Key Points
-
The International Agency for Research on Cancer has determined that, based on results from epidemiological studies, people who are overweight or obese are at increased risk of developing several cancer types, including adenocarcinoma of the oesophagus, colon cancer, breast cancer (in postmenopausal women), endometrial cancer and kidney (renal-cell) cancer.
-
Epidemiological evidence also indicates that cancers of the liver, gallbladder and pancreas are obesity related, and that obesity might also increase risk for haematopoietic cancers and for aggressive prostate cancer. No association is seen between obesity and lung cancer. Results for other cancers have been inconsistent.
-
Insulin resistance develops as a metabolic adaptation to increased levels of circulating free fatty acids released from adipose tissue, especially intra-abdominal adipose. Insulin resistance is generally compensated by increased pancreatic insulin secretion. There is mounting epidemiological and experimental evidence to indicate that chronic hyperinsulinaemia increases risk of cancers of the colon and endometrium, and probably other tumours (for example, of the pancreas and kidney).
-
Serum levels of insulin-like growth factor 1 (IGF1) are also associated with different forms of cancer. However, there is no simple, direct relationship between circulating levels of IGF1 and the degree of adiposity.
-
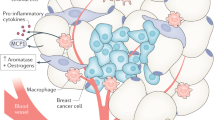
Circulating levels of oestrogens are strongly related to adiposity. For cancers of the breast (in postmenopausal women) and endometrium, the effects of overweight and obesity on cancer risk are largely mediated by increased oestrogen levels.
-
In 4–8% of premenopausal women, obesity and ensuing insulin resistance can either cause or aggravate syndromes of ovarian androgen excess (polycystic ovary syndrome) and chronic progesterone deficiency. There is strong evidence that such syndromes, along with reduced progesterone production, increase the risk of endometrial cancer.
-
Successful intervention strategies for weight loss and maintenance at the individual and community level are needed to reduce cancer risk.
Abstract
The prevalence of obesity is rapidly increasing globally. Epidemiological studies have associated obesity with a range of cancer types, although the mechanisms by which obesity induces or promotes tumorigenesis vary by cancer site. These include insulin resistance and resultant chronic hyperinsulinaemia, increased bioavailability of steroid hormones and localized inflammation. Gaining a better understanding of the relationship between obesity and cancer can provide new insight into mechanisms of cancer pathogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Heymsfield, S. B. et al. in Handbook of Obesity: Etiology and Pathophysiology (eds. Bray, G. & Bouchard, C.) 33–79 (Marcel Dekker, New York, 2004).
Flegal, K. M., Carroll, M. D., Ogden, C. L. & Johnson, C. L. Prevalence and trends in obesity among US adults, 1999–2000. JAMA 288 1723–1727 (2002).
Mokdad, A. H. et al. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA 289, 76–79 (2003).
Calle, E. E., Rodriguez, C., Walker-Thurmond, K. & Thun, M. J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U. S. adults. N. Engl. J. Med. 348, 1625–1638 (2003). A landmark epidemiological study indicating that overweight and obesity are associated with mortality from a wide range of cancer types, and might explain 14% and 20% of all deaths from cancer in men and women from the United States, respectively.
International Agency for Reasearch on Cancer. IARC Handbooks of Cancer Prevention. Weight Control and Physical Activity (International Agency for Research on Cancer, Lyon, 2002). An extensive report, prepared by an international expert panel, reviewing and summarizing epidemiological and experimental evidence relating increased cancer risk of excess weight. The report also includes a detailed discussion of hormonal and other mechanisms that might provide the physiological link between excess weight and tumour development.
Willett, W. C. in Nutritional Epidemiology (ed. Willett, W.) 244–273 (Oxford University Press, New York, 1998).
Kuczmarski, R. J. & Flegal, K. M. Criteria for definition of overweight in transition: background and recommendations for the United States. Am. J. Clin. Nutr. 72, 1074–1081 (2000).
James, P. T., Leach, R., Kalamara, E. & Shayeghi, M. The worldwide obesity epidemic. Obes. Res. 9 (Suppl. 4), 228–233 (2001).
Seidell, J. C. in Progress in Obesity Research (eds. Medeiros-Neto, G., Halpern, A. & Bouchard, C.) 571–578 (John Libbey and Company, London, 2003).
Seidell, J. & Rissanen, A. in Handbook of Obesity: Etiology and Pathophysiology (eds. Bray, G. & Bouchard, C.) 93–107 (Marcel Dekker, New York, 2004).
Bjorntorp, P. Obesity. Lancet 350, 423–426 (1997).
Giovannucci, E., Colditz, G. A., Stampfer, M. J. & Willett, W. C. Physical activity, obesity, and risk of colorectal adenoma in women (United States). Cancer Causes Control 7, 253–263 (1996).
Giovannucci, E. et al. Physical activity, obesity, and risk for colon cancer and adenoma in men. Ann. Intern. Med. 122, 327–334 (1995).
Calle, E. E., Miracle-McMahill, H. L., Thun, M. J. & Heath, C. W. Jr. Estrogen replacement therapy and risk of fatal colon cancer in a prospective cohort of postmenopausal women. J. Natl Cancer Inst. 87, 517–523 (1995).
Writing Group for the Women's Health Initiative Investigators. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results from the Women's Health Initiative randomized controlled trial. JAMA 288, 321–333 (2002).
Tchernof, A. & Despres, J. P. Sex steroid hormones, sex hormone-binding globulin, and obesity in men and women. Horm. Metab. Res. 32, 526–536 (2000).
Stephenson, G. D. & Rose, D. P. Breast cancer and obesity: an update. Nutr. Cancer 45, 1–16 (2003). An excellent and comprehensive review of the impact of adiposity on both breast cancer risk and the clinical behaviour of the established disease.
Potischman, N., Swanson, C. A., Siiteri, P. & Hoover, R. N. Reversal of relation between body mass and endogenous estrogen concentrations with menopausal status. J. Natl Cancer Inst. 88, 756–758 (1996).
Ballard-Barbash, R. & Swanson, C. A. Body weight: estimation of risk for breast and endometrial cancer. Am. J. Clin. Nutr. 63 (Suppl. 3), 437–441 (1996).
Galanis, D. J. et al. Anthropometric predictors of breast cancer incidence and survival in a multi-ethnic cohort of female residents of Hawaii, United States. Cancer Causes Control 9, 217–224 (1998).
Trentham-Dietz, A. et al. Body size and risk of breast cancer. Am. J. Epidemiol. 145, 1011–1119 (1997).
Hunter, D. J. & Willett, W. C. Diet, body size and breast cancer. Epidemiol. Rev. 15, 110–132 (1993).
Harvie, M., Hooper, L. & Howell, A. H. Central obesity and breast cancer risk: a systematic review. Obesity Rev. 4, 157–173 (2003).
Folsom, A. R. et al. Increased incidence of carcinoma of the breast associated with abdominal adiposity in postmenopausal women. Am. J. Epidemiol. 131, 794–803 (1990).
Huang, Z. et al. Dual effects of weight and weight gain on breast cancer risk. JAMA 278, 1407–1411 (1997).
Barnes-Josiah, D., Potter, J. D., Sellers, T. A. & Himes, J. H. Early body size and subsequent weight gain as predictors of breast cancer incidence (Iowa, United States). Cancer Causes Control 6, 112–118 (1995).
Feigelson, H. S., Jonas, C. R., Teras, L. R., Thun, M. J. & Calle, E. E. Weight gain, body mass index, hormone replacement therapy, and postmenopausal breast cancer in a large prospective study. Cancer Epidemiol. Biomark. Prev. 13, 220–224 (2004).
Collaborative Group on Hormonal Factors in Breast Cancer. Breast cancer and hormone replacement therapy: collaborative reanalysis of data from 51 epidemiological studies of 52 705 women with breast cancer and 108 411 women without breast cancer. Lancet 350, 1047–1059 (1997).
Schairer, C. et al. Menopausal estrogen and estrogen-progestin replacement therapy and breast cancer risk. JAMA 283, 485–491 (2000).
Rock, C. L. & Demark-Wahnefried, W. Nutrition and survival after the diagnosis of breast cancer: a review of the evidence. J. Clin. Oncol. 20, 3302–3316 (2002).
Chlebowski, R. T., Aiello, E. & McTiernan, A. Weight loss in breast cancer patient management. J. Clin. Oncol. 20, 1128–1143 (2002).
Petrelli, J. M., Calle, E. E., Rodriguez, C. & Thun, M. J. Body mass index, height, and postmenopausal breast cancer mortality in a prospective cohort of U. S. women. Cancer Causes Control 13, 325–332 (2002).
Wee, C., McCarthy, E., Davis, R. & Phillips, R. Screening for cervical and breast cancer: is obesity an unrecognized barrier to preventive care? Ann. Intern. Med. 132, 697–704 (2000).
Reeves, M., Newcomb, P. A., Remington, P., Marcus, P. M. & MacKenzie, W. Body mass and breast cancer, relationship between method of detection and stage of disease. Cancer 77, 301–307 (1996).
Kaaks, R., Lukanova, A. & Kurzer, M. A. Obesity, endogenous hormones, and endometrial cancer risk: a synthetic review. Cancer Epidemiol. Biomark. Prev. 11, 1531–1543 (2002).
Lew, E. A. & Garfinkel, L. Variations in mortality by weight among 750,000 men and women. J. Chron. Dis. 32, 563–576 (1979).
Bergstrom, A. et al. Obesity and renal cell cancer — a quantitative review. Br. J. Cancer 85, 984–990 (2001).
Hu, J., Mao, Y., White, K. & The Canadian Cancer Registries Epidemiology Research Group. Overweight and obesity in adults and risk of renal cell carcinoma in Canada. Soz Praventivmed. 48, 178–185 (2003).
Pan, S. Y. et al. Association of obesity and cancer risk in Canada. Am. J. Epidemiol. 159, 259–268 (2004).
Samanic, C. et al. Obesity and cancer risk among white and black United States veterans. Cancer Causes Control 15, 35–43 (2004).
World Cancer Research Fund & American Institute for Cancer Research. in Food, Nutrition and the Prevention of Cancer: a global perspective 371–373 (Am. Instit. Cancer Res., Washington, 1997).
Hill, H. A. & Austin, H. Nutrition and endometrial cancer. Cancer Causes Control 7, 19–32 (1996).
Wolk, A., Lindblad, P. & Adami, H. -O. Nutrition and renal cell cancer. Cancer Causes Control 7, 5–18 (1996).
Chow, W. H. et al. Obesity and risk of renal cell cancer. Cancer Epidemiol. Biomarkers Prev. 5, 17–21 (1996).
Mellemgaard, A., Engholm, G., McLaughlin, J. K. & Olsen, J. H. Risk factors for renal-cell carcinoma in Denmark. III. Role of weight, physical activity and reproductive factors. Int. J. Cancer 56, 66–71 (1994).
McLaughlin, J. et al. A population-based case-control study of renal cell carcinoma. J. Natl Cancer Inst. 72, 275–284 (1984).
McLaughlin, J. K. et al. Risk factors for renal-cell cancer in Shanghai, China. Int. J. Cancer 52, 562–565 (1992).
Chow, W. H., Gridley, G., Fraumeni, J. F. & Jarvholm, B. Obesity, hypertension, and the risk of kidney cancer in men. N. Engl. J. Med. 343, 1305–1311 (2000).
Devesa, S. S., Blot, W. J. & Fraumeni, J. F. Changing patterns in the incidence of esophageal and gastric carcinoma in the United States. Cancer 83, 2049–2053 (1998).
Wild, C. P. & Hardie, L. J. Reflux, Barrett's oesophagus and adenocarcinoma: burning questions. Nature Rev. Cancer 3, 676–685 (2003).
Wu, A. H. & Bernstein, L. A multiethnic population-based study of smoking, alcohol and body size and risk of adenocarcinomas of the stomach and esophagus. Cancer Causes Control 12, 721–732 (2001).
Lagergren, J., Bergstrom, R., Lindgren, A. & Nyren, O. Symptomatic gastroesophageal reflux as a risk factor for esophageal adenocarcinoma. N. Engl. J. Med. 340, 825–831 (1999).
Chow, W. H. et al. The relation of gastroesophageal reflux disease and its treatment to adenocarcinomas of the esophagus and gastric cardia. JAMA 274, 474–477 (1995).
Nilsson, M., Johnsen, R., Weimin, Y., Hveem, K. & Lagergren, J. Obesity and estrogen as risk factors for gastroesophageal reflux symptoms. JAMA 290, 66–72 (2003).
Lagergren, J., Bergstrom, R. & Nyren, O. No relation between body mass and gastro-esophageal reflux symptoms in a Swedish population based study. Gut 47, 26–29 (2000).
Chow, W. H. et al. Body mass index and risk of adenocarcinomas of the esophagus and gastric cardia. J. Natl Cancer Inst. 90, 150–55 (1998).
Lagergren, J., Bergstrom, R., Adami, H. O. & Nyren, O. Association between medications that relax the lower esophageal sphincter and risk for esophageal adenocarcinoma. Ann. Intern. Med. 133, 165–175 (2000).
Moller, H., Mellemgaard, A., Lindvig, K. & Olsen, J. H. Obesity and cancer risk: a Danish record-linkage study. Eur. J. Cancer 30A, 344–350 (1994).
Gapstur, S. M. et al. Abnormal glucose metabolism and pancreatic cancer mortality. JAMA 283, 2552–2558 (2000).
Michaud, D. S. et al. Physical activity, obesity, height, and the risk of pancreatic cancer. JAMA 286, 921–929 (2001).
Silverman, D. T. et al. Dietary and nutritional factors and pancreatic cancer: a case-control study based on direct interviews. J. Natl Cancer Inst. 90, 1710–1719 (1998).
Hanley, A. J. G., Johnson, K. C., Villeneuve, P. J. & Mao, Y. Physical activity, anthropometric factors and risk of pancreatic cancer: results from the Canadian enhanced cancer surveillance system. Int. J. Cancer 94, 140–147 (2001).
Berrington de Gonzalez, A., Sweetland, S. & Spencer, E. A meta-analysis of obesity and the risk of pancreatic cancer. Br. J. Cancer 89, 519–523 (2003).
Lee, I. -M., Sesso, H. D., Oguma, Y. & Paffenbarger, R. S. Physical activity, body weight, and pancreatic cancer mortality. Br. J. Cancer 88, 679–683 (2003).
Zatonski, W. et al. Nutritional factors and pancreatic cancer: a case-control study from south-west Poland. Int. J. Cancer 48, 390–394 (1991).
Howe, G. R., Jain, M. & Miller, A. B. Dietary factors and risk of pancreatic cancer: results of a Canadian population-based case-control study. Int. J. Cancer 45, 604–608 (1990).
Ghadirian, P., Simard, A., Baillargeon, J., Maisonneuve, P. & Boyle, P. Nutritional factors and pancreatic cancer in the francophone community in Montreal, Canada. Int. J. Cancer 47, 1–6 (1991).
Bueno de Mesquita, H. B., Moerman, C. J., Runia, S. & Maisonneuve, P. Are energy and energy-providing nutrients related to exocrine carcinoma of the pancreas? Int. J. Cancer 46, 435–444 (1990).
Wolk, A. et al. A prospective study of obesity and cancer risk (Sweden). Cancer Causes Control 12, 13–21 (2001).
Strom, B. L. et al. Risk factors for gallbladder cancer. Cancer 76, 1747–1756 (1995).
Zatonski, W. A. et al. Epidemiologic aspects of gallbladder cancer: a case-control study of the SEARCH Program of the International Agency for Research on Cancer. J. Natl Cancer Inst. 89, 1132–1138 (1997).
Ji, B. -T. et al. Body mass index and the risk of cancers of the gastric cardia and distal stomach in Shanghai, China. Cancer Epidemiol. Biomarkers Prev. 6, 481–485 (1997).
Vaughan, T. L., Davis, S., Kristal, A. & Thomas, D. B. Obesity, alcohol, and tobacco as risk factors fo cancers of the esophagus and gastric cardia: adenocarcinoma versus squamous cell carcinoma. Cancer Epidemiol. Biomarkers Prev. 4, 85–92 (1995).
Henley, S. J., Flanders, W. D., Manatunga, A. & Thun, M. J. Leanness and lung cancer risk: fact or artifact? Epidemiology 13, 268–276 (2002).
Tornberg, S. A. & Carstensen, J. M. Relationship between Quetelet's index and cancer of breast and female genital tract in 47,000 women followed for 25 years. Br. J. Cancer 69, 358–361 (1994).
Lacey, J. V. et al. Obesity as a potential risk factor for adenocarcinomas and squamous cell carcinomas of the uterine cervix. Cancer 98, 814–821 (2003).
Risch, H. A. Hormonal etiology of epithelial ovarian cancer, with a hypothesis concerning the role of androgens and progesterone. J. Natl Cancer Inst. 90, 1774–1786 (1998).
Rodriguez, C., Calle, E. E., Fakhrabadi-Shokoohi, D., Jacobs, E. J. & Thun, M. J. Body mass index, height, and the risk of ovarian cancer mortality in a prospective cohort of postmenopausal women. Cancer Epidemiol. Biomark. Prev. 11, 822–828 (2002).
Engeland, A., Tretli, S. & Bjorge, T. Height, body mass index, and ovarian cancer: a follow-up of 1. 1 million Norwegian women. J. Natl Cancer Inst. 95, 1244–1248 (2003).
Fairfield, K. M. et al. Obesity, weight gain, and ovarian cancer. Obstet. Gynecol. 100, 288–296 (2002).
Kuper, H., Cramer, D. W. & Titus-Ernstoff, L. Risk of ovarian cancer in the United States in relation to anthropometric measures: does the association depend on menopausal status? Cancer Causes Control 13, 455–463 (2002).
Lubin, F. et al. Body mass index at age 18 years and during adult life and ovarian cancer risk. Am. J. Epidemiol. 157, 113–120 (2003).
Schouten, L. J., Goldbohm, A. & van den Brandt, P. A. Height, weight, weight change, and ovarian cancer risk in the Netherlands cohort study on diet and cancer. Am. J. Epidemiol. 157, 424–433 (2003).
Calle, E. E. Invited commentary: Do anthropometric measures predict risk of prostate cancer? Am. J. Epidemiol. 151, 550–553 (2000).
Engeland, A., Tretli, S. & Bjorge, T. Height, body mass index, and prostate cancer: a follow-up of 950,000 Norwegian men. Br. J. Cancer 89, 1237–1242 (2003).
Giovannucci, E., Rimm, E. B., Stampfer, M. J., Colditz, G. A. & Willett, W. C. Height, body weight, and risk of prostate cancer. Cancer Epidemiol. Biomark. Prev. 6, 557–563 (1997).
Andersson, S. O. et al. Body size and prostate cancer: a 20-year follow-up study among 135,006 Swedish construction workers. J. Natl Cancer Inst. 89, 385–389 (1997).
Rodriguez, C. et al. Body mass index, height, and prostate cancer mortality in two large cohorts of adult men in the United States. Cancer Epidemiol. Biomark. Prev. 10, 345–353 (2001).
Amling, C. L. et al. Pathologic variables and recurrence rates as related to obesity and race in men with prostate cancer undergoing radical prostatectomy. J. Clin. Oncol. 22, 439–445 (2004).
Freedland, S. J. et al. Impact of obesity on biochemical control after radical prostatectomy for clinically localized prostate cancer: a report by the Shared Equal Access Regional Cancer Hospital Database Study Group. J. Clin. Oncol. 22, 446–453 (2004).
Holly, E. A., Lele, C., Bracci, P. M. & McGrath, M. S. Case-control study of non-Hodgkin's lymphoma among women and heterosexual men in the San Francisco Bay area, California. Am. J. Epidemiol. 150, 375–389 (1999).
Cerhan, J. R. et al. Anthropometric characteristics, physical activity and risk of non-Hodgkin's lymphoma subtypes and B-cell lymphocytic leukemia: a prospective study. Am. J. Epidemiol. 156, 527–535 (2003).
Rajala, M. W. & Scherer, P. E. Minireview: the adipocyte — at the crossroads of energy homeostasis, inflammation, and atherosclerosis. Endocrinology 144, 3765–3773 (2003).
Reaven, G. M. Banting lecture 1988. Role of insulin resistance in human disease. Diabetes 37, 1595–1607 (1988).
Wajchenberg, B. L. Subcutaneous and visceral adipose tissue: their relation to the metabolic syndrome. Endocr. Rev. 21, 697–738 (2000).
McKeown-Eyssen, G. Epidemiology of colorectal cancer revisited: are serum triglycerides and/or plasma glucose associated with risk? Cancer Epidemiol. Biomarkers Prev. 3, 687–695 (1994).
Giovannucci, E. Insulin and colon cancer. Cancer Causes Control 6, 164–179 (1995).
Kaaks, R. Nutrition, hormones, and breast cancer: is insulin the missing link? Cancer Causes Control 7, 605–625 (1996).
Stoll, B. A. Western nutrition and the insulin resistance syndrome: a link to breast cancer. Eur. J. Clin. Nutr. 53, 83–87 (1999).
Weiderpass, E. et al. Occurrence, trends and environment etiology of pancreatic cancer. Scand. J. Work Environ. Health 24, 165–174 (1998).
Lawlor, M. A. & Alessi, D. R. PKB/Akt: a key mediator of cell proliferation, survival and insulin responses? J. Cell Sci. 114, 2903–2910 (2001).
Prisco, M., Romano, G., Peruzzi, F., Valentinis, B. & Baserga, R. Insulin and IGF-I receptors signaling in protection from apoptosis. Horm. Metab. Res. 31, 80–89 (1999).
Ish-Shalom, D. et al. Mitogenic properties of insulin and insulin analogues mediated by the insulin receptor. Diabetologia 40 (Suppl. 2), 25–31 (1997).
Le Roith, D. Regulation of proliferation and apoptosis by the insulin-like growth factor I receptor. Growth Horm. IGF Res. 10 (Suppl. A), 12–13 (2000).
Khandwala, H. M., McCutcheon, I. E., Flyvbjerg, A. & Friend, K. E. The effects of insulin-like growth factors on tumorigenesis and neoplastic growth. Endocr. Rev. 21, 215–244 (2000).
Shafie, S. M. & Grantham, F. H. Role of hormones in the growth and regression of human breast cancer cells (MCF-7) transplanted into athymic nude mice. J. Natl Cancer Inst. 67, 51–56 (1981).
Shafie, S. M. & Hilf, R. Insulin receptor levels and magnitude of insulin-induced responses in 7,12-dimethylbenz(a)-anthracene-induced mammary tumors in rats. Cancer Res. 41, 826–829 (1981).
Heuson, J. C. & Legros, N. Effect of insulin and of alloxan diabetes on growth of the rat mammary carcinoma in vivo. Eur. J. Cancer 6, 349–351 (1970).
Heuson, J. C. & Legros, N. Influence of insulin deprivation on growth of the 7,12-dimethylbenz(a)anthracene-induced mammary carcinoma in rats subjected to alloxan diabetes and food restriction. Cancer Res. 32, 226–232 (1972).
Cocca, C. et al. An experimental model of diabetes and cancer in rats. Eur. J. Cancer 34, 889–894 (1998).
Cocca, C. et al. Suppression of mammary gland tumorigenesis in diabetic rats. Cancer Detect. Prev. 27, 37–46 (2003).
LeRoith, D. & Roberts, C. T. Jr. The insulin-like growth factor system and cancer. Cancer Lett. 195, 127–137 (2003).
Wideroff, L. et al. Cancer incidence in a population-based cohort of patients hospitalized with diabetes mellitus in Denmark. J. Natl Cancer Inst. 89, 1360–1365 (1997).
Lindblad, P. et al. The role of diabetes mellitus in the aetiology of renal cell cancer. Diabetologia 42, 107–112 (1999).
Everhart, J. & Wright, D. Diabetes mellitus as a risk factor for pancreatic cancer. A meta-analysis. JAMA 273, 1605–1609 (1995).
Kaaks, R. et al. Serum C-peptide, insulin-like growth factor (IGF)-I, Igf-binding proteins, and colorectal cancer risk in women. J. Natl Cancer Inst. 92, 1592–1600 (2000). Provides the first concrete evidence, from a prospective cohort study, that increased circulating insulin levels are a risk factor for the development of colon cancer.
Stattin, P. et al. Obesity and colon cancer: does leptin provide a link? Int. J. Cancer 109, 149–152 (2004).
Schoen, R. E. et al. Increased blood glucose and insulin, body size, and incident colorectal cancer. J. Natl Cancer Inst. 91, 1147–1154 (1999).
Lukanova, A. et al. Prediagnostic levels of C-peptide, IGF-I, IGFBP-1-2 and-3 and risk of endometrial cancer. Int. J. Cancer 108, 262–268 (2004). Gives the first solid evidence, from a prospective cohort study, that increased circulating insulin levels are a risk factor for the development of endometrial cancer.
Bruning, P. F. et al. Insulin resistance and breast cancer risk. Int. J. Cancer 52, 511–516 (1992).
Del Giudice, M. E. et al. Insulin and related factors in premenopausal breast cancer. Breast Cancer Res. Treat. 47, 111–120 (1998).
Hirose, K. et al. Insulin, insulin-like growth factor-I and breast cancer risk in Japanese women. Asian Pac. J. Cancer Prev. 4, 239–246 (2003).
Yang, G. et al. Population-based, case-control study of blood C-peptide level and breast cancer risk. Cancer Epidemiol. Biomarkers Prev. 10, 1207–1211 (2001).
Schairer, C. et al. Serum concentrations of IGF-I, IGFBP-3 and c-peptide and risk of hyperplasia and cancer of the breast in postmenopausal women. Int. J. Cancer 108, 773–779 (2004).
Kaaks, R. et al. Prospective study of IGF-I, IGF-binding proteins, and breast cancer risk, in northern and southern Sweden. Cancer Causes Control 13, 307–316 (2002).
Keinan-Boker, L. et al. Circulating levels of insulin-like growth factor I, its binding proteins-1,-2,-3, C-peptide and risk of postmenopausal breast cancer. Int. J. Cancer 106, 90–95 (2003).
Muti, P. et al. Fasting glucose is a risk factor for breast cancer: a prospective study. Cancer Epidemiol. Biomarkers Prev. 11, 1361–1368 (2002).
Toniolo, P. et al. Serum insulin-like growth factor-I and breast cancer. Int. J. Cancer 88, 828–832 (2000).
Clemmons, D. R. Role of insulin-like growth factor binding proteins in controlling IGF actions. Mol. Cell Endocrinol. 140, 19–24 (1998).
Kaaks, R. & Lukanova, A. Energy balance and cancer: the role of insulin and insulin-like growth factor-I. Proc. Nutr. Soc. 60, 91–106 (2001).
Thissen, J. P., Ketelslegers, J. M. & Underwood, L. E. Nutritional regulation of the insulin-like growth factors. Endocr. Rev. 15, 80–101 (1994).
Allen, N. E. et al. Lifestyle determinants of serum insulin-like growth-factor-I (IGF-I), C-peptide and hormone binding protein levels in British women. Cancer Causes Control 14, 65–74 (2003).
Holmes, M. D., Pollak, M. N. & Hankinson, S. E. Lifestyle correlates of plasma insulin-like growth factor I and insulin-like growth factor binding protein 3 concentrations. Cancer Epidemiol. Biomarkers Prev. 11, 862–867 (2002).
Lukanova, A. et al. Circulating levels of insulin-like growth factor-I and risk of ovarian cancer. Int. J. Cancer 101, 549–554 (2002).
Chapman, I. M. et al. Recovery of growth hormone release from suppression by exogenous insulin-like growth factor I (IGF-I): evidence for a suppressive action of free rather than bound IGF-I. J. Clin. Endocrinol. Metab. 83, 2836–2842 (1998).
Tannenbaum, G. S., Guyda, H. J. & Posner, B. I. Insulin-like growth factors: a role in growth hormone negative feedback and body weight regulation via brain. Science 220, 77–79 (1983).
Hankinson, S. E. et al. Circulating concertrations of insulin-like growth factor-I and risk of breast cancer. Lancet 351, 1393–1396 (1998).
Stattin, P. et al. Plasma insulin-like growth factor-I, insulin-like growth factor-binding proteins, and prostate cancer risk: a prospective study. J. Natl Cancer Inst. 92, 1910–1917 (2000).
Chan, J. M. et al. Plasma insulin-like growth factor-I and prostate cancer risk: a prospective study. Science 279, 563–566 (1998).
Giovannucci, E. et al. A prospective study of plasma insulin-like growth factor-1 and binding protein-3 and risk of colorectal neoplasia in women. Cancer Epidemiol. Biomark. Prev. 9, 345–349 (2000).
Ma, J. et al. A prospective study of plasma levels of insulin-like growth factor I, insulin-like growth factor binding protein 3 and colorectal cancer risk among men. J. Natl Cancer Inst. 91, 620–625 (1999).
Palmqvist, R. et al. Plasma insulin-like growth factor 1, insulin-like growth factor binding protein 3, and risk of colorectal cancer: a prospective study in northern Sweden. Gut 50, 642–646 (2002).
Key, T. J., Allen, N. E., Verkasalo, P. K. & Banks, E. Energy balance and cancer: the role of sex hormones. Proc. Nutr. Soc. 60, 81–89 (2001).
Key, T. J. et al. Body mass index, serum sex hormones, and breast cancer risk in postmenopausal women. J. Natl Cancer Inst. 95, 1218–1226 (2003). This paper presents a re-analysis of the original data from nine prospective cohort studies on the relationships between excess weight, alterations in circulating sex-hormone levels and breast cancer risk.
Pugeat, M. et al. Pathophysiology of sex hormone binding globulin (SHBG): relation to insulin. J. Steroid Biochem. Mol. Biol. 40, 841–849 (1991).
Kokkoris, P. & Pi-Sunyer, F. X. Obesity and endocrine disease. Endocrinol. Metab. Clin. North Am. 32, 895–914 (2003).
Kaaks, R., Lukanova, A. & Sommersberg, B. Plasma androgens, IGF-1, body size, and prostate cancer risk: a synthetic review. Prostate Cancer Prostatic Dis. 3, 157–172 (2000).
Strain, G. W. et al. Mild hypogonadotropic hypogonadism in obese men. Metabolism 31, 871–875 (1982).
Amatruda, J. M., Harman, S. M., Pourmotabbed, G. & Lockwood, D. H. Depressed plasma testosterone and fractional binding of testosterone in obese males. J. Clin. Endocrinol. Metab. 47, 268–271 (1978).
Poretsky, L., Cataldo, N. A., Rosenwaks, Z. & Giudice, L. C. The insulin-related ovarian regulatory system in health and disease. Endocr. Rev. 20, 535–582 (1999).
Ehrmann, D. A., Barnes, R. B. & Rosenfield, R. L. Polycystic ovary syndrome as a form of functional ovarian hyperandrogenism due to dysregulation of androgen secretion. Endocr. Rev. 16, 322–353 (1995).
Dunaif, A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocr. Rev. 18, 774–800 (1997).
Robinson, S. et al. The relationship of insulin insensitivity to menstrual pattern in women with hyperandrogenism and polycystic ovaries. Clin. Endocrinol. 39, 351–355 (1993).
Dickson, R. B. & Stancel, G. M. Estrogen receptor-mediated processes in normal and cancer cells. J. Natl Cancer Inst. Monogr. 27, 135–145 (2000).
Flototto, T. et al. Hormones and hormone antagonists: mechanisms of action in carcinogenesis of endometrial and breast cancer. Horm. Metab. Res. 33, 451–457 (2001).
Key, T. J. & Pike, M. C. The dose-effect relationship between 'unopposed' estrogens and endometrial mitotic rate: its central role in explaining and predicting endometrial cancer. Br. J. Cancer 57, 205–212 (1988).
Endogenous Hormones and Breast Cancer Collaborative Group. Endogenous sex hormones and breast cancer in postmenopausal women: reanalysis of nine prospective studies. J. Natl Cancer Inst. 94, 606–616 (2002).
Zeleniuch-Jacquotte, A. et al. Postmenopausal levels of oestrogen, androgen, and SHBG and breast cancer: long-term results of a prospective study. Br. J. Cancer 90, 153–159 (2004).
Coates, R. J. et al. Race, nutritional status, and survival from breast cancer. J. Natl Cancer Inst. 82, 1684–1692 (1990).
Tretli, S., Haldorsen, T. & Ottestad, L. The effect of pre-morbid height and weight on the survival of breast cancer patients. Br. J. Cancer 62, 299–303 (1990).
Maehle, B. O. & Tretli, S. Pre-morbid body-mass-index in breast cancer: reversed effect on survival in hormone receptor negative patients. Breast Cancer Res. Treat. 41, 123–130 (1996).
Lukanova, A. et al. Circulating levels of sex steroid hormones and risk of endometrial cancer in postmenopausal women. Int. J. Cancer 108, 425–432 (2004).
Zeleniuch-Jacquotte, A. et al. Postmenopausal endogenous oestrogens and risk of endometrial cancer: results of a prospective study. Br. J. Cancer 84, 975–981 (2001).
Hsing, A. W., Reichardt, J. K. & Stanczyk, F. Z. Hormones and prostate cancer: current perspectives and future directions. Prostate 52, 213–235 (2002).
Bosland, M. C. The role of steroid hormones in prostate carcinogenesis. J. Natl Cancer Inst. Monogr. 27, 39–66 (2000).
Harrison, S. A. & Diehl, A. M. Fat and the liver: a molecular overview. Semin. Gastroint. Dis. 13, 3–16 (2002).
Festi, D. et al. Hepatic steatosis in obese patients: clinical aspects and prognostic significance. Obes. Rev. 5, 27–42 (2004).
Hill, J. O., Wyatt, H. R., Reed, G. W. & Peters, J. C. Obesity and the environment: where do we go from here? Science 299, 853–855 (2003).
Allison, D. B., Fontaine, K. R., Manson, J. E., Stevens, J. & VanItallie, T. B. Annual deaths attributable to obesity in the United States. JAMA 282, 1530–1538 (1999).
Banegas, J. R., Lopez-Garcia, E., Gutierriz-Fisac, J. L., Guallar-Castillon, P. & Rodriguez-Artalejo, F. A simple estimate of mortality attributable to excess weight in the European Union. Eur. J. Clin. Nutr. 57, 201–208 (2003).
Finkelstein, E. A., Fiebelkorn, I. C. & Wang, G. State-level estimates of annual medical expenditures attributable to obesity. Obes. Res. 12, 18–24 (2004).
Sturm, R. The effects of obesity, smoking, and drinking on medical problems and costs. Health Affairs 21, 245–253 (2002).
Mokdad, A., Marks, J., Stroup, D. & Gerberding, J. Actual causes of death in the United States, 2000. JAMA 291, 1238–1245 (2004).
McTigue, K. M. et al. Screening and interventions for obesity in adults: summary of the evidence for the U. S. Preventive Services Task Force. Ann. Intern. Med. 139, 933–949 (2003). Current clinical guidelines to promote effective screening and intervention for obesity in adult populations, based on systematic reviews of all trials and observation studies of the health outcomes of obesity and efficacy of obesity treatment.
Chopra, M., Galbraith, S. & Darnton-Hill, I. A global response to a global problem: the epidemic of overnutrition. Bull. World Health Organ. 80, 952–958 (2002).
Byers, T. et al. American Cancer Society guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J. Clin. 52, 92–119 (2002).
Royal College of Physicians, Royal College of Paediatrics and Child Health, and Faculty of Public Health Medicine. Storing up problems: the medical case for a slimmer nation [online], <www.rcplondon.ac.uk/pubs/brochures/pub_print_SUP.htm> (2004).
Mercer, J. G., O'Reilly, J. M. & Morgan, P. J. Increasing the impact of European obesity research in preparation for the European research area: a report on the 2003 European Commission obesity workshop. Obes. Rev. 5, 79–86 (2004).
World Health Organization. Global strategy on diet, physical activity and health [online], <www.who.int/dietphysicalactivity/strategy/eb11344/en/> (2004).
Borkan, G., Hultz, D., Gerzof, S., Robbins, A. H. & Silbert, C. K. Age changes in body composition revealed by computed tomography. J. Gerontol. 38, 673–677 (1983).
US Department of Agriculture and US Department of Health and Human Services. Nutrition and your health: dietary guidelines for Americans (US Government Printing Office, Washington, DC, 2000).
WHO Expert Committee. Physical Status: the Use and Interpretation of Anthropometry. WHO Technical Report Series 854 (World Health Organization, Geneva, 1995).
National Institutes of Health and National Heart Lung and Blood Institute. Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults — the evidence report. Obes. Res. 6 (Suppl. 2), 51–209 (1998).
Rockhill, B., Newman, B. & Weinberg, C. Use and misuse of population attributable fractions. Am. J. Public Health 88, 15–19 (1998).
Bergman, R. N. & Ader, M. Free fatty acids and pathogenesis of type 2 diabetes mellitus. Trends Endocrinol. Metab. 11, 351–356 (2000).
Ebeling, P. & Koivisto, V. A. Non-esterified fatty acids regulate lipid and glucose oxidation and glycogen synthesis in healthy man. Diabetologia 37, 202–209 (1994).
Randle, P. J. Regulatory interactions between lipids and carbohydrates: the glucose fatty acid cycle after 35 years. Diabetes Metab. Rev. 14, 263–283 (1998).
Havel, P. J. Control of energy homeostasis and insulin action by adipocyte hormones: leptin, acylation stimulating protein, and adiponectin. Curr. Opin. Lipidol. 13, 51–59 (2002).
Hotamisligil, G. S. Molecular mechanisms of insulin resistance and the role of the adipocyte. Int. J. Obes. Relat. Metab. Disord. 24 (Suppl. 4), 23–27 (2000).
Kahn, B. B. & Flier, J. S. Obesity and insulin resistance. J. Clin. Invest. 106, 473–481 (2000). An excellent review of the physiological mechanisms leading to insulin resistance.
Steppan, C. M. & Lazar, M. A. Resistin and obesity-associated insulin resistance. Trends Endocrinol. Metab. 13, 18–23 (2002).
Trayhurn, P. & Beattie, J. H. Physiological role of adipose tissue: white adipose tissue as an endocrine and secretory organ. Proc. Nutr. Soc. 60, 329–339 (2001).
Ukropec, J., Sebokova, E. & Klimes, I. Nutrient sensing, leptin and insulin action. Arch. Physiol. Biochem. 109, 38–51 (2001).
Le Roith, D. & Zick, Y. Recent advances in our understanding of insulin action and insulin resistance. Diabetes Care 24, 588–597 (2001).
Moller, D. E. & Flier, J. S. Insulin resistance — mechanisms, syndromes, and implications. N. Engl. J. Med. 325, 938–948 (1991).
Virkamaki, A., Ueki, K. & Kahn, C. R. Protein–protein interaction in insulin signaling and the molecular mechanisms of insulin resistance. J. Clin. Invest. 103, 931–943 (1999).
Henriksen, E. J. Invited review: Effects of acute exercise and exercise training on insulin resistance. J. Appl. Physiol. 93, 788–796 (2002).
Borghouts, L. B. & Keizer, H. A. Exercise and insulin sensitivity: a review. Int. J. Sports Med. 21, 1–12 (2000).
Pedersen, O. Genetics of insulin resistance. Exp. Clin. Endocrinol. Diabetes 107, 113–118 (1999).
Grundy, S. M., Abate, N. & Chandalia, M. Diet composition and the metabolic syndrome: what is the optimal fat intake? Am. J. Med. 113 (Suppl. 9B), 25–29 (2002).
Kopp, W. High-insulinogenic nutrition — an etiologic factor for obesity and the metabolic syndrome? Metabolism 52, 840–844 (2003).
Vessby, B. Dietary fat, fatty acid composition in plasma and the metabolic syndrome. Curr. Opin. Lipidol. 14, 15–19 (2003).
Grundy, S. M., Brewer, H. B. Jr., Cleeman, J. I., Smith, S. C. Jr. & Lenfant, C. Definition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 109, 433–438 (2004).
Reaven, G. M. Pathophysiology of insulin resistance in human disease. Physiol. Rev. 75, 473–486 (1995).
National Center for Health Statistics, Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey 1999–2000 [online], <www.cdc.gov/nchs/about/major/nhanes/NHANES99_00.htm.> (2004)
Bergstrom, A., Pisani, P., Tenet, V., Wolk, A. & Adami, H. O. Overweight as an avoidable cause of cancer in Europe. Int. J. Cancer 91, 421–430 (2001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Cancer.gov
Entrez Gene
OMIM
gastro-oesophageal reflux disease
FURTHER INFORMATION
International Agency for Research on Cancer
National Center for Health Statistics
US Preventive Services Task Force
World Health Organization Global Strategy on Diet, Physical Activity and Health
Glossary
- TRIGLYCERIDE
-
A fat that is synthesized from carbohydrates and stored in animal adipose cells. On hydrolysis, it releases free fatty acids into the blood.
- ANTHROPOMETRIC
-
A measurement of the size or proportions of the human body; for example, weight, height and waist circumference.
- RELATIVE RISK
-
The risk of cancer (or other disease) in a group of exposed persons divided by the risk in a group of unexposed persons. The relative risk is a commonly used measure of association in epidemiological studies.
- CENTRAL ADIPOSITY
-
The storage of adipose tissue preferentially in adipocytes on or within the trunk rather than the extremities.
- ANOVULATORY
-
A menstrual cycle that is not accompanied by the discharge of an egg from the ovary.
- PROXY RESPONDERS
-
Also called surrogate responders, individuals who provide information regarding exposure in place of the individual involved in the study.
- SEX STEROIDS
-
A family of hormones that all share a basic chemical (steroidal) structure. These hormones include androgens, oestrogens and progesterone, and have important effects on sexual development and reproductive functions.
- FASTING AND NON-FASTING GLUCOSE LEVELS
-
Glucose is the end product of carbohydrate metabolism and the chief source of energy for living organisms; its utilization is controlled by insulin. It is found in the blood of all animals. Fasting plasma glucose levels are a measurement of the concentration of glucose in the plasma after the patient has not eaten for at least 8 hours.
- BIOAVAILABLE
-
The portion of a substance that can be used physiologically by target tissues.
- NON-ALCOHOLIC STEATOHEPATITIS.
-
A liver lesion characterized by liver cell injury and death, and hepatic inflammation that can be accompanied by progressive hepatic fibrosis.
Rights and permissions
About this article
Cite this article
Calle, E., Kaaks, R. Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat Rev Cancer 4, 579–591 (2004). https://doi.org/10.1038/nrc1408
Issue Date:
DOI: https://doi.org/10.1038/nrc1408
This article is cited by
-
White adipocyte dysfunction and obesity-associated pathologies in humans
Nature Reviews Molecular Cell Biology (2024)
-
Body mass index, triglyceride-glucose index, and prostate cancer death: a mediation analysis in eight European cohorts
British Journal of Cancer (2024)
-
Visceral adiposity in patients with lipomatous hypertrophy of the interatrial septum
Heart and Vessels (2024)
-
Pathological mechanisms of cold and mechanical stress in modulating cancer progression
Human Cell (2024)
-
Association of physical weight statuses defined by body mass index (BMI) with molecular subtypes of premenopausal breast cancer: a systematic review and meta-analysis
Breast Cancer Research and Treatment (2024)