Abstract
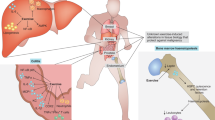
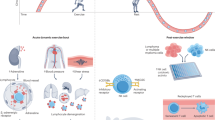
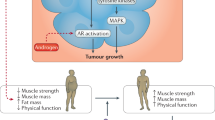
The integrity and composition of the tumour microenvironment (TME) is highly plastic, undergoing constant remodelling in response to instructive signals derived from alterations in the availability and nature of systemic host factors. This 'systemic milieu' is directly modulated by host exposure to modifiable lifestyle factors such as exercise. Host exposure to regular exercise markedly reduces the risk of the primary development of several cancers and might improve clinical outcomes following a diagnosis of a primary disease. However, the molecular mechanisms that underpin the apparent antitumour effects of exercise are poorly understood. In this Opinion article, we explore the putative effects of exercise in reprogramming the interaction between the host and the TME. Specifically, we speculate on the possible effects of exercise on reprogramming 'distant' tissue microenvironments (those not directly involved in the exercise response) by analysing how alterations in the systemic milieu might modulate key TME components to influence cancer hallmarks.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rozhok, A. I. & DeGregori, J. The evolution of lifespan and age-dependent cancer risk. Trends Cancer 2, 552–560 (2016).
Wu, S., Powers, S., Zhu, W. & Hannun, Y. A. Substantial contribution of extrinsic risk factors to cancer development. Nature 529, 43–47 (2016).
Govindan, R. et al. Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell 150, 1121–1134 (2012).
Jhappan, C., Noonan, F. P. & Merlino, G. Ultraviolet radiation and cutaneous malignant melanoma. Oncogene 22, 3099–3112 (2003).
[No authors listed.] TOBACCO and carcinoma of the lung. N. Engl. J. Med. 250, 125 (1954).
Richmond, B. G. & Jungers, W. L. Orrorin tugenensis femoral morphology and the evolution of hominin bipedalism. Science 319, 1662–1665 (2008).
Bramble, D. M. & Lieberman, D. E. Endurance running and the evolution of Homo. Nature 432, 345–352 (2004).
Booth, F. W., Chakravarthy, M. V. & Spangenburg, E. E. Exercise and gene expression: physiological regulation of the human genome through physical activity. J. Physiol. 543, 399–411 (2002).
Hopkins, B. D., Goncalves, M. D. & Cantley, L. C. Obesity and cancer mechanisms: cancer metabolism. J. Clin. Oncol. 34, 4277–4283 (2016).
Lee, I. M. et al. Effect of physical inactivity on major non-communicable diseases worldwide: an analysis of burden of disease and life expectancy. Lancet 380, 219–229 (2012).
Kohl, H. W. III et al. The pandemic of physical inactivity: global action for public health. Lancet 380, 294–305 (2012).
Bauman, A. et al. Tackling obesity: challenges ahead. Lancet 386, 741–742 (2015).
Calle, E. E., Rodriguez, C., Walker-Thurmond, K. & Thun, M. J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U. S. adults. N. Engl. J. Med. 348, 1625–1638 (2003).
Pontzer, H. et al. Hunter-gatherer energetics and human obesity. PLoS ONE 7, e40503 (2012).
Hallal, P. C. et al. Global physical activity levels: surveillance progress, pitfalls, and prospects. Lancet 380, 247–257 (2012).
World Health Organization. Global health risks: mortality and burden of disease attributable to selected major risks (WHO, 2009).
Myers, J. et al. Exercise capacity and mortality among men referred for exercise testing. N. Engl. J. Med. 346, 793–801 (2002).
Gulati, M. et al. The prognostic value of a nomogram for exercise capacity in women. N. Engl. J. Med. 353, 468–475 (2005).
Moore, S. C. et al. Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA Intern. Med. 176, 816–825 (2016).
Friedenreich, C. M., Neilson, H. K., Farris, M. S. & Courneya, K. S. Physical activity and cancer outcomes: a precision medicine approach. Clin. Cancer Res. 22, 4766–4775 (2016).
Betof, A. S., Dewhirst, M. W. & Jones, L. W. Effects and potential mechanisms of exercise training on cancer progression: a translational perspective. Brain Behav. Immun. 30, S75–S87 (2013).
Ashcraft, K. A., Peace, R. M., Betof, A. S., Dewhirst, M. W. & Jones, L. W. Efficacy and mechanisms of aerobic exercise on cancer initiation, progression, and metastasis: a critical systematic review of in vivo preclinical data. Cancer Res. 76, 4032–4050 (2016).
Neufer, P. D. et al. Understanding the cellular and molecular mechanisms of physical activity-induced health benefits. Cell Metab. 22, 4–11 (2015).
Jones, N. L. & Killian, K. J. Exercise limitation in health and disease. N. Engl. J. Med. 343, 632–641 (2000).
Jones, L. W., Eves, N. D., Haykowsky, M., Freedland, S. J. & Mackey, J. R. Exercise intolerance in cancer and the role of exercise therapy to reverse dysfunction. Lancet Oncol. 10, 598–605 (2009).
Levine, B. D. VO2, max: what do we know, and what do we still need to know? J. Physiol. 586, 25–34 (2008).
Peake, J. M. et al. Modulating exercise-induced hormesis: does less equal more? J. Appl. Physiol. 119, 172–189 (2015).
Hawley, J. A., Hargreaves, M., Joyner, M. J. & Zierath, J. R. Integrative biology of exercise. Cell 159, 738–749 (2014).
Fan, W. & Evans, R. M. Exercise Mimetics: Impact on Health and Performance. Cell. Metabolism 25, 242–247 (2017).
Egan, B., Hawley, J. A. & Zierath, J. R. SnapShot: exercise metabolism. Cell Metab. 24, 342–342.e1 (2016).
Lin, J., Handschin, C. & Spiegelman, B. M. Metabolic control through the PGC-1 family of transcription coactivators. Cell. Metabolism 1, 361–370 (2005).
Fan, W. & Evans, R. PPARs and ERRs: molecular mediators of mitochondrial metabolism. Curr. Opin. Cell Biol. 33, 49–54 (2015).
Dufour, C. R. et al. Genome-wide orchestration of cardiac functions by the orphan nuclear receptors ERRalpha and gamma. Cell. Metabolism 5, 345–356 (2007).
Kelly, D. P. & Scarpulla, R. C. Transcriptional regulatory circuits controlling mitochondrial biogenesis and function. Genes Dev. 18, 357–368 (2004).
Silveira, L. R., Pilegaard, H., Kusuhara, K., Curi, R. & Hellsten, Y. The contraction induced increase in gene expression of peroxisome proliferator-activated receptor (PPAR)-γ coactivator 1α (PGC-1α), mitochondrial uncoupling protein 3 (UCP3) and hexokinase II (HKII) in primary rat skeletal muscle cells is dependent on reactive oxygen species. Biochim. Biophys. Acta 1763, 969–976 (2006).
Irrcher, I., Ljubicic, V. & Hood, D. A. Interactions between ROS and AMP kinase activity in the regulation of PGC-1α transcription in skeletal muscle cells. Am. J. Physiol. Cell Physiol. 296, C116–C123 (2009).
Yan, Z., Okutsu, M., Akhtar, Y. N. & Lira, V. A. Regulation of exercise-induced fiber type transformation, mitochondrial biogenesis, and angiogenesis in skeletal muscle. J. Appl. Physiol. 110, 264–274 (2011).
Prior, B. M., Yang, H. T. & Terjung, R. L. What makes vessels grow with exercise training? J. Appl. Physiol. 97, 1119–1128 (2004).
Gustafsson, T. & Kraus, W. E. Exercise-induced angiogenesis-related growth and transcription factors in skeletal muscle, and their modification in muscle pathology. Front. Biosci. 6, D75–D89 (2001).
Arany, Z. et al. HIF-independent regulation of VEGF and angiogenesis by the transcriptional coactivator PGC-1α. Nature 451, 1008–1012 (2008).
Lindholm, M. E. & Rundqvist, H. Skeletal muscle hypoxia-inducible factor-1 and exercise. Exp. Physiol. 101, 28–32 (2016).
Semenza, G. L. Regulation of metabolism by hypoxia-inducible factor 1. Cold Spring Harb. Symp. Quant. Biol. 76, 347–353 (2011).
Fraisl, P., Aragones, J. & Carmeliet, P. Inhibition of oxygen sensors as a therapeutic strategy for ischaemic and inflammatory disease. Nat. Rev. Drug Discov. 8, 139–152 (2009).
Wong, B. W., Kuchnio, A., Bruning, U. & Carmeliet, P. Emerging novel functions of the oxygen-sensing prolyl hydroxylase domain enzymes. Trends Biochem. Sci. 38, 3–11 (2013).
Semenza, G. L. Oxygen sensing, homeostasis, and disease. N. Engl. J. Med. 365, 537–547 (2011).
Chazaud, B. Inflammation during skeletal muscle regeneration and tissue remodeling: application to exercise-induced muscle damage management. Immunol. Cell Biol. 94, 140–145 (2016).
Lieber, R. L., Thornell, L. E. & Friden, J. Muscle cytoskeletal disruption occurs within the first 15 min of cyclic eccentric contraction. J. Appl. Physiol. 80, 278–284 (1996).
Nunes-Silva, A. et al. Treadmill exercise induces neutrophil recruitment into muscle tissue in a reactive oxygen species-dependent manner. An intravital microscopy study. PLoS ONE 9, e96464 (2014).
Fielding, R. A. et al. Acute phase response in exercise. III. Neutrophil and IL-1ß accumulation in skeletal muscle. Am. J. Physiol. 265, R166–R172 (1993).
Marklund, P. et al. Extensive inflammatory cell infiltration in human skeletal muscle in response to an ultraendurance exercise bout in experienced athletes. J. Appl. Physiol. 114, 66–72 (1985).
Tidball, J. G. Regulation of muscle growth and regeneration by the immune system. Nat. Rev. Immunol. 17, 165–178 (2017).
Neubauer, O. et al. Time course-dependent changes in the transcriptome of human skeletal muscle during recovery from endurance exercise: from inflammation to adaptive remodeling. J. Appl. Physiol. 116, 274–287 (2014).
Peake, J. M., Neubauer, O., Walsh, N. P. & Simpson, R. J. Recovery of the immune system after exercise. J. Appl. Physiol. 122, 1077–1087 (1985).
Arnold, L. et al. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J. Exp. Med. 204, 1057–1069 (2007).
Deng, B., Wehling-Henricks, M., Villalta, S. A., Wang, Y. & Tidball, J. G. IL-10 triggers changes in macrophage phenotype that promote muscle growth and regeneration. J. Immunol. 189, 3669–3680 (2012).
Tonkin, J. et al. Monocyte/macrophage-derived IGF-1 orchestrates murine skeletal muscle regeneration and modulates autocrine polarization. Mol. Ther. 23, 1189–1200 (2015).
Ginhoux, F., Schultze, J. L., Murray, P. J., Ochando, J. & Biswas, S. K. New insights into the multidimensional concept of macrophage ontogeny, activation and function. Nat. Immunol. 17, 34–40 (2016).
Goh, J., Goh, K. P. & Abbasi, A. Exercise and adipose tissue macrophages: new frontiers in obesity research? Front. Endocrinol. (Lausanne) 7, 65 (2016).
Furrer, R., Eisele, P. S., Schmidt, A., Beer, M. & Handschin, C. Paracrine cross-talk between skeletal muscle and macrophages in exercise by PGC-1α-controlled BNP. Sci. Rep. 7, 40789 (2017).
Kotas, M. E. & Medzhitov, R. Homeostasis, inflammation, and disease susceptibility. Cell 160, 816–827 (2015).
Karsenty, G. & Olson, E. N. Bone and muscle endocrine functions: unexpected paradigms of inter-organ communication. Cell 164, 1248–1256 (2016).
Karsenty, G. & Ferron, M. The contribution of bone to whole-organism physiology. Nature 481, 314–320 (2012).
Safdar, A., Saleem, A. & Tarnopolsky, M. A. The potential of endurance exercise-derived exosomes to treat metabolic diseases. Nat. Rev. Endocrinol. 12, 504–517 (2016).
Goodwin, P. J., Meyerhardt, J. A. & Hursting, S. D. Host factors and cancer outcome. J. Clin. Oncol. 28, 4019–4021 (2010).
McTiernan, A. Mechanisms linking physical activity with cancer. Nat. Rev. Cancer 8, 205–211 (2008).
Hanahan, D. & Coussens, L. M. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21, 309–322 (2012).
Aaronson, S. A. Growth factors and cancer. Science 254, 1146–1153 (1991).
Rathmell, J. C., Vander Heiden, M. G., Harris, M. H., Frauwirth, K. A. & Thompson, C. B. In the absence of extrinsic signals, nutrient utilization by lymphocytes is insufficient to maintain either cell size or viability. Mol. Cell 6, 683–692 (2000).
Pavlova, N. N. & Thompson, C. B. The emerging hallmarks of cancer metabolism. Cell Metab. 23, 27–47 (2016).
Cairns, R. A., Harris, I. S. & Mak, T. W. Regulation of cancer cell metabolism. Nat. Rev. Cancer 11, 85–95 (2011).
Koppenol, W. H., Bounds, P. L. & Dang, C. V. Otto Warburg's contributions to current concepts of cancer metabolism. Nat. Rev. Cancer 11, 325–337 (2011).
Raitakari, O. T., Porkka, K. V., Rasanen, L. & Viikari, J. S. Relations of life-style with lipids, blood pressure and insulin in adolescents and young adults. The cardiovascular risk young finns study. Atherosclerosis 111, 237–246 (1994).
Barnard, R. J. Effects of life-style modification on serum lipids. Arch. Intern. Med. 151, 1389–1394 (1991).
Stanford, K. I. & Goodyear, L. J. Exercise and type 2 diabetes: molecular mechanisms regulating glucose uptake in skeletal muscle. Adv. Physiol. Educ. 38, 308–314 (2014).
Irwin, M. L. et al. Randomized controlled trial of aerobic exercise on insulin and insulin-like growth factors in breast cancer survivors: the Yale Exercise and Survivorship study. Cancer Epidemiol. 18, 306–313 (2009).
Fairey, A. S. et al. Effects of exercise training on fasting insulin, insulin resistance, insulin-like growth factors, and insulin-like growth factor binding proteins in postmenopausal breast cancer survivors: a randomized controlled trial. Cancer Epidemiol. 12, 721–727 (2003).
Zhu, Z. et al. Effect of nonmotorized wheel running on mammary carcinogenesis: circulating biomarkers, cellular processes, and molecular mechanisms in rats. Cancer Epidemiol. 17, 1920–1929 (2008).
Thompson, H. J., Wolfe, P., McTiernan, A., Jiang, W. & Zhu, Z. Wheel running-induced changes in plasma biomarkers and carcinogenic response in the 1-methyl-1-nitrosourea-induced rat model for breast cancer. Cancer Prevention Res. 3, 1484–1492 (2010).
Xie, L. et al. Effects of dietary calorie restriction or exercise on the PI3K and Ras signaling pathways in the skin of mice. J. Biol. Chem. 282, 28025–28035 (2007).
Glass, O. et al. Differential response to exercise in claudin-low breast cancer. Oncotarget (in the press).
Morikawa, T. et al. Association of CTNNB1 (ß-catenin) alterations, body mass index, and physical activity with survival in patients with colorectal cancer. JAMA 305, 1685–1694 (2011).
Klaus, A. & Birchmeier, W. Wnt signalling and its impact on development and cancer. Nat. Rev. Cancer 8, 387–398 (2008).
Hanyuda, A. et al. Survival benefit of exercise differs by tumor IRS1 expression status in colorectal cancer. Ann. Surg. Oncol. 23, 908–917 (2016).
Slattery, M. L. et al. Associations among IRS1, IRS2, IGF1, and IGFBP3 genetic polymorphisms and colorectal cancer. Cancer Epidemiol. 13, 1206–1214 (2004).
Pollak, M. Insulin and insulin-like growth factor signalling in neoplasia. Nat. Rev. Cancer 8, 915–928 (2008).
Kalaany, N. Y. & Sabatini, D. M. Tumours with PI3K activation are resistant to dietary restriction. Nature 458, 725–731 (2009).
Jiang, X., Overholtzer, M. & Thompson, C. B. Autophagy in cellular metabolism and cancer. J. Clin. Invest. 125, 47–54 (2015).
Amaravadi, R. & Debnath, J. Mouse models address key concerns regarding autophagy inhibition in cancer therapy. Cancer Discov. 4, 873–875 (2014).
Yang, A. et al. Autophagy is critical for pancreatic tumor growth and progression in tumors with p53 alterations. Cancer Discov. 4, 905–913 (2014).
White, E. Deconvoluting the context-dependent role for autophagy in cancer. Nat. Rev. Cancer 12, 401–410 (2012).
He, C. et al. Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis. Nature 481, 511–515 (2012).
He, C., Sumpter, R. Jr & Levine, B. Exercise induces autophagy in peripheral tissues and in the brain. Autophagy 8, 1548–1551 (2012).
Aveseh, M., Nikooie, R. & Aminaie, M. Exercise-induced changes in tumour LDH-B and MCT1 expression are modulated by oestrogen-related receptor alpha in breast cancer-bearing BALB/c mice. J. Physiol. 593, 2635–2648 (2015).
Semenza, G. L. Targeting HIF-1 for cancer therapy. Nat. Rev. Cancer 3, 721–732 (2003).
Carmeliet, P. & Jain, R. K. Angiogenesis in cancer and other diseases. Nature 407, 249–257 (2000).
Betof, A. S. et al. Modulation of murine breast tumor vascularity, hypoxia and chemotherapeutic response by exercise. J. Natl Cancer Inst. 107, djv040 (2015).
Jones, L. W. et al. Effect of aerobic exercise on tumor physiology in an animal model of human breast cancer. J. Appl. Physiol. 108, 343–348 (2010).
McCullough, D. J., Stabley, J. N., Siemann, D. W. & Behnke, B. J. Modulation of blood flow, hypoxia, and vascular function in orthotopic prostate tumors during exercise. J. Natl Cancer Inst. 106, dju036 (2014).
Schadler, K. L. et al. Tumor vessel normalization after aerobic exercise enhances chemotherapeutic efficacy. Oncotarget 7, 65429–65440 (2016).
Kashiwagi, S. et al. Perivascular nitric oxide gradients normalize tumor vasculature. Nat. Med. 14, 255–257 (2008).
Fukumura, D., Kashiwagi, S. & Jain, R. K. The role of nitric oxide in tumour progression. Nat. Rev. Cancer 6, 521–534 (2006).
Psaila, B. & Lyden, D. The metastatic niche: adapting the foreign soil. Nat. Rev. Cancer 9, 285–293 (2009).
Erler, J. T. et al. Hypoxia-induced lysyl oxidase is a critical mediator of bone marrow cell recruitment to form the premetastatic niche. Cancer Cell 15, 35–44 (2009).
Cox, T. R. et al. The hypoxic cancer secretome induces pre-metastatic bone lesions through lysyl oxidase. Nature 522, 106–110 (2015).
Sceneay, J. et al. Primary tumor hypoxia recruits CD11b+/Ly6Cmed/Ly6G+ immune suppressor cells and compromises NK cell cytotoxicity in the premetastatic niche. Cancer Res. 72, 3906–3911 (2012).
Chafe, S. C. et al. Carbonic anhydrase IX promotes myeloid-derived suppressor cell mobilization and establishment of a metastatic niche by stimulating G-CSF production. Cancer Res. 75, 996–1008 (2015).
Quail, D. F. & Joyce, J. A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19, 1423–1437 (2013).
Gao, D. et al. Endothelial progenitor cells control the angiogenic switch in mouse lung metastasis. Science 319, 195–198 (2008).
Jones, L. W. et al. Modulation of circulating angiogenic factors and tumor biology by aerobic training in breast cancer patients receiving neoadjuvant chemotherapy. Cancer Prevention Res. 6, 925–937 (2013).
Pearson, M. J. & Smart, N. A. Effect of exercise training on endothelial function in heart failure patients: a systematic review meta-analysis. Int. J. Cardiol. 231, 234–243 (2017).
Jain, R. K. Normalization of tumor vasculature: an emerging concept in antiangiogenic therapy. Science 307, 58–62 (2005).
Hatfield, S. M. et al. Immunological mechanisms of the antitumor effects of supplemental oxygenation. Sci. Trans. Med. 7, 277ra230 (2015).
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Kruger, K., Lechtermann, A., Fobker, M., Volker, K. & Mooren, F. C. Exercise-induced redistribution of T lymphocytes is regulated by adrenergic mechanisms. Brain Behav. Immun. 22, 324–338 (2008).
Walsh, N. P. et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 17, 6–63 (2011).
Timmerman, K. L., Flynn, M. G., Coen, P. M., Markofski, M. M. & Pence, B. D. Exercise training-induced lowering of inflammatory (CD14+CD16+) monocytes: a role in the anti-inflammatory influence of exercise? J. Leukoc. Biol. 84, 1271–1278 (2008).
Sloan, R. P. et al. Aerobic exercise attenuates inducible TNF production in humans. J. Appl. Physiol. 103, 1007–1011 (1985).
Pyne, D. B. et al. Effects of an intensive 12-wk training program by elite swimmers on neutrophil oxidative activity. Med. Sci. Sports Exerc. 27, 536–542 (1995).
Hack, V., Strobel, G., Weiss, M. & Weicker, H. PMN cell counts and phagocytic activity of highly trained athletes depend on training period. J. Appl. Physiol. 77, 1731–1735 (1985).
Woods, J. A. et al. Effects of 6 months of moderate aerobic exercise training on immune function in the elderly. Mech. Ageing Dev. 109, 1–19 (1999).
Kawanishi, N., Yano, H., Yokogawa, Y. & Suzuki, K. Exercise training inhibits inflammation in adipose tissue via both suppression of macrophage infiltration and acceleration of phenotypic switching from M1 to M2 macrophages in high-fat-diet-induced obese mice. Exerc. Immunol. Rev. 16, 105–118 (2010).
Kizaki, T. et al. Adaptation of macrophages to exercise training improves innate immunity. Biochem. Biophys. Res. Commun. 372, 152–156 (2008).
Sugiura, H., Nishida, H., Sugiura, H. & Mirbod, S. M. Immunomodulatory action of chronic exercise on macrophage and lymphocyte cytokine production in mice. Acta Physiol. Scand. 174, 247–256 (2002).
Engblom, C., Pfirschke, C. & Pittet, M. J. The role of myeloid cells in cancer therapies. Nat. Rev. Cancer 16, 447–462 (2016).
Abdalla, D. R., Aleixo, A. A., Murta, E. F. & Michelin, M. A. Innate immune response adaptation in mice subjected to administration of DMBA and physical activity. Oncol. Lett. 7, 886–890 (2014).
Murphy, E. A. et al. Effects of moderate exercise and oat beta-glucan on lung tumor metastases and macrophage antitumor cytotoxicity. J. Appl. Physiol. 97, 955–959 (1985).
Noy, R. & Pollard, J. W. Tumor-associated macrophages: from mechanisms to therapy. Immunity 41, 49–61 (2014).
Bai, J. et al. Contact-dependent carcinoma aggregate dispersion by M2a macrophages via ICAM-1 and ß2 integrin interactions. Oncotarget 6, 25295–25307 (2015).
Almeida, P. W. et al. Swim training suppresses tumor growth in mice. J. Appl. Physiol. 107, 261–265 (1985).
Zielinski, M. R., Muenchow, M., Wallig, M. A., Horn, P. L. & Woods, J. A. Exercise delays allogeneic tumor growth and reduces intratumoral inflammation and vascularization. J. Appl. Physiol. 96, 2249–2256 (1985).
McClellan, J. L. et al. Exercise effects on polyp burden and immune markers in the ApcMin/+ mouse model of intestinal tumorigenesis. Int. J. Oncol. 45, 861–868 (2014).
Fairey, A. S. et al. Randomized controlled trial of exercise and blood immune function in postmenopausal breast cancer survivors. J. Appl. Physiol. 98, 1534–1540 (1985).
Pedersen, L. et al. Voluntary running suppresses tumor growth through epinephrine- and IL-6-Dependent NK cell mobilization and redistribution. Cell Metab. 23, 554–562 (2016).
Simpson, R. J., Florida-James, G. D., Whyte, G. P. & Guy, K. The effects of intensive, moderate and downhill treadmill running on human blood lymphocytes expressing the adhesion/activation molecules CD54 (ICAM-1), CD18 (ß2 integrin) and CD53. Eur. J. Appl. Physiol. 97, 109–121 (2006).
LaVoy, E. C., Bosch, J. A., Lowder, T. W. & Simpson, R. J. Acute aerobic exercise in humans increases cytokine expression in CD27− but not CD27+ CD8+ T-cells. Brain Behav. Immun. 27, 54–62 (2013).
Silva, L. C. et al. Moderate and intense exercise lifestyles attenuate the effects of aging on telomere length and the survival and composition of T cell subpopulations. Age (Dordr) 38, 24 (2016).
Wang, J. et al. Effect of exercise training intensity on murine T-regulatory cells and vaccination response. Scand. J. Med. Sci. Sports 22, 643–652 (2012).
Glass, O. K. et al. Effect of aerobic training on the host systemic milieu in patients with solid tumours: an exploratory correlative study. Br. J. Cancer 112, 825–831 (2015).
Angelin, A. et al. Foxp3 reprograms T cell metabolism to function in low-glucose, high-lactate environments. Cell. Metab. 25, 1282–1293.e7 (2017).
Facciabene, A. et al. Tumour hypoxia promotes tolerance and angiogenesis via CCL28 and Treg cells. Nature 475, 226–230 (2011).
Carmona-Fontaine, C. et al. Emergence of spatial structure in the tumor microenvironment due to the Warburg effect. Proc. Natl Acad. Sci. USA 110, 19402–19407 (2013).
Colegio, O. R. et al. Functional polarization of tumour-associated macrophages by tumour-derived lactic acid. Nature 513, 559–563 (2014).
Carmona-Fontaine, C. et al. Metabolic origins of spatial organization in the tumor microenvironment. Proc. Natl Acad. Sci. USA 114, 2934–2939 (2017).
Roodman, G. D. Mechanisms of bone metastasis. N. Engl. J. Med. 350, 1655–1664 (2004).
Jones, L. W. Precision oncology framework for investigation of exercise as treatment for cancer. J. Clin. Oncol. 33, 4134–4137 (2015).
Schmitz, K. H. et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med. Sci. Sports Exerc. 42, 1409–1426 (2010).
Sasso, J. P. et al. A framework for prescription in exercise-oncology research. J. Cachexia Sarcopenia Muscle 6, 115–124 (2015).
van Waart, H. et al. Effect of low-intensity physical activity and moderate- to high-intensity physical exercise during adjuvant chemotherapy on physical fitness, fatigue, and chemotherapy completion rates: results of the PACES randomized clinical trial. J. Clin. Oncol. 33, 1918–1927 (2015).
Courneya, K. S. et al. Effects of exercise dose and type during breast cancer chemotherapy: multicenter randomized trial. J. Natl Cancer Inst. 105, 1821–1832 (2013).
Courneya, K. S. et al. Effects of aerobic and resistance exercise in breast cancer patients receiving adjuvant chemotherapy: a multicenter randomized controlled trial. J. Clin. Oncol. 25, 4396–4404 (2007).
Courneya, K. S. et al. The colon health and life-long exercise change trial: a randomized trial of the national cancer institute of canada clinical trials group. Curr. Oncol. 15, 279–285 (2008).
Kitano, H. Biological robustness. Nat. Rev. Genet. 5, 826–837 (2004).
Kitano, H. Cancer robustness: tumour tactics. Nature 426, 125 (2003).
Greenhaff, P. L. & Hargreaves, M. 'Systems biology' in human exercise physiology: is it something different from integrative physiology? J. Physiol. 589, 1031–1036 (2011).
Baker, J. M., De Lisio, M. & Parise, G. Endurance exercise training promotes medullary hematopoiesis. FASEB J. 25, 4348–4357 (2011).
David, V. et al. Mechanical loading down-regulates peroxisome proliferator-activated receptor gamma in bone marrow stromal cells and favors osteoblastogenesis at the expense of adipogenesis. Endocrinology 148, 2553–2562 (2007).
Wallace, J. M., Ron, M. S. & Kohn, D. H. Short-term exercise in mice increases tibial post-yield mechanical properties while two weeks of latency following exercise increases tissue-level strength. Calcif. Tissue Int. 84, 297–304 (2009).
Pichler, K. et al. RANKL is downregulated in bone cells by physical activity (treadmill and vibration stimulation training) in rat with glucocorticoid-induced osteoporosis. Histol. Histopathol. 28, 1185–1196 (2013).
Croucher, P. I., McDonald, M. M. & Martin, T. J. Bone metastasis: the importance of the neighbourhood. Nat. Rev. Cancer 16, 373–386 (2016).
De Lisio, M. & Parise, G. Characterization of the effects of exercise training on hematopoietic stem cell quantity and function. J. Appl. Physiol. 113, 1576–1584 (1985).
Reagan, M. R. & Rosen, C. J. Navigating the bone marrow niche: translational insights and cancer-driven dysfunction. Nat. Rev. Rheumatol 12, 154–168 (2016).
Roodman, G. D. Genes associate with abnormal bone cell activity in bone metastasis. Cancer Metastasis Rev. 31, 569–578 (2012).
De Lisio, M., Baker, J. M. & Parise, G. Exercise promotes bone marrow cell survival and recipient reconstitution post-bone marrow transplantation, which is associated with increased survival. Exp. Hematol. 41, 143–154 (2013).
Shiozawa, Y. et al. Human prostate cancer metastases target the hematopoietic stem cell niche to establish footholds in mouse bone marrow. J. Clin. Invest. 121, 1298–1312 (2011).
Pagnotti, G. M. & Styner, M. Exercise regulation of marrow adipose tissue. Front. Endocrinol. (Lausanne) 7, 94 (2016).
Emmons, R., Niemiro, G. M., Owolabi, O. & De Lisio, M. Acute exercise mobilizes hematopoietic stem and progenitor cells and alters the mesenchymal stromal cell secretome. J. Appl. Physiol. 120, 624–632 (2016) (1985).
Liu, F., Poursine-Laurent, J. & Link, D. C. Expression of the G-CSF receptor on hematopoietic progenitor cells is not required for their mobilization by G-CSF. Blood 95, 3025–3031 (2000).
Hardaway, A. L., Herroon, M. K., Rajagurubandara, E. & Podgorski, I. Bone marrow fat: linking adipocyte-induced inflammation with skeletal metastases. Cancer Metastasis Rev. 33, 527–543 (2014).
Fazeli, P. K. et al. Marrow fat and bone—new perspectives. J. Clin. Endocrinol. Metab. 98, 935–945 (2013).
American Thoracic Society & American College of Chest Physicians. ATS/ACCP Statement on cardiopulmonary exercise testing. Am. J. Respir. Crit. Care Med. 167, 211–277 (2003).
Davis, J. A. Anaerobic threshold: review of the concept and directions for future research. Med. Sci. Sports Exerc. 17, 6–21 (1985).
Convertino, V. A. Blood volume response to physical activity and inactivity. Am. J. Med. Sci. 334, 72–79 (2007).
Bishop, D. J., Granata, C. & Eynon, N. Can we optimise the exercise training prescription to maximise improvements in mitochondria function and content? Biochim. Biophys. Acta 1840, 1266–1275 (2014).
MacDougall, J. D. et al. Muscle performance and enzymatic adaptations to sprint interval training. J. Appl. Physiol. 84, 2138–2142 (1985).
Hickson, R. C., Hagberg, J. M., Ehsani, A. A. & Holloszy, J. O. Time course of the adaptive responses of aerobic power and heart rate to training. Med. Sci. Sports Exerc. 13, 17–20 (1981).
Kreher, J. B. & Schwartz, J. B. Overtraining syndrome: a practical guide. Sports Health 4, 128–138 (2012).
Fry, R. W., Morton, A. R. & Keast, D. Periodisation of training stress—a review. Can. J. Sport Sci. 17, 234–240 (1992).
Fry, R. W., Morton, A. R. & Keast, D. Periodisation and the prevention of overtraining. Can. J. Sport Sci. 17, 241–248 (1992).
Rusch, H. P. & Kline, B. E. The effect of exercise on the growth of a mouse tumor. Cancer Res. 4, 116–118 (1944).
Winningham, M. L., MacVicar, M. G., Bondoc, M., Anderson, J. I. & Minton, J. P. Effect of aerobic exercise on body weight and composition in patients with breast cancer on adjuvant chemotherapy. Oncol. Nurs. Forum 16, 683–689 (1989).
Segal, R. et al. Structured exercise improves physical functioning in women with stages I and II breast cancer: results of a randomized controlled trial. J. Clin. Oncol. 19, 657–665 (2001).
Brown, J. K. et al. Nutrition and physical activity during and after cancer treatment: an American Cancer Society guide for informed choices. CA Cancer J. Clin. 53, 268–291 (2003).
Holmes, M. D., Chen, W. Y., Feskanich, D., Kroenke, C. H. & Colditz, G. A. Physical activity and survival after breast cancer diagnosis. JAMA 293, 2479–2486 (2005).
Meyerhardt, J. A. et al. Interaction of molecular markers and physical activity on mortality in patients with colon cancer. Clin. Cancer Res. 15, 5931–5936 (2009).
Acknowledgements
L.W.J. is supported by research grants from the National Cancer Institute, AKTIV Against Cancer and the Memorial Sloan Kettering Cancer Center Support Grant/Core Grant (P30 CA008748). The authors would like to thank N. Eves and anonymous reviewers for insights and feedback on earlier versions of this manuscript as well as W. Underwood for administrative support. The authors apologize to the many authors whose work we were unable to cite owing to space constraints.
Author information
Authors and Affiliations
Contributions
G.J.K., D.F.Q. and L.W.J. researched data for the article, made substantial contributions to the discussion of content and wrote the manuscript. All authors reviewed and edited the manuscript before submission. G.J.K. and D.F.Q. are joint first authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Caloric restriction
-
Reduction in calorie consumption.
- Cardiorespiratory fitness
-
The integrative capacity of the pulmonary and cardiovascular systems, the vasculature, blood and skeletal muscle to deliver and/or utilize oxygen from the environment to the skeletal muscle mitochondria.
- Chronic exercise
-
Structured, repeated and purposeful physical activity with the objective of improving health or cardiorespiratory fitness.
- Exercise prescription
-
A specific regimen with details regarding the frequency, duration, modality, intensity and length of exercise training.
- Homeostasis
-
The need to maintain a stable internal environment in the face of pathological and physiological perturbations.
- Homeostatic control circuits
-
Specialized homeostatic systems that operate at the cellular, tissue and systemic levels to regulate physiological processes within predefined ranges ('set points').
- Pathological angiogenesis
-
Dysfunctional or non-productive angiogenesis that results in pathological phenotypes.
- Physical activity
-
Any bodily movement produced by skeletal muscles that requires energy expenditure above resting levels.
- Physiological angiogenesis
-
Functional or productive angiogenesis that results in physiological phenotypes.
- Pre-metastatic niche
-
Secondary organ microenvironments (for example, lung, liver, brain or bone) that undergo molecular and possibly architectural alterations to augment engraftment and survival of subsequent tumour cell arrival.
Rights and permissions
About this article
Cite this article
Koelwyn, G., Quail, D., Zhang, X. et al. Exercise-dependent regulation of the tumour microenvironment. Nat Rev Cancer 17, 620–632 (2017). https://doi.org/10.1038/nrc.2017.78
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2017.78
This article is cited by
-
Effect of home-based online training and activity feedback on oxygen uptake in patients after surgical cancer therapy: a randomized controlled trial
BMC Medicine (2023)
-
FAM171B stabilizes vimentin and enhances CCL2-mediated TAM infiltration to promote bladder cancer progression
Journal of Experimental & Clinical Cancer Research (2023)
-
The effect of acute exercise on circulating immune cells in newly diagnosed breast cancer patients
Scientific Reports (2023)
-
Renal enhanced CT images reveal the tandem mechanism between tumor cells and immunocytes based on bulk/single-cell RNA sequencing
Functional & Integrative Genomics (2023)
-
Cancer Stem Cells and the Tumor Microenvironment in Tumor Drug Resistance
Stem Cell Reviews and Reports (2023)