, M., Fuster, M. & Varner, J. A. Methods to study lymphatic vessel integrins. Methods Enzymol. 426, 415–438 (2007).
Gutheil, J. C. et al. Targeted antiangiogenic therapy for cancer using Vitaxin: a humanized monoclonal antibody to the integrin αvβ3. Clin. Cancer Res. 6, 3056–3061 (2000).
McNeel, D. G. et al. Phase I trial of a monoclonal antibody specific for αvβ3 integrin (MEDI-522) in patients with advanced malignancies, including an assessment of effect on tumor perfusion. Clin Cancer Res. 11, 7851–7860 (2005).
Hersey, P. et al. A phase II, randomized, open-label study evaluating the antitumor activity of MEDI-522, a humanized monoclonal antibody directed against the human αvβ3 (αvβ3) integrin, ± dacarbazine (DTIC) in patients with metastatic melanoma. J. Clin. Oncol. 2005 ASCO Annu. Meeting Proc. 23, 7507 (2005).
Zhang, D., Pier, T., McNeel, D. G., Wilding, G. & Friedl, A. Effects of a monoclonal anti-αvβ3 integrin antibody on blood vessels — a pharmacodynamic study. Invest. New Drugs 25, 49–55 (2007).
Trikha, M. et al. CNTO 95, a fully human monoclonal antibody that inhibits αv integrins, has antitumor and antiangiogenic activity in vivo. Int. J. Cancer 110, 326–335 (2004).
Martin, P. L. et al. Absence of adverse effects in cynomolgus macaques treated with CNTO 95, a fully human anti-αv integrin monoclonal antibody, despite widespread tissue binding. Clin. Cancer Res. 11, 6959–6965 (2005).
Mullamitha, S. A. et al. Phase I evaluation of a fully human anti-αv integrin monoclonal antibody (CNTO 95) in patients with advanced solid tumors. Clin. Cancer Res. 13, 2128–2135 (2007).
Brooks, P. C. et al. Insulin-like growth factor receptor cooperates with integrin αvβ5 to promote tumor cell dissemination in vivo. J. Clin. Invest. 99, 1390–1398 (1997).
Nabors, L. B. et al. Phase I and correlative biology study of cilengitide in patients with recurrent malignant glioma. J. Clin. Oncol. 25, 1651–1657 (2007).
Albert, J. M. et al. Integrin α v β 3 antagonist Cilengitide enhances efficacy of radiotherapy in endothelial cell and non-small-cell lung cancer models. Int. J. Radiat. Oncol. Biol. Phys. 65, 1536–1543 (2006).
Friess, H. et al. A randomized multi-center phase II trial of the angiogenesis inhibitor Cilengitide (EMD 121974) and gemcitabine compared with gemcitabine alone in advanced unresectable pancreatic cancer. BMC Cancer 6, 285 (2006).
Beekman, K. W. et al. Phase II evaluations of cilengitide in asymptomatic patients with androgen-independent prostate cancer: scientific rationale and study design. Clin. Genitourin. Cancer 4, 299–302 (2006).
Bradley, D. A. et. al. EMD121974 (NSC 707544, cilengitide) in asymptomatic metastatic androgen independent prostate cancer (AIPCa) patients (pts): A randomized trial by the Prostate Cancer Clinical Trials Consortium (NCI 6372). J. Clin. Oncol. 2007 ASCO Annu. Meeting Proc. 25, 5137 (2007).
Figlin, R. A., Kondagunta, G. V., Yazji, S., Motzer, R. J. & Bukowski, R. M. Phase II study of volociximab (M200), an α5β1 anti-integrin antibody in refractory metastatic clear cell renal cell cancer (RCC). J. Clin. Oncol. ASCO Annu. Meeting Proc. 24, 4535 (2006).
Kuwada, S. K. Drug evaluation: Volociximab, an angiogenesis-inhibiting chimeric monoclonal antibody. Curr. Opin. Mol. Ther. 9, 92–98 (2007).
Stoeltzing, O. et al. Inhibition of integrin α5β1 function with a small peptide (ATN-161) plus continuous 5-FU infusion reduces colorectal liver metastases and improves survival in mice. Int. J. Cancer 104, 496–503 (2003).
Cianfrocca, M. E. et al. Phase 1 trial of the antiangiogenic peptide ATN-161 (Ac-PHSCN-NH2), a β integrin antagonist, in patients with solid tumours. Br. J. Cancer 94, 1621–1626 (2006).
Lim, E. H., Danth, N., Bednarski, M. & Li, K. C. A review: Integrin αvβ3-targeted molecular imaging and therapy in angiogenesis. Nanomedicine 1, 110–114 (2005).
Garanger, E., Boturyn, D. & Dumy, P. Tumor targeting with RGD peptide ligands-design of new molecular conjugates for imaging and therapy of cancers. Anticancer Agents Med. Chem. 7, 552–558 (2007).
Sipkins, D. A. et al. Detection of tumor angiogenesis in vivo by αvβ3-targeted magnetic resonance imaging. Nature Med. 4, 623–626 (1998).
Leong-Poi, H. et al. Noninvasive assessment of angiogenesis by ultrasound and microbubbles targeted to αv-integrins. Circulation 107, 455–460 (2003).
Hood, J. D. et al. Tumor regression by targeted gene delivery to the neovasculature. Science 296, 2404–2407 (2002). This landmark paper established the potential of integrin-targeted nanoparticles in cancer therapy.
Arnaout, M. A., Mahalingam, B. & Xiong, J. P. Integrin structure, allostery, and bidirectional signaling. Annu. Rev. Cell Dev. Biol. 21, 381–410 (2005).
Lu, C., Takagi, J. & Springer, T. A. Association of the membrane proximal regions of the α and β subunit cytoplasmic domains constrains an integrin in the inactive state. J. Biol. Chem. 276, 14642–14648 (2001).
Beglova, N., Blacklow, S. C., Takagi, J. & Springer, T. A. Cysteine-rich module structure reveals a fulcrum for integrin rearrangement upon activation. Nature Struct. Biol. 9, 282–287 (2002).
Vinogradova, O. et al. A structural mechanism of integrin αIIbβ3 “inside-out” activation as regulated by its cytoplasmic face. Cell 110, 587–597 (2002).
Grabovsky, V. et al. Subsecond induction of α4 integrin clustering by immobilized chemokines stimulates leukocyte tethering and rolling on endothelial vascular cell adhesion molecule 1 under flow conditions. J. Exp. Med. 192, 495–506 (2000).
O'Toole, T. E. et al. Integrin cytoplasmic domains mediate inside-out signal transduction. J. Cell Biol. 124, 1047–1059 (1994).
Lin, E. Y. et al. Macrophages regulate the angiogenic switch in a mouse model of breast cancer. Cancer Res. 66, 11238–11246 (2006).
Lin, E. Y. & Pollard, J. W. Tumor-associated macrophages press the angiogenic switch in breast cancer. Cancer Res. 67, 5064–5066 (2007).
Peng, X. et al. Overexpression of focal adhesion kinase in vascular endothelial cells promotes angiogenesis in transgenic mice. Cardiovasc. Res. 64, 421–430 (2004).
Shen, T. L. et al. Conditional knockout of focal adhesion kinase in endothelial cells reveals its role in angiogenesis and vascular development in late embryogenesis. J. Cell Biol. 169, 941–952 (2005).
Lai, K. M. & Pawson, T. The ShcA phosphotyrosine docking protein sensitizes cardiovascular signaling in the mouse embryo. Genes Dev. 14, 1132–1145 (2000).
Hüser, M. et al. MEK kinase activity is not necessary for Raf-1 function. EMBO J. 20, 1940–1951 (2001).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Bolat, F. et al. Microvessel density, VEGF expression, and tumor-associated macrophages in breast tumors: correlations with prognostic parameters. J. Exp. Clin. Cancer Res. 25, 365–372 (2006).
Tsutsui, S. et al. Macrophage infiltration and its prognostic implications in breast cancer: the relationship with VEGF expression and microvessel density. Oncol. Rep. 14, 425–431 (2005).
Valkovi´c, T. et al. Correlation between vascular endothelial growth factor, angiogenesis, and tumor-associated macrophages in invasive ductal breast carcinoma. Virchows Arch. 440, 583–588 (2002).
Esposito, I. et al. Inflammatory cells contribute to the generation of an angiogenic phenotype in pancreatic ductal adenocarcinomas. J. Clin. Pathol. 6, 630–636 (2004).
Leek, R. D. & Harris, A. L. Tumor-associated macrophages in breast cancer. J. Mammary Gland Biol. Neoplasia 2, 177–189 (2002).
Nishie, A. Macrophage infiltration and heme oxygenase-1 expression correlate with angiogenesis in human gliomas. Clin. Cancer Res. 5, 1107–1113 (1999).
Yamashiro, S. et al. Tumor-derived monocyte chemoattractant protein-1 induces intratumoral infiltration of monocyte-derived macrophage subpopulation in transplanted rat tumors. Am. J. Pathol. 4, 856–867 (1994).
Lewis, J. S. et al. Expression of vascular endothelial growth factor by macrophages is up-regulated in poorly vascularized areas of breast carcinomas. J. Pathol. 2, 150–158 (2000).
Sica, A. & Bronte, V. Altered macrophage differentiation and immune dysfunction in tumor development. J. Clin. Invest. 117, 1155–1166 (2007).
Jodele, S. et al. The contribution of bone marrow-derived cells to the tumor vasculature in neuroblastoma is matrix metalloproteinase-9 dependent. Cancer Res. 65, 3200–3208 (2005).
Giraudo, E., Inoue, M. & Hanahan, D. An amino-bisphosphonate targets MMP-9-expressing macrophages and angiogenesis to impair cervical carcinogenesis. J. Clin. Invest. 5, 623–633 (2004).
Ueno, T. et al. Significance of macrophage chemoattractant protein-1 in macrophage recruitment, angiogenesis, and survival in human breast cancer. Clin. Cancer Res. 8, 3282–3289 (2000).
Niwa, Y. et al. Correlation of tissue and plasma RANTES levels with disease course in patients with breast or cervical cancer. Clin. Cancer Res. 2, 285–289 (2001).
Murdoch, C., Giannoudis, A. & Lewis, C. E. Mechanisms regulating the recruitment of macrophages into hypoxic areas of tumors and other ischemic tissues. Blood 8, 2224–2234 (2004).
Gerszten, R. E. et al. MCP-1 and IL-8 trigger firm adhesion of monocytes to vascular endothelium under flow conditions. Nature 398, 718–723 (1999).
Goswami, S. et al. Macrophages promote the invasion of breast carcinoma cells via a colony-stimulating factor-1/epidermal growth factor paracrine loop. Cancer Res. 12, 5278–5283 (2005).
Luo, J. L. et al. Nuclear cytokine-activated IKKα controls prostate cancer metastasis by repressing Maspin. Nature 446, 690–694 (2007).
Greten, F. R. et al. IKKβ links inflammation and tumorigenesis in a mouse model of colitis-associated cancer. Cell 118, 285–296 (2004).
Schafer, Z. T. & Brugge, J. S. IL-6 involvement in epithelial cancers. J. Clin. Invest. 117, 3660–3663 (2007).
Sansone, P. et al. Il-6 triggers malignant features in mammospheres from human ductal breast carcinoma and normal mammary gland. J. Clin. Invest. 117, 3988–4002 (2007).
Gao, S. P. et al. Mutations in the EGFR kinase domain mediate STAT3 activation via IL-6 production in human lung adenocarcinomas J. Clin. Invest. 117, 3846–3856 (2007).
Lin, E. Y., Nguyen, A. V., Russell, R. G. & Pollard, J. W. Colony-stimulating factor 1 promotes progression of mammary tumors to malignancy. J. Exp. Med. 6, 727–740 (2001).
Lu, H. et al. LFA-1 is sufficient in mediating neutrophil emigration in Mac-1-deficient mice. J. Clin. Invest. 99, 1340–1350 (1997).
Acknowledgements
This work was supported by NIH grants R01CA83133 and R01CA126820 to J.V. and a postdoctoral research fellowship from the California Breast Cancer Research Program to B.G.-S.
Author information
Authors and Affiliations
Corresponding author
Related links
Related links
DATABASES
National Cancer Institute
National Cancer Institute Drug Dictionary
FURTHER INFORMATION
Glossary
- Peyer's Patches
-
Secondary lymphoid organs, named after the 17th century Swiss anatomist Joseph Conrad Peyer, that are comprised of round lymphoid follicles in the mucosa of the small intestine.
- Angiogenic switch
-
The induction of new blood vessel sprouting at an early time point in tumour development that leads to rapid, exponential tumour growth.
- Chick chorioallantoic membrane
-
A thin, highly vascularized fetal membrane formed by fusion of the chorion and allantois in fertilized chicken eggs that is often used to evaluate pro- and anti-angiogenic agents.
- Choroidal angiogenesis
-
The development of new blood vessels in the highly vascular area of the eye that lies between the retina and the sclera.
- Teratoma
-
A germ cell tumour composed of undifferentiated and differentiated cells derived from the three germ layers: mesoderm, ectoderm and endoderm. Teratomas may include hair, teeth and other complex structures.
- Forskolin
-
A diterpene derived from the Indian coleus plant that raises cAMP levels in cells by activating adenyl cyclase.
- Posterior somites
-
Cuboidal, segmented masses of mesoderm organized in pairs and distributed along the developing neural tube. Posterior somites give rise to the thoracic, lumbar and sacral vertebrae.
- Pericyte
-
A mesenchymal cell precursor to vascular smooth muscle that associates with endothelial cells during angiogenesis and provides support to small capillaries.
- Hemidesmosome
-
An organized adhesive structure on the surface of epithelial cells comprising integrin α6β4 attached on the exterior of the cell to laminin and on the interior of the cells to plectin and cytosolic keratins.
- Alpha granules
-
Endosomes or granules in platelets that contain growth factors such as VEGF, TGFβ and PDGF.
- Humanized antibody
-
A humanized antibody is a synthetic monoclonal antibody comprised of a human antibody backbone fused with the antigen recognition regions of a mouse monoclonal antibody through recombinant DNA techniques, which is developed to eliminate immunogenic sequences.
- Leiomyosarcoma
-
A neoplasm comprising tumour cells arising from smooth muscle (sarcoma) and frequently found in the stomach and small intestines.
- Dacarbazine
-
A chemotherapeutic, DNA-alkylating agent used in the treatment of malignant melanoma and Hodgkin disease.
- PEGylated
-
Covalently modified with poly(ethylene glycol) to make a hydrophobic drug more soluble and to mask a drug from the host immune system.
- Microbubbles
-
Small (≥3 μm) gas-filled bubbles that serve as contrast-enhancing agents in diagnostic medical ultrasound imaging.
Rights and permissions
About this article
Cite this article
Avraamides, C., Garmy-Susini, B. & Varner, J. Integrins in angiogenesis and lymphangiogenesis. Nat Rev Cancer 8, 604–617 (2008). https://doi.org/10.1038/nrc2353
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc2353
This article is cited by
-
The characteristics and the multiple functions of integrin β1 in human cancers
Journal of Translational Medicine (2023)
-
Angiogenic signaling pathways and anti-angiogenic therapy for cancer
Signal Transduction and Targeted Therapy (2023)
-
Recent Research Progress of RGD Peptide–Modified Nanodrug Delivery Systems in Tumor Therapy
International Journal of Peptide Research and Therapeutics (2023)
-
Preclinical evaluation of a dual-receptor targeted tracer [68Ga]Ga-HX01 in 10 different subcutaneous and orthotopic tumor models
European Journal of Nuclear Medicine and Molecular Imaging (2023)
-
Downregulation of ITGA5 inhibits lymphangiogenesis and cell migration and invasion in male laryngeal squamous cell carcinoma
Protoplasma (2023)