Abstract
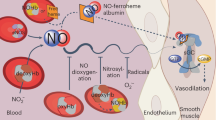
Alkyl nitrates, in vivo, are metabolized to yield nitric oxide, and thiol groups are considered necessary cofactors. This statement is based on studies that underline how these species potentiate hemodynamic responsiveness to nitrates in patients with ischemic heart disease. However, the role of thiols might be mediated by the formation of corresponding S-nitrosothiols, and a redox process is responsible for the nitrates' degradation: an enzyme, probably the cytochrome P450, is involved in vivo. Here, we report evidence that, in vitro, no reaction between thiols and alkyl nitrates takes place, but that stronger reducing agents, such as iron (II) derivatives, are necessary: alkoxy radicals and the nitrite anion are the reaction intermediates. The latter, in slightly acidic conditions, for instance mimicking ischemic conditions, is shown to nitrosilate thiols to the corresponding S-nitrosothiols: the real NO suppliers. Therefore, the direct release of NO from nitrates is excluded. Finally, the in vivo role of thiols on depletion and tolerance is also accounted for.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grossi, L. The nitrite anion: the key intermediate in alkyl nitrates degradative mechanism.. Nat Prec (2007). https://doi.org/10.1038/npre.2007.235.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2007.235.1