Key Points
-
Advanced non-small-cell lung cancer (NSCLC) is the leading cause of cancer-related deaths in the world.
-
Epidermal growth factor receptor (EGFR) is expressed in 50% of NSCLCs, and its expression is correlated with poor prognosis. These two factors make EGFR and its family members prime candidates for the development of targeted therapeutics.
-
Two EGFR-targeting small-molecule inhibitors, gefitinib (Iressa: AstraZeneca, approved in May 2003) and erlotinib (Tarceva: OSI-Genentech, approved in November 2004) received fast-track approval from the US Food and Drug Administration as treatment for patients with advanced NSCLC who had failed to respond to conventional chemotherapy.
-
Early clinical data showed that 10% of patients with NSCLC responded to gefitinib or erlotinib. Although infrequent, the speed and magnitude of clinical responses were unique, as was the fact that they were seen in specific subsets of cases (non-smokers, women, East Asians and patients with adenocarcinomas with bronchioloalveolar histology).
-
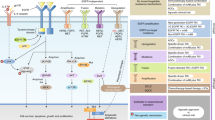
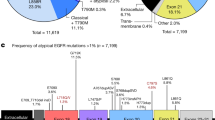
Molecular analysis showed that in most instances, responders harboured specific mutations in the gene that encodes EGFR. Exon 19 mutations characterized by in-frame deletions of amino-acids 747–750 account for 45% of mutations, exon 21 mutations resulting in L858R substitutions account for 40–45% of mutations, and the remaining 10% of mutations involve exon 18 and 20.
-
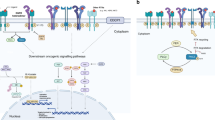
EGFR kinase domain mutations hyperactivate the kinase and confer a dependence on the mutated kinase for the survival of the NSCLC tumour cells.
-
The treatment of sensitive cells with targeted therapeutics such as gefitinib and erlotinib seems to trigger a form of 'oncogenic shock', which is postulated to result from the differential decay of downstream signals leading to a temporary predominance of apoptotic signals.
-
Acquired resistance to gefitinib and erlotinib might involve the recurrent mutation T790M which affects the gatekeeper residue in the catalytic domain of the kinase that weakens the interaction of the inhibitor with its target. Resistance can be overcome in vitro by irreversible inhibitors of EGFR
Abstract
The development and clinical application of inhibitors that target the epidermal growth factor receptor (EGFR) provide important insights for new lung cancer therapies, as well as for the broader field of targeted cancer therapies. We review the results of genetic, biochemical and clinical studies focused on somatic mutations of EGFR that are associated with the phenomenon of oncogene addiction, describing 'oncogenic shock' as a mechanistic explanation for the apoptosis that follows the acute treatment of susceptible cells with kinase inhibitors. Understanding the genetic heterogeneity of epithelial tumours and devising strategies to circumvent their rapid acquisition of resistance to targeted kinase inhibitors are essential to the successful use of targeted therapies in common epithelial cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Brambilla, E., Travis, W. D., Colby, T. V., Corrin, B. & Shimosato, Y. The new World Health Organization classification of lung tumours. Eur. Respir. J. 18, 1059–1068 (2001).
Rapp, E. et al. Chemotherapy can prolong survival in patients with advanced non-small-cell lung cancer — report of a Canadian multicenter randomized trial. J. Clin. Oncol. 6, 633–641 (1988).
Breathnach, O. S. et al. Twenty-two years of phase III trials for patients with advanced non-small-cell lung cancer: sobering results. J. Clin. Oncol. 19, 1734–1742 (2001).
Blackhall, F. H., Shepherd, F. A. & Albain, K. S. Improving survival and reducing toxicity with chemotherapy in advanced non-small cell lung cancer: a realistic goal? Treat Respir. Med. 4, 71–84 (2005).
Blackhall, F., Ranson, M. & Thatcher, N. Where next for gefitinib in patients with lung cancer? Lancet Oncol. 7, 499–507 (2006).
Krause, D. S. & Van Etten, R. A. Tyrosine kinases as targets for cancer therapy. N. Engl. J. Med. 353, 172–187 (2005).
Hynes, N. E. & Lane, H. A. ERBB receptors and cancer: the complexity of targeted inhibitors. Nature Rev. Cancer 5, 341–354 (2005).
Nicholson, R. I., Gee, J. M. & Harper, M. E. EGFR and cancer prognosis. Eur. J. Cancer 37 (Suppl. 4), S9–S15 (2001).
Veale, D., Ashcroft, T., Marsh, C., Gibson, G. J. & Harris, A. L. Epidermal growth factor receptors in non-small cell lung cancer. Br. J. Cancer 55, 513–516 (1987).
Haeder, M. et al. Epidermal growth factor receptor expression in human lung cancer cell lines. Cancer Res. 48, 1132–1136 (1988).
Ohsaki, Y. et al. Epidermal growth factor receptor expression correlates with poor prognosis in non-small cell lung cancer patients with p53 overexpression. Oncol. Rep. 7, 603–607 (2000).
Hirsch, F. R. et al. Epidermal growth factor receptor in non-small-cell lung carcinomas: correlation between gene copy number and protein expression and impact on prognosis. J. Clin. Oncol. 21, 3798–3807 (2003).
Testa, J. R. & Siegfried, J. M. Chromosome abnormalities in human non-small cell lung cancer. Cancer Res. 52, 2702s–2706s (1992).
Putnam, E. A. et al. Autocrine growth stimulation by transforming growth factor-α in human non-small cell lung cancer. Surg. Oncol. 1, 49–60 (1992).
Rusch, V. et al. Differential expression of the epidermal growth factor receptor and its ligands in primary non-small cell lung cancers and adjacent benign lung. Cancer Res. 53, 2379–2385 (1993).
Mendelsohn, J. Epidermal growth factor receptor as a target for therapy with antireceptor monoclonal antibodies. J. Natl Cancer Inst. Monogr. 125–131 (1992).
Giaccone, G. Epidermal growth factor receptor inhibitors in the treatment of non-small-cell lung cancer. J. Clin. Oncol. 23, 3235–3242 (2005).
Barker, A. J. et al. Studies leading to the identification of ZD1839 (IRESSA): an orally active, selective epidermal growth factor receptor tyrosine kinase inhibitor targeted to the treatment of cancer. Bioorg. Med. Chem. Lett. 11, 1911–1914 (2001).
Wakeling, A. E. et al. ZD1839 (Iressa): an orally active inhibitor of epidermal growth factor signaling with potential for cancer therapy. Cancer Res. 62, 5749–5754 (2002).
Lynch, T. J. et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 350, 2129–2139 (2004). Identified mutations in the ATP-binding pocket of EGFR that confer hypersensitivity to gefitinib and translates to a gefitinib response accompanied by tumour shrinkage in patients with NSCLC in a clinical setting.
Paez, J. G. et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304, 1497–1500 (2004). Identified mutations in the kinase domain of EGFR that predict gefitinib response.
Pao, W. et al. EGF receptor gene mutations are common in lung cancers from 'never smokers' and are associated with sensitivity of tumors to gefitinib and erlotinib. Proc. Natl Acad. Sci. USA 101, 13306–13311 (2004).
Sequist, L. V. et al. Epidermal growth factor receptor mutation testing in the care of lung cancer patients. Clin. Cancer Res. 12, 4403s–4408s (2006).
Settleman, J. Mutant kinases as targets for cancer drugs. Drug Discov. Today Dis. Mech. 2, 139–144 (2005).
Kosaka, T. et al. Mutations of the epidermal growth factor receptor gene in lung cancer: biological and clinical implications. Cancer Res. 64, 8919–8923 (2004).
Janne, P. A., Engelman, J. A. & Johnson, B. E. Epidermal growth factor receptor mutations in non-small-cell lung cancer: implications for treatment and tumor biology. J. Clin. Oncol. 23, 3227–3234 (2005).
Shigematsu, H. et al. Clinical and biological features associated with epidermal growth factor receptor gene mutations in lung cancers. J. Natl Cancer Inst. 97, 339–346 (2005).
Han, S. W. et al. Predictive and prognostic impact of epidermal growth factor receptor mutation in non-small-cell lung cancer patients treated with gefitinib. J. Clin. Oncol. 23, 2493–2501 (2005).
Marchetti, A. et al. EGFR mutations in non-small-cell lung cancer: analysis of a large series of cases and development of a rapid and sensitive method for diagnostic screening with potential implications on pharmacologic treatment. J. Clin. Oncol. 23, 857–865 (2005).
Mitsudomi, T. et al. Mutations of the epidermal growth factor receptor gene predict prolonged survival after gefitinib treatment in patients with non-small-cell lung cancer with postoperative recurrence. J. Clin. Oncol. 23, 2513–2520 (2005).
Cappuzzo, F. et al. Epidermal growth factor receptor gene and protein and gefitinib sensitivity in non-small-cell lung cancer. J. Natl Cancer Inst. 97, 643–655 (2005).
Tsao, M. S. et al. Erlotinib in lung cancer- molecular and clinical predictors of outcome. N. Engl J. Med. 353, 133–144 (2005).
Chou, T. Y. et al. Mutation in the tyrosine kinase domain of epidermal growth factor receptor is a predictive and prognostic factor for gefitinib treatment in patients with non-small cell lung cancer. Clin. Cancer Res. 11, 3750–3757 (2005).
Haneda, H. et al. A correlation between EGFR gene mutation status and bronchioloalveolar carcinoma features in Japanese patients with adenocarcinoma. Jpn J. Clin. Oncol. 36, 69–75 (2006).
Kobayashi, S. et al. EGFR mutation and resistance of non-small-cell lung cancer to gefitinib. N. Engl. J. Med. 352, 786–792 (2005). Identified a second site intragenic mutation in EGFR (T790M) that confers acquired resistance to gefitinib in patients with NSCLC that stop responding to gefitinib therapy.
Pao, W. et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PLoS Med. 2, e73 (2005). Identified the T790M mutation in exon 20 of EGFR that confers acquired resistance to EGFR-TKIs.
Kwak, E. L. et al. Irreversible inhibitors of the EGF receptor may circumvent acquired resistance to gefitinib. Proc. Natl Acad. Sci. USA 102, 7665–7670 (2005). In vitro data showing that T790M-mediated acquired resistance to gefitinib can be overcome by irreversible EGFR-TKIs.
Gow, C. H., Shih, J. Y., Chang, Y. L. & Yu, C. J. Acquired gefitinib-resistant mutation of EGFR in a chemonaive lung adenocarcinoma harboring gefitinib-sensitive mutation L858R. PLoS Med. 2, e269 (2005).
Inukai, M. et al. Presence of epidermal growth factor receptor gene T790M mutation as a minor clone in non-small cell lung cancer. Cancer Res. 66, 7854–7858 (2006).
Greulich, H. et al. Oncogenic transformation by inhibitor-sensitive and-resistant EGFR mutants. PLoS Med. 2, e313 (2005).
Ji, H. et al. The impact of human EGFR kinase domain mutations on lung tumorigenesis and in vivo sensitivity to EGFR-targeted therapies. Cancer Cell 9, 485–495 (2006). Showed the relevance of EGFR mutations to the development of NSCLC and response to EGFR-TKIs in a mouse tumour model.
Politi, K. et al. Lung adenocarcinomas induced in mice by mutant EGF receptors found in human lung cancers respond to a tyrosine kinase inhibitor or to down-regulation of the receptors. Genes Dev. 20, 1496–1510 (2006). Modelled NSCLC in mice using transgenic kinase domain mutant EGFR and showed their response to EGFR-TKIs.
Sordella, R., Bell, D. W., Haber, D. A. & Settleman, J. Gefitinib-sensitizing EGFR mutations in lung cancer activate anti-apoptotic pathways. Science 305, 1163–1167 (2004). EGFR kinase domain mutations hyperactivate survival pathways such as Akt and STAT 3, and gefitinib attenuates them in sensitive cell lines.
Jiang, J. et al. Epidermal growth factor-independent transformation of Ba/F3 cells with cancer-derived epidermal growth factor receptor mutants induces gefitinib-sensitive cell cycle progression. Cancer Res. 65, 8968–8974 (2005).
Prudkin, L. & Wistuba, II. Epidermal growth factor receptor abnormalities in lung cancer. Pathogenetic and clinical implications. Ann. Diagn. Pathol. 10, 306–315 (2006).
Kobayashi, S. et al. An alternative inhibitor overcomes resistance caused by a mutation of the epidermal growth factor receptor. Cancer Res. 65, 7096–7101 (2005).
Nagatomo, I. et al. The gefitinib-sensitizing mutant epidermal growth factor receptor enables transformation of a mouse fibroblast cell line. DNA Cell Biol. 25, 246–251 (2006).
Carey, K. D. et al. Kinetic analysis of epidermal growth factor receptor somatic mutant proteins shows increased sensitivity to the epidermal growth factor receptor tyrosine kinase inhibitor, erlotinib. Cancer Res. 66, 8163–8171 (2006).
Mulloy, R. et al. EGF receptor mutants from human lung cancers exhibit enhanced catalytic activity and increased sensitivity to gefitinib. Cancer Res. (in the press).
Fabian, M. A. et al. A small molecule-kinase interaction map for clinical kinase inhibitors. Nature Biotechnol. 23, 329–336 (2005).
Okamoto, I. et al. EGFR mutation in gefitinib-responsive small-cell lung cancer. Ann. Oncol. 17, 1028–1029 (2006).
Gwak, G. Y. et al. Detection of response-predicting mutations in the kinase domain of the epidermal growth factor receptor gene in cholangiocarcinomas. J. Cancer Res. Clin. Oncol. 131, 649–652 (2005).
Schilder, R. J. et al. Phase II study of gefitinib in patients with relapsed or persistent ovarian or primary peritoneal carcinoma and evaluation of epidermal growth factor receptor mutations and immunohistochemical expression: a Gynecologic Oncology Group Study. Clin. Cancer Res. 11, 5539–5548 (2005).
Barber, T. D., Vogelstein, B., Kinzler, K. W. & Velculescu, V. E. Somatic mutations of EGFR in colorectal cancers and glioblastomas. N. Engl. J. Med. 351, 2883 (2004).
Lee, J. W. et al. Somatic mutations of EGFR gene in squamous cell carcinoma of the head and neck. Clin. Cancer Res. 11, 2879–2882 (2005).
Kwak, E. L. et al. Epidermal growth factor receptor kinase domain mutations in esophageal and pancreatic adenocarcinomas. Clin. Cancer Res. 12, 4283–4287 (2006).
Fukuoka, M. et al. Multi-institutional randomized phase II trial of gefitinib for previously treated patients with advanced non-small-cell lung cancer (The IDEAL 1 Trial). J. Clin. Oncol. 21, 2237–2246 (2003).
Kris, M. G. et al. Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer: a randomized trial. JAMA 290, 2149–2158 (2003).
Shepherd, F. A. et al. Erlotinib in previously treated non-small-cell lung cancer. N. Engl. J. Med. 353, 123–132 (2005). Reports on a phase III clinical trial that showed a significant survival benefit for individuals with advanced NSCLC who were treated with erlotinib.
Perez-Soler, R. et al. Determinants of tumor response and survival with erlotinib in patients with non-small-cell lung cancer. J. Clin. Oncol. 22, 3238–3247 (2004).
Bell, D. W. et al. Epidermal growth factor receptor mutations and gene amplification in non-small-cell lung cancer: molecular analysis of the IDEAL/INTACT gefitinib trials. J. Clin. Oncol. 23, 8081–8092 (2005).
Cortes-Funes, H. et al. Epidermal growth factor receptor activating mutations in Spanish gefitinib-treated non-small-cell lung cancer patients. Ann. Oncol. 16, 1081–1086 (2005).
Huang, S. F. et al. High frequency of epidermal growth factor receptor mutations with complex patterns in non-small cell lung cancers related to gefitinib responsiveness in Taiwan. Clin. Cancer Res. 10, 8195–8203 (2004).
Kim, K. S. et al. Predictors of the response to gefitinib in refractory non-small cell lung cancer. Clin. Cancer Res. 11, 2244–2251 (2005).
Kondo, M. et al. Mutations of epidermal growth factor receptor of non-small cell lung cancer were associated with sensitivity to gefitinib in recurrence after surgery. Lung Cancer 50, 385–391 (2005).
Mu, X. L. et al. Gefitinib-sensitive mutations of the epidermal growth factor receptor tyrosine kinase domain in chinese patients with non-small cell lung cancer. Clin. Cancer Res. 11, 4289–4294 (2005).
Niho, S. et al. First-line single agent treatment with gefitinib in patients with advanced non-small-cell lung cancer: a phase II study. J. Clin. Oncol. 24, 64–69 (2006).
Rosell, R. et al. Mutations in the tyrosine kinase domain of the EGFR gene associated with gefitinib response in non-small-cell lung cancer. Lung Cancer 50, 25–33 (2005).
Takano, T. et al. Epidermal growth factor receptor gene mutations and increased copy numbers predict gefitinib sensitivity in patients with recurrent non-small-cell lung cancer. J. Clin. Oncol. 23, 6829–6837 (2005).
Taron, M. et al. Activating mutations in the tyrosine kinase domain of the epidermal growth factor receptor are associated with improved survival in gefitinib-treated chemorefractory lung adenocarcinomas. Clin. Cancer Res. 11, 5878–5885 (2005).
Tokumo, M. et al. Double mutation and gene copy number of EGFR in gefitinib refractory non-small-cell lung cancer. Lung Cancer 53, 117–121 (2006).
Tomizawa, Y. et al. Clinicopathologic significance of the mutations of the epidermal growth factor receptor gene in patients with non-small cell lung cancer. Clin. Cancer Res. 11, 6816–6822 (2005).
Zhang, X. T. et al. The EGFR mutation and its correlation with response of gefitinib in previously treated Chinese patients with advanced non-small-cell lung cancer. Ann. Oncol. 16, 1334–1342 (2005).
Jackman, D. M. et al. Exon 19 deletion mutations of epidermal growth factor receptor are associated with prolonged survival in non-small cell lung cancer patients treated with gefitinib or erlotinib. Clin. Cancer Res. 12, 3908–3914 (2006).
Riely, G. J. et al. Clinical course of patients with non-small cell lung cancer and epidermal growth factor receptor exon 19 and exon 21 mutations treated with gefitinib or erlotinib. Clin. Cancer Res. 12, 839–844 (2006).
Tokumo, M. et al. The relationship between epidermal growth factor receptor mutations and clinicopathologic features in non-small cell lung cancers. Clin. Cancer Res. 11, 1167–1173 (2005).
Hirsch, F. R. et al. Increased epidermal growth factor receptor gene copy number detected by fluorescence in situ hybridization associates with increased sensitivity to gefitinib in patients with bronchioloalveolar carcinoma subtypes: a Southwest Oncology Group Study. J. Clin. Oncol. 23, 6838–6845 (2005).
Cappuzzo, F. et al. Increased HER2 gene copy number is associated with response to gefitinib therapy in epidermal growth factor receptor-positive non-small-cell lung cancer patients. J. Clin. Oncol. 23, 5007–5018 (2005).
Cappuzzo, F. et al. HER3 genomic gain and sensitivity to gefitinib in advanced non-small-cell lung cancer patients. Br. J. Cancer 93, 1334–1340 (2005).
Suzuki, R. et al. A phase II study of single-agent gefitinib as first-line therapy in patients with stage IV non-small-cell lung cancer. Br. J. Cancer 94, 1599–1603 (2006).
Inoue, A. et al. Prospective phase II study of gefitinib for chemotherapy-naive patients with advanced non-small-cell lung cancer with epidermal growth factor receptor gene mutations. J. Clin. Oncol. 24, 3340–3346 (2006).
Hirsch, F. R. et al. Molecular predictors of outcome with gefitinib in a phase III placebo-controlled study in advanced non-small-cell lung cancer. J. Clin. Oncol. 24, 5034–42 (2006).
Thatcher, N. et al. Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: results from a randomised, placebo-controlled, multicentre study (Iressa Survival Evaluation in Lung Cancer). Lancet 366, 1527–1537 (2005). Reports on a phase III clinical trial that showed a lack of survival benefit in individuals with advanced NSCLC who were treated with gefitinib.
Ono, M. et al. Sensitivity to gefitinib (Iressa, ZD1839) in non-small cell lung cancer cell lines correlates with dependence on the epidermal growth factor (EGF) receptor/extracellular signal-regulated kinase 1/2 and EGF receptor/Akt pathway for proliferation. Mol. Cancer Ther. 3, 465–472 (2004).
Han, S. W. et al. Epidermal growth factor receptor (EGFR) downstream molecules as response predictive markers for gefitinib (Iressa, ZD1839) in chemotherapy-resistant non-small cell lung cancer. Int. J. Cancer 113, 109–115 (2005).
Mukohara, T. et al. Differential effects of gefitinib and cetuximab on non-small-cell lung cancers bearing epidermal growth factor receptor mutations. J. Natl Cancer Inst. 97, 1185–1194 (2005).
Anderson, N. G., Ahmad, T., Chan, K., Dobson, R. & Bundred, N. J. ZD1839 (Iressa), a novel epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor, potently inhibits the growth of EGFR-positive cancer cell lines with or without erbB2 overexpression. Int. J. Cancer 94, 774–782 (2001).
Moasser, M. M., Basso, A., Averbuch, S. D. & Rosen, N. The tyrosine kinase inhibitor ZD1839 ('Iressa') inhibits HER2-driven signaling and suppresses the growth of HER2-overexpressing tumor cells. Cancer Res. 61, 7184–7188 (2001).
She, Q. B., Solit, D., Basso, A. & Moasser, M. M. Resistance to gefitinib in PTEN-null HER-overexpressing tumor cells can be overcome through restoration of PTEN function or pharmacologic modulation of constitutive phosphatidylinositol 3′-kinase/Akt pathway signaling. Clin. Cancer Res. 9, 4340–4346 (2003).
Engelman, J. A. et al. ErbB-3 mediates phosphoinositide 3-kinase activity in gefitinib-sensitive non-small cell lung cancer cell lines. Proc. Natl Acad. Sci. USA 102, 3788–3793 (2005). Gefitinib sensitivity is correlated with the heterodimerization of EGFR with ERBB2 and the attenuation of Akt activation in gefitinib-treated NSCLC cell lines.
Weinstein, I. B. Disorders in cell circuitry during multistage carcinogenesis: the role of homeostasis. Carcinogenesis 21, 857–864 (2000).
Sharma, S. V. et al. A common signaling cascade may underlie 'addiction' to the Src, BCR-ABL, and EGF receptor oncogenes. Cancer Cell 10, 425–435 (2006). Details the molecular mechanistic basis for cancer cell killing by oncogene-targeted therapeutics.
Evan, G. & Littlewood, T. A matter of life and cell death. Science 281, 1317–1322 (1998).
Chen, C. Y. & Faller, D. V. Direction of p21ras-generated signals towards cell growth or apoptosis is determined by protein kinase C and Bcl-2. Oncogene 11, 1487–1498 (1995).
Mayo, M. W. et al. Requirement of NF-κB activation to suppress p53-independent apoptosis induced by oncogenic Ras. Science 278, 1812–1815 (1997).
Johnson, D. et al. Regulation of both apoptosis and cell survival by the v-Src oncoprotein. Cell Death Differ. 7, 685–696 (2000).
Webb, B. L., Jimenez, E. & Martin, G. S. v-Src generates a p53-independent apoptotic signal. Mol. Cell. Biol. 20, 9271–9280 (2000).
Wang, J. Y. Regulation of cell death by the Abl tyrosine kinase. Oncogene 19, 5643–5650 (2000).
Reginato, M. J. et al. Integrins and EGFR coordinately regulate the pro-apoptotic protein Bim to prevent anoikis. Nature Cell Biol. 5, 733–740 (2003).
Evan, G. I. et al. Induction of apoptosis in fibroblasts by c-myc protein. Cell 69, 119–128 (1992).
Shi, Y. et al. Role for c-myc in activation-induced apoptotic cell death in T cell hybridomas. Science 257, 212–214 (1992).
White, E. & Stillman, B. Expression of adenovirus E1B mutant phenotypes is dependent on the host cell and on synthesis of E1A proteins. J. Virol. 61, 426–435 (1987).
Rodenhuis, S. et al. Mutational activation of the K-ras oncogene. A possible pathogenetic factor in adenocarcinoma of the lung. N. Engl. J. Med. 317, 929–935 (1987).
Mitsudomi, T. et al. ras gene mutations in non-small cell lung cancers are associated with shortened survival irrespective of treatment intent. Cancer Res. 51, 4999–5002 (1991).
Husgafvel-Pursiainen, K. et al. K-ras mutations in human adenocarcinoma of the lung: association with smoking and occupational exposure to asbestos. Int. J. Cancer 53, 250–256 (1993).
Ahrendt, S. A. et al. Cigarette smoking is strongly associated with mutation of the K-ras gene in patients with primary adenocarcinoma of the lung. Cancer 92, 1525–1530 (2001).
Soung, Y. H. et al. Mutational analysis of EGFR and K-RAS genes in lung adenocarcinomas. Virchows Arch. 446, 483–488 (2005).
Pao, W. et al. KRAS mutations and primary resistance of lung adenocarcinomas to gefitinib or erlotinib. PLoS Med. 2, e17 (2005). Shows the correlation between the presence of activated KRAS with NSCLC insensitivity to EGFR-TKIs.
Cully, M., You, H., Levine, A. J. & Mak, T. W. Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nature Rev. Cancer 6, 184–192 (2006).
Kohno, T., Takahashi, M., Manda, R. & Yokota, J. Inactivation of the PTEN/MMAC1/TEP1 gene in human lung cancers. Genes Chromosomes Cancer 22, 152–156 (1998).
Soria, J. C. et al. Lack of PTEN expression in non-small cell lung cancer could be related to promoter methylation. Clin. Cancer Res. 8, 1178–1184 (2002).
Bianco, R. et al. Loss of PTEN/MMAC1/TEP in EGF receptor-expressing tumor cells counteracts the antitumor action of EGFR tyrosine kinase inhibitors. Oncogene 22, 2812–2822 (2003).
Chakravarti, A., Loeffler, J. S. & Dyson, N. J. Insulin-like growth factor receptor I mediates resistance to anti-epidermal growth factor receptor therapy in primary human glioblastoma cells through continued activation of phosphoinositide 3-kinase signaling. Cancer Res. 62, 200–207 (2002).
Hurbin, A., Dubrez, L., Coll, J. L. & Favrot, M. C. Inhibition of apoptosis by amphiregulin via an insulin-like growth factor-1 receptor-dependent pathway in non-small cell lung cancer cell lines. J. Biol. Chem. 277, 49127–49133 (2002).
Erjala, K. et al. Signaling via ErbB2 and ErbB3 associates with resistance and epidermal growth factor receptor (EGFR) amplification with sensitivity to EGFR inhibitor gefitinib in head and neck squamous cell carcinoma cells. Clin. Cancer Res. 12, 4103–4111 (2006).
Cappuzzo, F. et al. Insulin-like growth factor receptor 1 (IGFR-1) is significantly associated with longer survival in non-small-cell lung cancer patients treated with gefitinib. Ann. Oncol. 17, 1120–1127 (2006).
Kosaka, T. et al. Analysis of epidermal growth factor receptor gene mutation in patients with non-small cell lung cancer and acquired resistance to gefitinib. Clin. Cancer Res. 12, 5764–5769 (2006).
Engelman, J. A. et al. Allelic dilution obscures detection of a biologically significant resistance mutation in EGFR-amplified lung cancer. J. Clin. Invest. 116, 2695–2706 (2006).
Bell, D. W. et al. Inherited susceptibility to lung cancer may be associated with the T790M drug resistance mutation in EGFR. Nature Genet. 37, 1315–1316 (2005). Shows that the T790M mutation that confers acquired resistance to EGFR-TKIs might also predispose individuals to develop NSCLC.
Shih, J. Y., Gow, C. H. & Yang, P. C. EGFR mutation conferring primary resistance to gefitinib in non-small-cell lung cancer. N. Engl. J. Med. 353, 207–208 (2005).
Gorre, M. E. et al. Clinical resistance to STI-571 cancer therapy caused by BCR-ABL gene mutation or amplification. Science 293, 876–880 (2001).
Blencke, S. et al. Characterization of a conserved structural determinant controlling protein kinase sensitivity to selective inhibitors. Chem. Biol. 11, 691–701 (2004).
Daub, H., Specht, K. & Ullrich, A. Strategies to overcome resistance to targeted protein kinase inhibitors. Nature Rev. Drug Discov. 3, 1001–1010 (2004).
Zhou, B. B. et al. Targeting ADAM-mediated ligand cleavage to inhibit HER3 and EGFR pathways in non-small cell lung cancer. Cancer Cell 10, 39–50 (2006).
Koizumi, F., Shimoyama, T., Taguchi, F., Saijo, N. & Nishio, K. Establishment of a human non-small cell lung cancer cell line resistant to gefitinib. Int. J. Cancer 116, 36–44 (2005).
Haber, D. A. & Settleman, J. Overcoming acquired resistance to Iressa/Tarceva with inhibitors of a different class. Cell Cycle 4, 1057–1059 (2005).
Elkind, N. B. et al. Multidrug transporter ABCG2 prevents tumor cell death induced by the epidermal growth factor receptor inhibitor Iressa (ZD1839, Gefitinib). Cancer Res. 65, 1770–1777 (2005).
Ozvegy-Laczka, C., Cserepes, J., Elkind, N. B. & Sarkadi, B. Tyrosine kinase inhibitor resistance in cancer: role of ABC multidrug transporters. Drug Resist. Updat. 8, 15–26 (2005).
Yanase, K. et al. Gefitinib reverses breast cancer resistance protein-mediated drug resistance. Mol. Cancer Ther. 3, 1119–1125 (2004).
Nakamura, Y. et al. Gefitinib ('Iressa', ZD1839), an epidermal growth factor receptor tyrosine kinase inhibitor, reverses breast cancer resistance protein/ABCG2-mediated drug resistance. Cancer Res. 65, 1541–1546 (2005).
Nagashima, S. et al. BCRP/ABCG2 levels account for the resistance to topoisomerase I inhibitors and reversal effects by gefitinib in non-small cell lung cancer. Cancer Chemother. Pharmacol. 58, 594–600 (2006).
Kitazaki, T. et al. Gefitinib, an EGFR tyrosine kinase inhibitor, directly inhibits the function of P-glycoprotein in multidrug resistant cancer cells. Lung Cancer 49, 337–343 (2005).
Yang, C. H. et al. Gefitinib reverses chemotherapy resistance in gefitinib-insensitive multidrug resistant cancer cells expressing ATP-binding cassette family protein. Cancer Res. 65, 6943–6949 (2005).
Singh, J. et al. Structure-based design of a potent, selective, and irreversible inhibitor of the catalytic domain of the erbB receptor subfamily of protein tyrosine kinases. J. Med. Chem. 40, 1130–1135 (1997).
Fry, D. W. et al. Specific, irreversible inactivation of the epidermal growth factor receptor and erbB2, by a new class of tyrosine kinase inhibitor. Proc. Natl Acad. Sci. USA 95, 12022–12027 (1998).
Rabindran, S. K. et al. Antitumor activity of HKI-272, an orally active, irreversible inhibitor of the HER-2 tyrosine kinase. Cancer Res. 64, 3958–3965 (2004).
Wissner, A. et al. Synthesis and structure-activity relationships of 6, 7-disubstituted 4-anilinoquinoline-3-carbonitriles. The design of an orally active, irreversible inhibitor of the tyrosine kinase activity of the epidermal growth factor receptor (EGFR) and the human epidermal growth factor receptor-2 (HER-2). J. Med. Chem. 46, 49–63 (2003).
Haber, D. A. et al. Molecular targeted therapy of lung cancer: EGFR mutations and response to EGFR inhibitors. Cold Spring Harb. Symp. Quant. Biol. 70, 419–426 (2005).
Discafani, C. M. et al. Irreversible inhibition of epidermal growth factor receptor tyrosine kinase with in vivo activity by N-[4-[(3-bromophenyl)amino]-6-quinazolinyl]-2-butynamide (CL-387, 785). Biochem. Pharmacol. 57, 917–925 (1999).
Smaill, J. B. et al. Tyrosine kinase inhibitors. 17. Irreversible inhibitors of the epidermal growth factor receptor: 4-(phenylamino)quinazoline- and 4-(phenylamino)pyrido[3, 2-d]pyrimidine-6-acrylamides bearing additional solubilizing functions. J. Med. Chem. 43, 1380–1397 (2000).
Carter, T. A. et al. Inhibition of drug-resistant mutants of ABL, KIT, and EGF receptor kinases. Proc. Natl Acad. Sci. USA 102, 11011–11016 (2005). Details the development of second generation TKIs that overcome resistance that arises from mutations of the gatekeeper residue in various tyrosine kinase oncogenes.
Shimamura, T. et al. Non-small-cell lung cancer and Ba/F3 transformed cells harboring the ERBB2 G776insV_G/C mutation are sensitive to the dual-specific epidermal growth factor receptor and ERBB2 inhibitor HKI-272. Cancer Res. 66, 6487–6491 (2006).
Ji, H. et al. Epidermal growth factor receptor variant III mutations in lung tumorigenesis and sensitivity to tyrosine kinase inhibitors. Proc. Natl Acad. Sci. USA 103, 7817–7822 (2006).
Shimamura, T., Lowell, A. M., Engelman, J. A. & Shapiro, G. I. Epidermal growth factor receptors harboring kinase domain mutations associate with the heat shock protein 90 chaperone and are destabilized following exposure to geldanamycins. Cancer Res. 65, 6401–6408 (2005).
Yang, S. et al. Association with HSP90 inhibits Cbl-mediated down-regulation of mutant epidermal growth factor receptors. Cancer Res. 66, 6990–6997 (2006).
Ge, J. et al. Design, synthesis, and biological evaluation of hydroquinone derivatives of 17-amino-17-demethoxygeldanamycin as potent, water-soluble inhibitors of Hsp90. J. Med. Chem. 49, 4606–4615 (2006).
Herbst, R. S., Fukuoka, M. & Baselga, J. Gefitinib–a novel targeted approach to treating cancer. Nature Rev. Cancer 4, 956–965 (2004).
Shaw, R. J. & Cantley, L. C. Ras, PI(3)K and mTOR signalling controls tumour cell growth. Nature 441, 424–430 (2006).
Ihle, N. T. et al. The phosphatidylinositol-3-kinase inhibitor PX-866 overcomes resistance to the epidermal growth factor receptor inhibitor gefitinib in A-549 human non-small cell lung cancer xenografts. Mol. Cancer Ther. 4, 1349–1357 (2005).
Fan, Q. W. et al. A dual PI3 kinase/mTOR inhibitor reveals emergent efficacy in glioma. Cancer Cell 9, 341–349 (2006).
Goudar, R. K. et al. Combination therapy of inhibitors of epidermal growth factor receptor/vascular endothelial growth factor receptor 2 (AEE788) and the mammalian target of rapamycin (RAD001) offers improved glioblastoma tumor growth inhibition. Mol. Cancer Ther. 4, 101–112 (2005).
Rao, R. D. et al. Disruption of parallel and converging signaling pathways contributes to the synergistic antitumor effects of simultaneous mTOR and EGFR inhibition in GBM cells. Neoplasia 7, 921–929 (2005).
Doherty, L. et al. Pilot study of the combination of EGFR and mTOR inhibitors in recurrent malignant gliomas. Neurology 67, 156–158 (2006).
Easton, J. B. & Houghton, P. J. Therapeutic potential of target of rapamycin inhibitors. Expert Opin. Ther. Targets 8, 551–564 (2004).
Margolin, K. et al. CCI-779 in metastatic melanoma: a phase II trial of the California Cancer Consortium. Cancer 104, 1045–1048 (2005).
Takeuchi, H. et al. Synergistic augmentation of rapamycin-induced autophagy in malignant glioma cells by phosphatidylinositol 3-kinase/protein kinase B inhibitors. Cancer Res. 65, 3336–3346 (2005).
Hay, N. The Akt-mTOR tango and its relevance to cancer. Cancer Cell 8, 179–183 (2005).
Adjei, A. A. Novel combinations based on epidermal growth factor receptor inhibition. Clin. Cancer Res. 12, 4446s–4450s (2006).
Hennequin, L. F. et al. Novel 4-anilinoquinazolines with C-7 basic side chains: design and structure activity relationship of a series of potent, orally active, VEGF receptor tyrosine kinase inhibitors. J. Med. Chem. 45, 1300–1312 (2002).
Wedge, S. R. et al. ZD6474 inhibits vascular endothelial growth factor signaling, angiogenesis, and tumor growth following oral administration. Cancer Res. 62, 4645–4655 (2002).
Ciardiello, F. et al. Antitumor activity of ZD6474, a vascular endothelial growth factor receptor tyrosine kinase inhibitor, in human cancer cells with acquired resistance to antiepidermal growth factor receptor therapy. Clin. Cancer Res. 10, 784–793 (2004).
Taguchi, F. et al. Anticancer effects of ZD6474, a VEGF receptor tyrosine kinase inhibitor, in gefitinib ('Iressa')-sensitive and resistant xenograft models. Cancer Sci. 95, 984–989 (2004).
Traxler, P. et al. AEE788: a dual family epidermal growth factor receptor/ErbB2 and vascular endothelial growth factor receptor tyrosine kinase inhibitor with antitumor and antiangiogenic activity. Cancer Res. 64, 4931–4941 (2004).
Lee, D. Phase II data with ZD6474, a small-molecule kinase inhibitor of epidermal growth factor receptor and vascular endothelial growth factor receptor, in previously treated advanced non-small-cell lung cancer. Clin. Lung Cancer 7, 89–91 (2005).
Stephens, P. et al. Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature 431, 525–526 (2004).
Sjoblom, T. et al. The consensus coding sequences of human breast and colorectal cancers. Science 314, 268–274 (2006). Highlights the genetic complexity and inter-tumour variability of human cancers.
Ngo, V. N. et al. A loss-of-function RNA interference screen for molecular targets in cancer. Nature 441, 106–110 (2006). New approaches to anticancer target discovery.
Weinstein, I. B. Cancer. Addiction to oncogenes--the Achilles heal of cancer. Science 297, 63–64 (2002).
Weinstein, I. B. & Joe, A. K. Mechanisms of disease: Oncogene addiction-a rationale for molecular targeting in cancer therapy. Nature Clin. Pract. Oncol. 3, 448–457 (2006).
Druker, B. J. et al. Effects of a selective inhibitor of the Abl tyrosine kinase on the growth of Bcr-Abl positive cells. Nature Med. 2, 561–566 (1996).
Chin, L. et al. Essential role for oncogenic Ras in tumour maintenance. Nature 400, 468–472 (1999).
Fisher, G. H. et al. Induction and apoptotic regression of lung adenocarcinomas by regulation of a K-Ras transgene in the presence and absence of tumor suppressor genes. Genes Dev. 15, 3249–3262 (2001).
Felsher, D. W. & Bishop, J. M. Reversible tumorigenesis by MYC in hematopoietic lineages. Mol. Cell 4, 199–207 (1999).
Felsher, D. W. Cancer revoked: oncogenes as therapeutic targets. Nature Rev. Cancer 3, 375–380 (2003).
Shigematsu, H. & Gazdar, A. F. Somatic mutations of epidermal growth factor receptor signaling pathway in lung cancers. Int. J. Cancer 118, 257–262 (2006).
Balak, M. N. et al. Novel D761Y and common secondary T790M mutations in epidermal growth factor receptor-mutant lung adenocarcinomas with acquired resistance to kinase inhibitors. Clin. Cancer Res. 12, 6494–6500 (2006).
Asahina, H. et al. Non-responsiveness to gefitinib in a patient with lung adenocarcinoma having rare EGFR mutations S768I and V769L. Lung Cancer 54, 419–422 (2006).
Baselga, J. et al. Phase I safety, pharmacokinetic, and pharmacodynamic trial of ZD1839, a selective oral epidermal growth factor receptor tyrosine kinase inhibitor, in patients with five selected solid tumor types. J. Clin. Oncol. 20, 4292–4302 (2002).
Janmaat, M. L., Kruyt, F. A., Rodriguez, J. A. & Giaccone, G. Response to epidermal growth factor receptor inhibitors in non-small cell lung cancer cells: limited antiproliferative effects and absence of apoptosis associated with persistent activity of extracellular signal-regulated kinase or Akt kinase pathways. Clin. Cancer Res. 9, 2316–2326 (2003).
Tracy, S. et al. Gefitinib induces apoptosis in the EGFRL858R non-small-cell lung cancer cell line H3255. Cancer Res. 64, 7241–7244 (2004).
Acknowledgements
The authors would like to thank T.-M. Chin and L. Sequist Van Dam for their helpful comments. We apologize to our many colleagues whose work is not cited owing to space constraints. Another notable omission is the very important area of antibody-based ErbB-targeted therapies, which is dealt with very well in other reviews.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
National Cancer Institute
FURTHER INFORMATION
Glossary
- Neural crest
-
A pluripotent, ectodermally derived ridge-like cluster of cells found on either side of the neural tube in vertebrate embryos.
- Cytotoxic chemotherapy
-
Chemicals or drugs that kill proliferating cells, especially cancer cells. Their side effects are typically related to the inhibition of normal cell proliferation, with a narrow window of selectivity for cancer cells.
- Molecular-targeted therapeutics
-
Chemicals or drugs that target known proteins that are important in cancer cell proliferation or survival at the same time as being dispensable to normal cells. Although side effects are typically less severe than with cytotoxic agents, the effective inhibition of the target protein might not translate into generally effective therapies, hence the importance of reliable biomarkers.
- Autocrine loop
-
A mode of cell signalling in which soluble ligands released by cells stimulate receptors on their own cell surfaces.
- Reversible inhibitors
-
Inhibitors that bind non-covalently with biological molecules and interfere with their activity.
- Unselected patients
-
A cohort of patients identified on the basis of tissue diagnosis but not correlated with biomarkers (that is, sequencing of the EGFR gene was not used as a selection criterion).
- Ligand independence
-
The activation of a receptor in the absence of interaction with its cognate ligand.
- Kcat
-
The overall catalytic rate of an enzyme (that is, the number of substrate molecules converted to product by each catalytic site per unit of time.
- Km
-
The Michaelis–Menten constant. Km is a measure of the affinity of a substrate for an enzyme, and is the substrate concentration at half the maximal velocity of an enzyme.
- Ki
-
The dissociation constant for the binding of an inhibitor to an enzyme.
- Phage-display method
-
A method in which proteins or peptides are displayed on the surface of filamentous bacteriophages, which can then be used to study the interaction of the peptide with other proteins or chemicals.
- Oncogenic shock
-
A mechanism to explain oncogene addiction, in which the acute inactivation of an oncoprotein is associated with differential attenuation rates of pro-survival and pro-apoptotic signals emanating from the oncoprotein, such that apoptotic signals become predominant and kill the cancer cell.
- Differential signal decay
-
A signalling imbalance created by the rapid decay of pro-survival signals and persistence of the relatively long-lived pro-apoptotic signals after acute oncogene inactivation.
- Gatekeeper residue
-
Amino acids with small side chains found at the catalytic site of enzymes that, when mutated to amino acids with bulkier side chains, can sterically impede the binding of a drug at the active site of the enzyme at the same time as retaining substrate binding.
- Irreversible inhibitors
-
Inhibitors that bind covalently with biological molecules and interfere with their activity.
Rights and permissions
About this article
Cite this article
Sharma, S., Bell, D., Settleman, J. et al. Epidermal growth factor receptor mutations in lung cancer. Nat Rev Cancer 7, 169–181 (2007). https://doi.org/10.1038/nrc2088
Issue Date:
DOI: https://doi.org/10.1038/nrc2088
This article is cited by
-
Targeting pyruvate dehydrogenase kinase 1 overcomes EGFR C797S mutation-driven osimertinib resistance in non-small cell lung cancer
Experimental & Molecular Medicine (2024)
-
Using Vision Transformer for high robustness and generalization in predicting EGFR mutation status in lung adenocarcinoma
Clinical and Translational Oncology (2024)
-
Nanoengineered approaches to improve the efficacy of targeted drug delivery for the treatment of malignancy: a comprehensive review
Future Journal of Pharmaceutical Sciences (2023)
-
The development of amivantamab for the treatment of non-small cell lung cancer
Respiratory Research (2023)
-
Assessment of respiratory and reproductive impacts of artisanal refinery activities on male Albino Wistar rats: implications for environmental health
Bulletin of the National Research Centre (2023)