Abstract
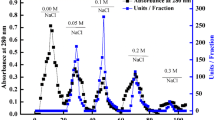
A detailed procedure for the purification of cotinifolin from the latex of Euphorbia cotinifolia using ion exchange chromatography and hydrophobic interaction chromatography is described.Various biochemical characteristics of cotinifolin and other enzymatic properties along with spectroscopic parameters like circular dichorism and fluorescence have been reported. Cotinifolin is stable over a wide range of pH and under extreme conditions of chaotrops and organic solvents where most proteins lost their functions. Further studies on the identification of physiological substrates, biophysical and structural studies will give insight into the utility of the enzyme in the biotechnological industries and protein engineering.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumar, R. Purification and physicochemical studies of metalloprotease, cotinifolin from Euphorbia cotinifolia. Nat Prec (2011). https://doi.org/10.1038/npre.2011.6397.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2011.6397.1