Abstract
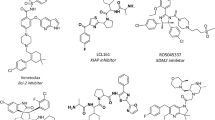
The three-dimensional quantitative structure-activity relationship (3D-QSAR) model of sulfonamide analogs binding a monoclonal antibody (MabSMR) produced against sulfamerazine, was carried out by comparative molecular field analysis (CoMFA). The affinities of MabSMR, expressed as Log10IC50, for 17 sulfonamide analogs were determined by competitive fluorescence polarization immunoassay (FPIA). Removal of two outliers from the initial set of 17 sulfonamide analogs improved the predictability of the models. The 3D-QSAR model of 15 sulfonamides resulted in q2cv values of 0.600, and r2 values of 0.995, respectively. This novel study combining FPIA with CoMFA demonstrates that multidisciplinary research can be used as a useful tool to investigate antigen-antibody interactions and provide information required for design of novel haptens, which may result in new antibodies with properties already optimized by an antibody-based immunoassay.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, Z., Cao, X., Kai, Z. et al. Three Dimensional Quantitative Structure-Activity Relationships of Sulfonamides Binding Monoclonal Antibody by Comparative Molecular Field Analysis. Nat Prec (2008). https://doi.org/10.1038/npre.2008.2646.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2008.2646.1
Keywords
This article is cited by
-
Precise Hapten Design of Sulfonamides by Combining Machine Learning and 3D-QSAR Approaches
Food Analytical Methods (2022)