Key Points
-
Malignant mesothelioma is a universally lethal cancer that usually affects the pleura, and it is increasing in incidence worldwide. Carcinogenesis in this disease is unique in that the direct causal relationship between exposure to the causative environmental carcinogen, asbestos, and the development of mesothelioma is so well established.
-
Currently, platinum–antifolate combination chemotherapy remains the only established treatment, with pemetrexed and cisplatin combination chemotherapy the only licensed regimen. Here, despite tumour response rates of 45.5%, as well as improvements in progression-free survival of 6.1 months, overall survival of 13.3 months and cancer-related symptoms, benefits are usually modest at best and prognosis remains poor, with population-level survival estimated at approximately 8 months, as many patients are unfit for active treatment.
-
There has been recent exponential growth in our understanding of mesothelioma pathobiology, with an improvement in our knowledge of mesothelioma genetics, epigenetics, tumour microenvironment and immunobiology. The translational outputs from these data have now led to the discovery and development of promising therapeutic strategies.
-
Extensive interrogation of the mesothelioma genome has revealed the most frequent mutational events to involve tumour suppressor inactivation, mediated by multiple mechanisms, which include single nucleotide variation, copy number losses, gene fusions and splicing alterations. Tumour suppressors commonly inactivated include those encoded by cyclin-dependent kinase inhibitor 2A (CDKN2A), BRCA1 associated protein 1 (BAP1) and neurofibromin 2 (NF2).
-
There are now several promising novel antitumour agents under investigation in mesothelioma, including mesothelin-targeted therapies, arginine deprivation in arginosuccinate synthetase 1-deficient mesothelioma and immunotherapeutics such as immune checkpoint inhibitors.
Abstract
Malignant mesothelioma is a universally lethal cancer that is increasing in incidence worldwide. There is a dearth of effective therapies, with only one treatment (pemetrexed and cisplatin combination chemotherapy) approved in the past 13 years. However, the past 5 years have witnessed an exponential growth in our understanding of mesothelioma pathobiology, which is set to revolutionize therapeutic strategies. From a genomic standpoint, mesothelioma is characterized by a preponderance of tumour suppressor alterations, for which novel therapies are currently in development. Other promising antitumour agents include inhibitors against angiogenesis, mesothelin and immune checkpoints, which are at various phases of clinical trial testing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bianchi, C. & Bianchi, T. Malignant mesothelioma: global incidence and relationship with asbestos. Ind. Health 45, 379–387 (2007).
Mutsaers, S. E. The mesothelial cell. Int. J. Biochem. Cell Biol. 36, 9–16 (2004).
Wagner, J. C., Sleggs, C. A. & Marchand, P. Diffuse pleural mesothelioma and asbestos exposure in the North Western Cape Province. Br. J. Ind. Med. 17, 260–271 (1960).
Carbone, M. et al. Malignant mesothelioma: facts, myths, and hypotheses. J. Cell. Physiol. 227, 44–58 (2012).
Farioli, A. et al. Radiation-induced mesothelioma among long-term solid cancer survivors: a longitudinal analysis of SEER database. Cancer Med. 5, 950–959 (2016).
Peto, J., Hodgson, J. T., Matthews, F. E. & Jones, J. R. Continuing increase in mesothelioma mortality in Britain. Lancet 345, 535–539 (1995).
Pott, F. et al. Carcinogenicity studies on fibres, metal compounds, and some other dusts in rats. Exp. Pathol. 32, 129–152 (1987).
Boutin, C. & Rey, F. Thoracoscopy in pleural malignant mesothelioma: a prospective study of 188 consecutive patients. Part 1: diagnosis. Cancer 72, 389–393 (1993).
Choe, N., Tanaka, S. & Kagan, E. Asbestos fibers and interleukin-1 upregulate the formation of reactive nitrogen species in rat pleural mesothelial cells. Am. J. Respir. Cell Mol. Biol. 19, 226–236 (1998).
Huang, S. X., Jaurand, M. C., Kamp, D. W., Whysner, J. & Hei, T. K. Role of mutagenicity in asbestos fiber-induced carcinogenicity and other diseases. J. Toxicol. Environ. Health B Crit. Rev. 14, 179–245 (2011).
Zanella, C. L., Posada, J., Tritton, T. R. & Mossman, B. T. Asbestos causes stimulation of the extracellular signal-regulated kinase 1 mitogen-activated protein kinase cascade after phosphorylation of the epidermal growth factor receptor. Cancer Res. 56, 5334–5338 (1996).
Marzo, A. L., Fitzpatrick, D. R., Robinson, B. W. & Scott, B. Antisense oligonucleotides specific for transforming growth factor beta2 inhibit the growth of malignant mesothelioma both in vitro and in vivo. Cancer Res. 57, 3200–3207 (1997).
Yang, H. et al. Programmed necrosis induced by asbestos in human mesothelial cells causes high-mobility group box 1 protein release and resultant inflammation. Proc. Natl Acad. Sci. USA 107, 12611–12616 (2010).
Qi, F. et al. Continuous exposure to chrysotile asbestos can cause transformation of human mesothelial cells via HMGB1 and TNF-alpha signaling. Am. J. Pathol. 183, 1654–1666 (2013).
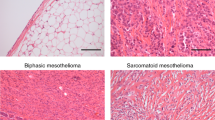
Attanoos, R. L. & Gibbs, A. R. Pathology of malignant mesothelioma. Histopathology 30, 403–418 (1997).
Travis, W. D., Brambilla, E., Burke, A., Marx, A. & Nicholson, A. G. WHO Classification of Tumours of the Lung, Pleura, Thymus and Heart (WHO, 2015).
Kao, S. C. et al. Accuracy of diagnostic biopsy for the histological subtype of malignant pleural mesothelioma. J. Thorac. Oncol. 6, 602–605 (2011).
Comertpay, S. et al. Evaluation of clonal origin of malignant mesothelioma. J. Transl Med. 12, 301 (2014).
National Lung Cancer Audit. National Lung Cancer Audit Report 2014: Mesothelioma Report for the Period 2008–2012 (Health and Social Care Information Centre, 2014).
American Cancer Society. Survival statistics for mesothelioma. Cancer.org http://www.cancer.org/cancer/malignantmesothelioma/detailedguide/malignant-mesothelioma-survival-statistics (2016).
Fennell, D. A. et al. Statistical validation of the EORTC prognostic model for malignant pleural mesothelioma based on three consecutive phase II trials. J. Clin. Oncol. 23, 184–189 (2005).
van Meerbeeck, J. P. et al. Randomized phase III study of cisplatin with or without raltitrexed in patients with malignant pleural mesothelioma: an intergroup study of the European Organisation for Research and Treatment of Cancer Lung Cancer Group and the National Cancer Institute of Canada. J. Clin. Oncol. 23, 6881–6889 (2005).
Vogelzang, N. J. et al. Phase III study of pemetrexed in combination with cisplatin versus cisplatin alone in patients with malignant pleural mesothelioma. J. Clin. Oncol. 21, 2636–2644 (2003). Key phase III trial confirming pemetrexed plus cisplatin as standard of care in mesothelioma.
Treasure, T. et al. Extra-pleural pneumonectomy versus no extra-pleural pneumonectomy for patients with malignant pleural mesothelioma: clinical outcomes of the Mesothelioma and Radical Surgery (MARS) randomised feasibility study. Lancet Oncol. 12, 763–772 (2011).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02040272 (2017).
Zalcman, G. et al. Bevacizumab for newly diagnosed pleural mesothelioma in the Mesothelioma Avastin Cisplatin Pemetrexed Study (MAPS): a randomised, controlled, open-label, phase 3 trial. Lancet 387, 1405–1414 (2016). Phase III MAPS trial demonstrating patient benefit with bevacizumab plus chemotherapy in mesothelioma.
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02991482 (2017).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02784171 (2017).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02628067 (2017).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT03063450 (2017).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02716272 (2016).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02899299 (2017).
Bronte, G. et al. The resistance related to targeted therapy in malignant pleural mesothelioma: why has not the target been hit yet? Crit. Rev. Oncol. Hematol. 107, 20–32 (2016).
Cheng, L. et al. Response evaluation in mesothelioma: beyond RECIST. Lung Cancer 90, 433–441 (2015).
Papadatos-Pastos, D. et al. Clinical outcomes and prognostic factors of patients with advanced mesothelioma treated in a phase I clinical trials unit. Eur. J. Cancer 75, 56–62 (2017).
Bueno, R. et al. Comprehensive genomic analysis of malignant pleural mesothelioma identifies recurrent mutations, gene fusions and splicing alterations. Nat. Genet. 48, 407–416 (2016). Important study detailing comprehensive genomic analysis of malignant pleural mesothelioma.
Guo, G. et al. Whole-exome sequencing reveals frequent genetic alterations in BAP1, NF2, CDKN2A, and CUL1 in malignant pleural mesothelioma. Cancer Res. 75, 264–269 (2015).
Lo Iacono, M. et al. Targeted next-generation sequencing of cancer genes in advanced stage malignant pleural mesothelioma: a retrospective study. J. Thorac. Oncol. 10, 492–499 (2015).
Ugurluer, G. et al. Genome-based mutational analysis by next generation sequencing in patients with malignant pleural and peritoneal mesothelioma. Anticancer Res. 36, 2331–2338 (2016).
Christensen, B. C. et al. Integrated profiling reveals a global correlation between epigenetic and genetic alterations in mesothelioma. Cancer Res. 70, 5686–5694 (2010).
Christensen, B. C. et al. Epigenetic profiles distinguish pleural mesothelioma from normal pleura and predict lung asbestos burden and clinical outcome. Cancer Res. 69, 227–234 (2009).
Jamal-Hanjani, M. et al. Tracking genomic cancer evolution for precision medicine: the lung TRACERx study. PLoS Biol. 12, e1001906 (2014).
Cheng, J. Q. et al. p16 alterations and deletion mapping of 9p21-p22 in malignant mesothelioma. Cancer Res. 54, 5547–5551 (1994).
Xio, S. et al. Codeletion of p15 and p16 in primary malignant mesothelioma. Oncogene 11, 511–515 (1995).
Wong, L., Zhou, J., Anderson, D. & Kratzke, R. A. Inactivation of p16INK4a expression in malignant mesothelioma by methylation. Lung Cancer 38, 131–136 (2002).
Christensen, B. C. et al. Asbestos exposure predicts cell cycle control gene promoter methylation in pleural mesothelioma. Carcinogenesis 29, 1555–1559 (2008).
Singhi, A. D. et al. The prognostic significance of BAP1, NF2, and CDKN2A in malignant peritoneal mesothelioma. Mod. Pathol. 29, 14–24 (2016).
Jongsma, J. et al. A conditional mouse model for malignant mesothelioma. Cancer Cell 13, 261–271 (2008). Study detailing the generation of a conditional mouse model to further dissect crucial pathways in mesothelioma development and progression; this model serves as an invaluable tool to test new intervention strategies.
Lopez-Rios, F. et al. Global gene expression profiling of pleural mesotheliomas: overexpression of aurora kinases and P16/CDKN2A deletion as prognostic factors and critical evaluation of microarray-based prognostic prediction. Cancer Res. 66, 2970–2979 (2006).
Dacic, S. et al. Prognostic significance of p16/cdkn2a loss in pleural malignant mesotheliomas. Virchows Arch. 453, 627–635 (2008).
Ivanov, S. V. et al. Genomic events associated with progression of pleural malignant mesothelioma. Int. J. Cancer 124, 589–599 (2009).
Jennings, C. J. et al. Differential p16/INK4A cyclin-dependent kinase inhibitor expression correlates with chemotherapy efficacy in a cohort of 88 malignant pleural mesothelioma patients. Br. J. Cancer 113, 69–75 (2015).
De Rienzo, A. et al. Gender-specific molecular and clinical features underlie malignant pleural mesothelioma. Cancer Res. 76, 319–328 (2016).
Serrano, M. et al. Role of the INK4a locus in tumor suppression and cell mortality. Cell 85, 27–37 (1996).
Pomerantz, J. et al. The Ink4a tumor suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2's inhibition of p53. Cell 92, 713–723 (1998).
Zhang, Y., Xiong, Y. & Yarbrough, W. G. ARF promotes MDM2 degradation and stabilizes p53: ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell 92, 725–734 (1998).
Oliner, J. D. et al. Oncoprotein MDM2 conceals the activation domain of tumour suppressor p53. Nature 362, 857–860 (1993).
Stott, F. J. et al. The alternative product from the human CDKN2A locus, p14ARF, participates in a regulatory feedback loop with p53 and MDM2. EMBO J. 17, 5001–5014 (1998).
Montes de Oca Luna, R., Wagner, D. S. & Lozano, G. Rescue of early embryonic lethality in mdm2-deficient mice by deletion of p53. Nature 378, 203–206 (1995).
Urso, L., Calabrese, F., Favaretto, A., Conte, P. & Pasello, G. Critical review about MDM2 in cancer: possible role in malignant mesothelioma and implications for treatment. Crit. Rev. Oncol. Hematol. 97, 220–230 (2016).
Walter, R. F. et al. MDM2 is an important prognostic and predictive factor for platin–pemetrexed therapy in malignant pleural mesotheliomas and deregulation of P14/ARF (encoded by CDKN2A) seems to contribute to an MDM2-driven inactivation of P53. Br. J. Cancer 112, 883–890 (2015).
Sui, G. et al. Yin Yang 1 is a negative regulator of p53. Cell 117, 859–872 (2004).
Frizelle, S. P. et al. Inhibition of both mesothelioma cell growth and Cdk4 activity following treatment with a TATp16INK4a peptide. Anticancer Res. 28, 1–7 (2008).
Frizelle, S. P. et al. Re-expression of p16INK4a in mesothelioma cells results in cell cycle arrest, cell death, tumor suppression and tumor regression. Oncogene 16, 3087–3095 (1998).
Eilers, G. et al. CDKN2A/p16 loss implicates CDK4 as a therapeutic target in imatinib-resistant dermatofibrosarcoma protuberans. Mol. Cancer Ther. 14, 1346–1353 (2015).
Huang, S. et al. CDK4/6 inhibitor suppresses gastric cancer with CDKN2A mutation. Int. J. Clin. Exp. Med. 8, 11692–11700 (2015).
Konecny, G. E. et al. Expression of p16 and retinoblastoma determines response to CDK4/6 inhibition in ovarian cancer. Clin. Cancer Res. 17, 1591–1602 (2011).
Wiedemeyer, W. R. et al. Pattern of retinoblastoma pathway inactivation dictates response to CDK4/6 inhibition in GBM. Proc. Natl Acad. Sci. USA 107, 11501–11506 (2010).
Kadariya, Y. et al. Bap1 is a bona fide tumor suppressor: genetic evidence from mouse models carrying heterozygous germline Bap1 mutations. Cancer Res. 76, 2836–2844 (2016).
Dey, A. et al. Loss of the tumor suppressor BAP1 causes myeloid transformation. Science 337, 1541–1546 (2012).
Testa, J. R. et al. Germline BAP1 mutations predispose to malignant mesothelioma. Nat. Genet. 43, 1022–1025 (2011). Important study describing germline BAP1 mutations in mesothelioma.
Bott, M. et al. The nuclear deubiquitinase BAP1 is commonly inactivated by somatic mutations and 3p21.1 losses in malignant pleural mesothelioma. Nat. Genet. 43, 668–672 (2011). Important study describing somatic inactivating BAP1 mutations in mesothelioma.
Yoshikawa, Y. et al. High-density array-CGH with targeted NGS unmask multiple noncontiguous minute deletions on chromosome 3p21 in mesothelioma. Proc. Natl Acad. Sci. USA 113, 13432–13437 (2016).
Jensen, D. E. et al. BAP1: a novel ubiquitin hydrolase which binds to the BRCA1 RING finger and enhances BRCA1-mediated cell growth suppression. Oncogene 16, 1097–1112 (1998).
Yu, H. et al. Tumor suppressor and deubiquitinase BAP1 promotes DNA double-strand break repair. Proc. Natl Acad. Sci. USA 111, 285–290 (2014).
Ismail, I. H. et al. Germline mutations in BAP1 impair its function in DNA double-strand break repair. CancerRes. 74, 4282–4294 (2014).
Hakiri, S. et al. Functional differences between wild-type and mutant-type BRCA1-associated protein 1 tumor suppressor against malignant mesothelioma cells. Cancer Sci. 106, 990–999 (2015).
Kemp, C. D. et al. Polycomb repressor complex-2 is a novel target for mesothelioma therapy. Clin. Cancer Res. 18, 77–90 (2012).
LaFave, L. M. et al. Loss of BAP1 function leads to EZH2-dependent transformation. Nat. Med. 21, 1344–1349 (2015).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02860286 (2017).
Bianchi, A. B. et al. High frequency of inactivating mutations in the neurofibromatosis type 2 gene (NF2) in primary malignant mesotheliomas. Proc. Natl Acad. Sci. USA 92, 10854–10858 (1995). Interesting study demonstrating high frequency of inactivating mutations in NF2 in mesothelioma.
Sekido, Y. et al. Neurofibromatosis type 2 (NF2) gene is somatically mutated in mesothelioma but not in lung cancer. Cancer Res. 55, 1227–1231 (1995).
Li, W. et al. Merlin/NF2 suppresses tumorigenesis by inhibiting the E3 ubiquitin ligase CRL4DCAF1 in the nucleus. Cell 140, 477–490 (2010).
Mizuno, T. et al. YAP induces malignant mesothelioma cell proliferation by upregulating transcription of cell cycle-promoting genes. Oncogene 31, 5117–5122 (2012).
Yu, F. X., Zhao, B. & Guan, K. L. Hippo pathway in organ size control, tissue homeostasis, and cancer. Cell 163, 811–828 (2015).
Yokoyama, T. et al. YAP1 is involved in mesothelioma development and negatively regulated by Merlin through phosphorylation. Carcinogenesis 29, 2139–2146 (2008).
Miyanaga, A. et al. Hippo pathway gene mutations in malignant mesothelioma: revealed by RNA and targeted exon sequencing. J. Thorac. Oncol. 10, 844–851 (2015).
Murakami, H. et al. LATS2 is a tumor suppressor gene of malignant mesothelioma. Cancer Res. 71, 873–883 (2011).
Cheng, J. Q. et al. Frequent mutations of NF2 and allelic loss from chromosome band 22q12 in malignant mesothelioma: evidence for a two-hit mechanism of NF2 inactivation. Genes Chromosomes Cancer 24, 238–242 (1999).
Altomare, D. A. et al. A mouse model recapitulating molecular features of human mesothelioma. Cancer Res. 65, 8090–8095 (2005).
Fleury-Feith, J. et al. Hemizygosity of Nf2 is associated with increased susceptibility to asbestos-induced peritoneal tumours. Oncogene 22, 3799–3805 (2003).
Poulikakos, P. I. et al. Re-expression of the tumor suppressor NF2/merlin inhibits invasiveness in mesothelioma cells and negatively regulates FAK. Oncogene 25, 5960–5968 (2006).
Shapiro, I. M. et al. Merlin deficiency predicts FAK inhibitor sensitivity: a synthetic lethal relationship. Sci. Transl Med. 6, 237ra68 (2014). Important study showing synthetic lethal relationship between Merlin and FAK.
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01870609 (2017).
Bueno, R. et al. Phase 2 neoadjuvant study of VX-6063, a FAK inhibitor, in subjects with surgically resectable malignant pleural mesothelioma [abstract]. Presented at 13th International Conference of the International Mesothelioma Interest Group 2016 MS10.04 (2016).
Zhou, S. et al. Multipoint targeting of the PI3K/mTOR pathway in mesothelioma. Br. J. Cancer 110, 2479–2488 (2014). Study rationalizing targeting of the PI3K–AKT pathway in mesothelioma.
Dolly, S. O. et al. Phase I study of apitolisib (GDC-0980), dual phosphatidylinositol-3-kinase and mammalian target of rapamycin kinase inhibitor, in patients with advanced solid tumors. Clin. Cancer Res. 22, 2874–2884 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01655225 (2017).
Yap, T. A., Omlin, A. & de Bono, J. S. Development of therapeutic combinations targeting major cancer signaling pathways. J. Clin. Oncol. 31, 1592–1605 (2013).
Sundar, R., Valeri, N., Harrington, K. J. & Yap, T. A. Combining molecularly targeted agents: is more always better? Clin. Cancer Res. 23, 1123–1125 (2017).
Harris, S. J., Brown, J., Lopez, J. & Yap, T. A. Immuno-oncology combinations: raising the tail of the survival curve. Cancer Biol. Med. 13, 171–193 (2016).
Soria, J. C., Massard, C. & Izzedine, H. From theoretical synergy to clinical supra-additive toxicity. J. Clin. Oncol. 27, 1359–1361 (2009).
Pastan, I. & Hassan, R. Discovery of mesothelin and exploiting it as a target for immunotherapy. Cancer Res. 74, 2907–2912 (2014). Important review on the role of mesothelin in mesothelioma.
Hassan, R. et al. Phase 1 study of the antimesothelin immunotoxin SS1P in combination with pemetrexed and cisplatin for front-line therapy of pleural mesothelioma and correlation of tumor response with serum mesothelin, megakaryocyte potentiating factor, and cancer antigen 125. Cancer 120, 3311–3319 (2014).
Hassan, R. et al. Major cancer regressions in mesothelioma after treatment with an anti-mesothelin immunotoxin and immune suppression. Sci. Transl Med. 5, 208ra147 (2013).
Hassan, R. et al. Preclinical evaluation of MORAb-009, a chimeric antibody targeting tumor-associated mesothelin. Cancer Immun. 7, 20 (2007).
Kaneko, O. et al. A binding domain on mesothelin for CA125/MUC16. J. Biol. Chem. 284, 3739–3749 (2009).
Hassan, R. et al. Phase II clinical trial of amatuximab, a chimeric antimesothelin antibody with pemetrexed and cisplatin in advanced unresectable pleural mesothelioma. Clin. Cancer Res. 20, 5927–5936 (2014).
Hassan, R. et al. A randomized, placebo-controlled study of amatuximab in combination with pemetrexed and cisplatin (P/C) as front-line therapy for subjects with malignant pleural mesothelioma (MPM) [abstract]. J. Clin. Oncol. 34 (Suppl.), TPS8577 (2016).
Golfier, S. et al. Anetumab ravtansine: a novel mesothelin-targeting antibody–drug conjugate cures tumors with heterogeneous target expression favored by bystander effect. Mol. Cancer Ther. 13, 1537–1548 (2014).
Blumenschein, G. R. et al. Phase I study of anti-mesothelin antibody drug conjugate anetumab ravtansine (AR) [abstract]. J. Clin. Oncol. 34 (Suppl.), 2509 (2016).
Hassan, R. et al. A pivotal randomized phase II study of anetumab ravtansine or vinorelbine in patients with advanced or metastatic pleural mesothelioma after progression on platinum/pemetrexed-based chemotherapy (NCT02610140) [abstract]. J. Clin. Oncol. 34 (Suppl.), TPS8576 (2016).
US National Library of Medicine. ClinicalTrials.gov http://www.clinicaltrials.gov/ct2/show/NCT02610140 (2017).
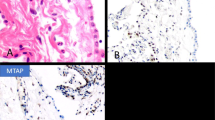
Mavrakis, K. J. et al. Disordered methionine metabolism in MTAP/CDKN2A-deleted cancers leads to dependence on PRMT5. Science 351, 1208–1213 (2016).
Kryukov, G. V. et al. MTAP deletion confers enhanced dependency on the PRMT5 arginine methyltransferase in cancer cells. Science 351, 1214–1218 (2016). References 114 and 115 showed that MTAP deletion leads to PRMT5 dependence.
Schmid, M. et al. Homozygous deletions of methylthioadenosine phosphorylase (MTAP) are more frequent than p16INK4A (CDKN2) homozygous deletions in primary non-small cell lung cancers (NSCLC). Oncogene 17, 2669–2675 (1998).
Illei, P. B., Rusch, V. W., Zakowski, M. F. & Ladanyi, M. Homozygous deletion of CDKN2A and codeletion of the methylthioadenosine phosphorylase gene in the majority of pleural mesotheliomas. Clin. Cancer Res. 9, 2108–2113 (2003).
Kindler, H. L., Burris, H. A. III, Sandler, A. B. & Oliff, I. A. A phase II multicenter study of L-alanosine, a potent inhibitor of adenine biosynthesis, in patients with MTAP-deficient cancer. Invest. New Drugs 27, 75–81 (2009).
Delage, B. et al. Arginine deprivation and argininosuccinate synthetase expression in the treatment of cancer. Int. J. Cancer 126, 2762–2772 (2010).
Szlosarek, P. W. et al. In vivo loss of expression of argininosuccinate synthetase in malignant pleural mesothelioma is a biomarker for susceptibility to arginine depletion. Clin. Cancer Res. 12, 7126–7131 (2006).
Szlosarek, P. W. et al. Arginine deprivation with pegylated arginine deiminase in patients with argininosuccinate synthetase 1-deficient malignant pleural mesothelioma: a randomized clinical trial. JAMA Oncol. 3, 58–66 (2017). Proof-of-concept study demonstrating the role of arginine deprivation in ASS1 -deficient mesothelioma.
Beddowes, E. et al. Phase 1 dose-escalation study of pegylated arginine deiminase, cisplatin, and pemetrexed in patients with argininosuccinate synthetase 1-deficient thoracic cancers. J. Clin. Oncol. 35, 1778–1785 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02709512 (2017).
Mossman, B. T. et al. New insights into understanding the mechanisms, pathogenesis, and management of malignant mesotheliomas. Am. J. Pathol. 182, 1065–1077 (2013).
Bograd, A. J. et al. Immune responses and immunotherapeutic interventions in malignant pleural mesothelioma. Cancer Immunol. Immunother. 60, 1509–1527 (2011).
Cornelissen, R. et al. Ratio of intratumoral macrophage phenotypes is a prognostic factor in epithelioid malignant pleural mesothelioma. PLoS ONE 9, e106742 (2014).
Ujiie, H. et al. The tumoral and stromal immune microenvironment in malignant pleural mesothelioma: a comprehensive analysis reveals prognostic immune markers. Oncoimmunology 4, e1009285 (2015).
Coussens, L. M., Zitvogel, L. & Palucka, A. K. Neutralizing tumor-promoting chronic inflammation: a magic bullet? Science 339, 286–291 (2013).
Lievense, L. A. et al. Precision immunotherapy; dynamics in the cellular profile of pleural effusions in malignant mesothelioma patients. Lung Cancer 107, 36–40 (2017).
Dostert, C. et al. Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 320, 674–677 (2008).
Sekido, Y. Molecular pathogenesis of malignant mesothelioma. Carcinogenesis 34, 1413–1419 (2013).
Balkwill, F., Charles, K. A. & Mantovani, A. Smoldering and polarized inflammation in the initiation and promotion of malignant disease. Cancer Cell 7, 211–217 (2005).
Lo, A. et al. Tumor-promoting desmoplasia is disrupted by depleting FAP-expressing stromal cells. Cancer Res. 75, 2800–2810 (2015).
Wang, L. C. et al. Targeting fibroblast activation protein in tumor stroma with chimeric antigen receptor T cells can inhibit tumor growth and augment host immunity without severe toxicity. Cancer Immunol. Res. 2, 154–166 (2014).
Meerang, M. et al. Antagonizing the Hedgehog pathway with vismodegib impairs malignant pleural mesothelioma growth in vivo by affecting stroma. Mol. Cancer Ther. 15, 1095–1105 (2016).
Lievense, L. A., Bezemer, K., Aerts, J. G. & Hegmans, J. P. Tumor-associated macrophages in thoracic malignancies. Lung Cancer 80, 256–262 (2013).
Holzel, M., Bovier, A. & Tuting, T. Plasticity of tumour and immune cells: a source of heterogeneity and a cause for therapy resistance? Nat. Rev. Cancer 13, 365–376 (2013).
Lievense, L. A. et al. Pleural effusion of patients with malignant mesothelioma induces macrophage-mediated T cell suppression. J. Thorac. Oncol. 11, 1755–1764 (2016).
Awad, M. M. et al. Cytotoxic T cells in PD-L1-positive malignant pleural mesotheliomas are counterbalanced by distinct immunosuppressive factors. Cancer Immunol. Res. 4, 1038–1048 (2016).
van der Most, R. G. et al. Tumor eradication after cyclophosphamide depends on concurrent depletion of regulatory T cells: a role for cycling TNFR2-expressing effector-suppressor T cells in limiting effective chemotherapy. Cancer Immunol. Immunother. 58, 1219–1228 (2009).
Veltman, J. D. et al. COX-2 inhibition improves immunotherapy and is associated with decreased numbers of myeloid-derived suppressor cells in mesothelioma. Celecoxib influences MDSC function. BMC Cancer 10, 464 (2010).
Veltman, J. D. et al. Zoledronic acid impairs myeloid differentiation to tumour-associated macrophages in mesothelioma. Br. J. Cancer 103, 629–641 (2010).
Veltman, J. D. et al. Low-dose cyclophosphamide synergizes with dendritic cell-based immunotherapy in antitumor activity. J. Biomed. Biotechnol. 2010, 798467 (2010).
Ellis, L. M. & Hicklin, D. J. VEGF-targeted therapy: mechanisms of anti-tumour activity. Nat. Rev. Cancer 8, 579–591 (2008).
Strizzi, L. et al. Vascular endothelial growth factor is an autocrine growth factor in human malignant mesothelioma. J. Pathol. 193, 468–475 (2001).
Linder, C., Linder, S., Munck-Wikland, E. & Strander, H. Independent expression of serum vascular endothelial growth factor (VEGF) and basic fibroblast growth factor (bFGF) in patients with carcinoma and sarcoma. Anticancer Res. 18, 2063–2068 (1998).
Hilberg, F. et al. BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. 68, 4774–4782 (2008).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01907100 (2017).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02863055 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02555007 (2015).
Kerbel, R. S. & Kamen, B. A. The anti-angiogenic basis of metronomic chemotherapy. Nat. Rev. Cancer 4, 423–436 (2004).
Lievense, L. A., Sterman, D. H., Cornelissen, R. & Aerts, J. G. Checkpoint blockade in lung cancer and mesothelioma. Am. J. Respir. Crit. Care Med. https://doi.org/10.1164/rccm.201608-1755CI (2017).
Combaz-Lair, C. et al. Immune biomarkers PD-1/PD-L1 and TLR3 in malignant pleural mesotheliomas. Hum. Pathol. 52, 9–18 (2016).
Kindler, H. L. et al. Tremelimumab as second- or third-line treatment of unresectable malignant mesothelioma (MM): results from the global, double-blind, placebo-controlled DETERMINE study [abstract]. J. Clin. Oncol. 34 (Suppl.) 8502 (2016).
Calabro, L. et al. Tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma: an open-label, single-arm, phase 2 trial. Lancet Oncol. 14, 1104–1111 (2013). Phase II trial of tremelimumab for patients with chemotherapy-resistant advanced malignant mesothelioma.
Guazzelli, A. et al. Anti-CTLA-4 therapy for malignant mesothelioma. Immunotherapy 9, 273–280 (2017).
Alley, E. W. et al. Clinical safety and activity of pembrolizumab in patients with malignant pleural mesothelioma (KEYNOTE-028): preliminary results from a non-randomised, open-label, phase 1b trial. Lancet Oncol. 18, 623–630 (2017). Phase Ib trial of pembrolizumab in patients with malignant pleural mesothelioma.
Quispel-Janssen, J. et al. A phase II study of ivolumab in malignant pleural mesothelioma (NivoMes): with translational research (TR) biopies [OA13.01]. J. Thorac. Oncol. 12, S292–S293 (2017).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT03063450 (2017).
Hassan, R. et al. Avelumab (MSB0010718C; anti-PD-L1) in patients with advanced unresectable mesothelioma from the JAVELIN solid tumor phase Ib trial: safety, clinical activity, and PD-L1 expression [abstract]. J. Clin. Oncol. 34 (Suppl.), 8503 (2016).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02588131 (2015).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02716272 (2016).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02899299 (2017).
Marcq, E., Pauwels, P., van Meerbeeck, J. P. & Smits, E. L. Targeting immune checkpoints: new opportunity for mesothelioma treatment? Cancer Treat. Rev. 41, 914–924 (2015).
Serrels, A. et al. Nuclear FAK controls chemokine transcription, Tregs, and evasion of anti-tumor immunity. Cell 163, 160–173 (2015).
US National Library of Medicine. ClinicalTrials.gov https://www.clinicaltrials.gov/ct2/show/NCT02758587 (2017).
Aerts, J. G., Lievense, L. A., Hoogsteden, H. C. & Hegmans, J. P. Immunotherapy prospects in the treatment of lung cancer and mesothelioma. Transl Lung Cancer Res. 3, 34–45 (2014).
Dammeijer, F. et al. Efficacy of tumor vaccines and cellular immunotherapies in non-small-cell lung cancer: a systematic review and meta-analysis. J. Clin. Oncol. 34, 3204–3212 (2016).
Cornelissen, R. et al. Extended tumor control after dendritic cell vaccination with low-dose cyclophosphamide as adjuvant treatment in patients with malignant pleural mesothelioma. Am. J. Respir. Crit. Care Med. 193, 1023–1031 (2016).
Hegmans, J. P. et al. Consolidative dendritic cell-based immunotherapy elicits cytotoxicity against malignant mesothelioma. Am. J. Respir. Crit. Care Med. 181, 1383–1390 (2010).
Aerts, J. et al. Autologous dendritic cells loaded with allogeneic tumor cell lysate (Pheralys®) in patients with mesothelioma: final results of a phase I study [abstract OA13.06]. J. Thorac. Oncol. 12 (Suppl.), S295 (2017).
Sterman, D. H. et al. Pilot and feasibility trial evaluating immuno-gene therapy of malignant mesothelioma using intrapleural delivery of adenovirus-IFNalpha combined with chemotherapy. Clin. Cancer Res. 22, 3791–3800 (2016).
Sterman, D. H. et al. A trial of intrapleural adenoviral-mediated Interferon-alpha2b gene transfer for malignant pleural mesothelioma. Am. J. Respir. Crit. Care Med. 184, 1395–1399 (2011).
Morello, A., Sadelain, M. & Adusumilli, P. S. Mesothelin-targeted CARs: driving T cells to solid tumors. Cancer Discov. 6, 133–146 (2016). Interesting article on using CAR T cells in solid tumours, including mesothelioma.
Beatty, G. L. et al. Mesothelin-specific chimeric antigen receptor mRNA-engineered T cells induce anti-tumor activity in solid malignancies. Cancer Immunol. Res. 2, 112–120 (2014).
Antonia, S. J., Vansteenkiste, J. F. & Moon, E. Immunotherapy: beyond anti-PD-1 and anti-PD-L1 therapies. Am. Soc. Clin. Oncol. Educ. Book 35, e450–e458 (2016).
Galateau-Salle, F., Churg, A., Roggli, V., Travis, W. D. & World Health Organization Committee for Tumors of the Pleura. The 2015 World Health Organization Classification of Tumors of the Pleura: advances since the 2004 Classification. J. Thorac. Oncol. 11, 142–154 (2016).
International Agency for Research on Cancer. A Review of Human Carcinogens: Arsenic, Metals, Fibres, and Dusts (WHO Press, 2012).
Bianchi, C. & Bianchi, T. Global mesothelioma epidemic: trend and features. Indian J. Occup. Environ. Med. 18, 82–88 (2014).
Joshi, T. K., Bhuva, U. B. & Katoch, P. Asbestos ban in India: challenges ahead. Ann. NY Acad. Sci. 1076, 292–308 (2006).
World Health Organization. Chrysotile asbestos. WHO http://apps.who.int/iris/bitstream/10665/143649/1/9789241564816_eng.pdf?ua=1 (2014).
Baumann, F., Ambrosi, J. P. & Carbone, M. Asbestos is not just asbestos: an unrecognised health hazard. Lancet Oncol. 14, 576–578 (2013). Important article on asbestos as a health hazard.
Artvinli, M. & Baris, Y. I. Malignant mesotheliomas in a small village in the Anatolian region of Turkey: an epidemiologic study. J. Natl Cancer Inst. 63, 17–22 (1979).
Comba, P., Gianfagna, A. & Paoletti, L. Pleural mesothelioma cases in Biancavilla are related to a new fluoro-edenite fibrous amphibole. Arch. Environ. Health 58, 229–232 (2003).
Ministry of Labour and Factory Inspectorate. Annual Report for 1947 79–80 (HMSO, 1949).
Doll, R. Mortality from lung cancer in asbestos workers. Br. J. Ind. Med. 12, 81–86 (1955).
Selikoff, I. J., Churg, J. & Hammond, E. C. Asbestos exposure and neoplasia. JAMA 188, 22–26 (1964).
Newhouse, M. L. & Thompson, H. Mesothelioma of pleura and peritoneum following exposure to asbestos in the London area. Br. J. Ind. Med. 22, 261–269 (1965).
The Asbestos Regulations 1969, No. 690, UK Statutory Instrument.
United States Clean Air Act, 42 U.S.C. section 7401 et seq. (1970).
United States Toxic Substances Control Act, 15 U.S.C. section 2601 et seq. (1976).
International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks of Chemicals to Man: volume 14, Asbestos (IARC, 1977).
The Asbestos (Prohibitions) Regulations 1985, No. 910, UK Statutory Instrument.
The Control of Asbestos at Work Regulations 1987, No. 2115, UK Statutory Instrument.
International Agency for Research on Cancer. Overall Evaluations of Carcinogenicity: an Updating of IARC Monographs 1–42 106–116 (IARC, 1987).
Dogan, A. U. et al. Genetic predisposition to fiber carcinogenesis causes a mesothelioma epidemic in Turkey. Cancer Res. 66, 5063–5068 (2006).
The Control of Asbestos Regulations 2006, No. 2739, UK Statutory Instrument.
Napolitano, A. et al. Minimal asbestos exposure in germline BAP1 heterozygous mice is associated with deregulated inflammatory response and increased risk of mesothelioma. Oncogene 35, 1996–2002 (2016).
Harvey, K. F., Zhang, X. & Thomas, D. M. The Hippo pathway and human cancer. Nat. Rev. Cancer 13, 246–257 (2013).
Carbone, M. & Yang, H. Molecular pathways: targeting mechanisms of asbestos and erionite carcinogenesis in mesothelioma. Clin. Cancer Res. 18, 598–604 (2012).
Acknowledgements
S.P. acknowledges UK National Health Service funding to the Royal Marsden Hospital/Institute of Cancer Research NIHR-Biomedical Research Centre, London, UK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
T.A.Y. is consultant to AstraZeneca, Roche, Pfizer, Bristol-Myers Squibb (BMS), EMD Serono, Clovis Oncology and Ignyta, Inc., has received research funding from AstraZeneca, Vertex and Clearbridge Biomedics, and has received travel support from AstraZeneca, GlaxoSmithKline, EMD Serono, Merck Sharp and Dohme (MSD) Oncology, Janssen-Cilag, Vertex and BMS. J.G.A. is consultant/speaker for AstraZeneca, Boehringer Ingelheim, Eli Lilly, MSD, BMS, Roche and Amphera, has received travel expenses from Pfizer, Merck, Boehringer Ingelheim, Verastem and Amphera, and has received research funding from Roche. S.P. is consultant to Ariad, AstraZeneca, Boehringer Ingelheim, BMS, Clovis Oncology, MSD, Novartis, Pfizer and Eli Lilly, has received honoraria from Boehringer Ingelheim, Pfizer and Eli Lilly, travel expenses from Boehringer Ingelheim, BMS, MSD and Pfizer, and research funding from Boehringer Ingelheim and Pierre Fabre. D.A.F. is consultant to Aduro, AstraZeneca, Bayer, Boehringer Ingelheim, Clovis, Medimmune, Lab 21, Lilly, Merck, Pierre Fabre, Roche-Genentech, member of speakers bureaux for BMS, Merck and AstraZeneca.
Supplementary information
Supplementary information S1 (table)
Ongoing clinical trials in mesothelioma (PDF 149 kb)
Glossary
- Pleura
-
A serous membrane formed of a single layer of epithelium of mesothelial origin (mesothelial cells) that forms a closed invaginated sac (the pleural cavity), which contains a minimal amount of serous fluid and surrounds the lung. The pleural membrane reflection covering the surface of the lung is termed visceral pleura, and the reflection attached to the internal chest wall is termed the parietal pleura.
- Asbestos
-
The generic commercial designation for a group of naturally occurring mineral silicate fibres.
- Serpentine asbestos
-
Also known as white asbestos, this is an asbestos subclassification that consists of the mineral chrysotile.
- Amphibole asbestos
-
Also known as brown and blue asbestos, this is an asbestos subclassification that consists of five minerals: actinolite and amosite (brown asbestos) and anthophyllite, crocidolite (blue asbestos) and tremolite.
- Window of opportunity studies
-
Trials in which patients receive one or more novel antitumour agents between their cancer diagnosis and standard-of-care therapy (usually surgery). Tumour sampling is undertaken before and after therapy for translational research.
- Ezrin, radixin and moesin (ERM) family
-
The ERM family proteins have structural and regulatory roles in the rearrangement of plasma membrane flexibility and protrusions through their reversible interaction with cortical actin filaments and plasma membrane. These ERM proteins are involved in cytoskeletal organization and offer a platform for the transmission of signals in response to various extracellular stimuli through their ability to crosslink transmembrane receptors with downstream signalling components.
- Switch maintenance clinical trial
-
Treating a patient with a different drug immediately after that patient obtains maximal response to an initial induction therapy
- Antimesothelin immunotoxin SS1P
-
(SS1(dsFv)PE38). A recombinant antimesothelin immunotoxin consisting of a mouse antimesothelin variable antibody fragment (Fv) linked to PE38, a truncated portion of Pseudomonas exotoxin A.
- Antibody–drug conjugate
-
A monoclonal antibody attached to an antitumour agent by a chemical linker. This enables the unique targeting with a monoclonal antibody that has the cancer-killing ability of a cytotoxic drug and sensitive discrimination between normal and tumour tissue.
- Antibody-dependent cell-mediated cytotoxicity
-
A mechanism of cell-mediated immune defence whereby an effector cell of the immune system actively lyses a target cell, whose membrane surface antigens have been bound by specific antibodies.
Rights and permissions
About this article
Cite this article
Yap, T., Aerts, J., Popat, S. et al. Novel insights into mesothelioma biology and implications for therapy. Nat Rev Cancer 17, 475–488 (2017). https://doi.org/10.1038/nrc.2017.42
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrc.2017.42
This article is cited by
-
Mesothelin-targeted MRI for assessing migration, invasion, and prognosis in malignant pleural mesothelioma
Cancer Nanotechnology (2024)
-
The BAP1 nuclear deubiquitinase is involved in the nonhomologous end-joining pathway of double-strand DNA repair through interaction with DNA-PK
Oncogene (2024)
-
Genomic characterization and detection of potential therapeutic targets for peritoneal mesothelioma in current practice
Clinical and Experimental Medicine (2024)
-
Predicting survival for patients with mesothelioma: development of the PLACE prognostic model
BMC Cancer (2023)
-
LAG3 is an independent prognostic biomarker and potential target for immune checkpoint inhibitors in malignant pleural mesothelioma: a retrospective study
BMC Cancer (2023)