Abstract
Methyl-CpG-binding protein 2 (MeCP2) is a ubiquitously expressed transcriptional regulator with functional importance in the central nervous system. Loss-of-function mutations in MECP2 results in the neurodevelopmental disorder, Rett syndrome, whereas increased expression levels are associated with the neurological disorder, MECP2 duplication syndrome. Previous characterization of a mouse line overexpressing Mecp2 demonstrated that this model recapitulated key behavioral features of MECP2 duplication syndrome with specific deficits in synaptic plasticity and neurotransmission. Alterations in excitation/inhibition balance have been suggested to underlie neurodevelopmental disorders with recent data suggesting that picrotoxin (PTX), a GABAA receptor antagonist, rescues certain behavioral and synaptic phenotypes in a mouse model of Down syndrome. We therefore examined whether a similar treatment regimen would impact the behavioral and synaptic phenotypes in a mouse model of MECP2 duplication syndrome. We report that chronic treatment with low doses of PTX ameliorates specific behavioral phenotypes, including motor coordination, episodic memory impairments, and synaptic plasticity deficits. These findings suggest that GABAA receptor antagonists may offer a possible therapeutic target for the treatment of MECP2 duplication syndrome.
Similar content being viewed by others
INTRODUCTION
Methyl-CpG-binding protein 2 (MeCP2) is widely expressed in neuronal and non-neuronal tissues and was originally purified from the brain as a heterochromatin protein that binds to DNA containing a single methyl-CpG dinucleotide (Lewis et al, 1992). Interest in the role of MeCP2 in the brain has been high since the identification of mutations in the MECP2 gene, predicted to result in the loss of function of the gene, are present in >95% of patients with classic Rett syndrome (Amir et al, 1999), an X-linked neurodevelopmental disorder, in which females express hand wringing behavior, autistic behavior, and mental retardation (Hagberg et al, 1983). Rather unexpectedly, duplication of chromosome Xq28, which results in increased expression of MECP2, has been shown to cause a neurodevelopmental disorder named MECP2 duplication syndrome, that predominantly affects males (del Gaudio et al, 2006; Friez et al, 2006; Meins et al, 2005; Ramocki et al, 2009; Van Esch et al, 2005). Individuals diagnosed with MECP2 duplication syndrome are often characterized with symptoms of progressive spasticity, difficulty with social interaction, mental retardation, and autistic behavior (Ramocki et al, 2010). Although there has been much research focused on the loss of function of MeCP2 in CNS, there have only been a few studies that have investigated the impact of MeCP2 overexpression (Collins et al, 2004; Jiang et al, 2013; Luikenhuis et al, 2004; Na et al, 2012).
To study the impact of MeCP2 overexpression in behavior, recent work has focused on the generation of animal models. One line of MeCP2-overexpressing mice was created in which Mecp2 was targeted into the Tau locus (Tau-Mecp2), creating a fusion protein with the first exon of Tau resulting in MeCP2 overexpression selectively in neurons (Luikenhuis et al, 2004). The Tau-Mecp2 mice have an ∼twofold increase in MeCP2 expression in the brain (Luikenhuis et al, 2004; Na et al, 2012). We recently characterized the Tau-Mecp2 mice in a wide array of behavioral paradigms and found that they have an elevated anxiety-like phenotype, motor coordination deficits, impaired novel object recognition, and extinction learning, recapitulating key phenotypes observed in MECP2 duplication syndrome patients. We also observed significant attenuations in long-term potentiation (LTP), a form of neural plasticity that may underlie memory storage (Abel and Lattal, 2001) and alterations in neurotransmission (Na et al, 2012). Indeed, recent data have suggested that loss or gain of MeCP2 expression alters specific aspects of synaptic plasticity and neurotransmission that may impact the balance of excitation/inhibition in the brain (Asaka et al, 2006; Calfa et al, 2011; Chao et al, 2007; Dani et al, 2005; Moretti et al, 2006; Nelson et al, 2011; Nelson et al, 2006; Zhang et al, 2008). Imbalances in excitatory and inhibitory neurotransmission have been suggested to be the result of global alterations in neuronal activity or specific antagonism of excitatory or inhibitory neurotransmission (Chubykin et al, 2007; Turrigiano, 2007). MeCP2 overexpression has been shown to impact excitation-evoked neurotransmission in the brain resulting in a decrease in probability of neurotransmitter release (Na et al, 2012).
Although various putative treatments for Rett syndrome and other forms of mental retardation and autism have been examined by animal studies, no data currently exist on possible therapeutic approaches for MECP2 duplication syndrome. Given that MeCP2 overexpression selectively impacts evoked excitatory neurotransmission consistent with a decrease in the probability of evoked glutamate release, leading to an excess of inhibition, we wondered whether rebalancing this alteration in neurotransmission would rescue the behavioral phenotypes in the Tau-Mecp2 mice. In the current study, we tested the impact of the non-competitive GABAA antagonist picrotoxin (PTX) on behavioral and synaptic plasticity deficits observed in Tau-Mecp2 mice. We report that a non-epileptic dose of PTX ameliorates some of the behavioral deficits, including cognitive and motor impairments, and also restores normal synaptic function in the Tau-Mecp2 mice. These data suggest that rebalancing excitation–inhibition using GABAA antagonists may hold promise as a therapeutic treatment for MECP2 duplication disorder.
MATERIALS AND METHODS
Animals
Tau-Mecp2 mice were generously provided by Dr Rudolf Jaenisch and have been previously described (Luikenhuis et al, 2004). We backcrossed this line for more than 10 generations to C57BL/6 male mice. Mouse genotype was confirmed by PCR using tail samples and the following primers, Tau138 (5′-CTG-GCA-GAT-CTT-CCC-GTC-TA-3′), Tau1078 (5′-TGC-CTG-ACA-GAG-TCC-AGA-TG-3′), and Neo1323 (5′-AGG-GGA-TCC-GTC-CTG-TAA-GT-3′), which amplifies a 941-bp fragment from the wild-type (WT) allele and a 796-bp fragment from the overexpressing allele. Mice were maintained on a 12-h light/dark cycle with ad libitum access to food and water. All experiments used male mice aged 10–16 weeks that were Tau-Mecp2 mice or wild-type littermate controls. All experiments were conducted by an observer who was blind to genotype and treatment. For all testing, mice were allowed to habituate in the behavioral room for 1 h. Mice were tested in the following order: locomotor activity, dark–light cycle, elevated plus maze (EPM), rotarod, fear conditioning, and extinction training. Two separate cohorts of mice were used for cue fear conditioning and novel object recognition. For all experiments, data were presented as mean±SEM, with significance set as p<0.05. All procedures were approved by the Institutional Animal Care and Use Committee at the University of Texas Southwestern Medical Center.
Drug Treatment
Mice were weighed at the start of experimentation and assigned to one of four groups: (1) WT vehicle (VEH), (2) Tau-Mecp2 (Tau) VEH, (3) WT PTX, and (4) Tau PTX. Mice were treated chronically for five consecutive days with a daily injection of VEH or PTX (1 mg/kg, i.p.) on the basis of the dose used by Fernandez et al (2007) and Ramwell and Shaw (1965). It has previously been shown that systemic doses of 4–8 mg/kg induce epileptic activity in mice; however, lower doses (eg, 0.4–2 mg/kg) do not induce any seizure activity (Ramwell and Shaw, 1965). PTX (Sigma) was dissolved (10 × ) with dimethyl sulfoxide (Sigma) and diluted with isotonic saline to a 1 × working concentration. Mice used for hippocampal slice electrophysiology were given systemic PTX injections chronically based on the timeline outlined by Fernandez et al (2007), who demonstrated robust changes in LTP after chronic PTX administration.
Locomotor Activity
Mice were placed in a standard cage with fresh bedding. Locomotor activity was assessed for 2 h under red light by photocell beams linked to computer data acquisition software (San Diego Instruments).
Dark–Light Test
To assess anxiety, mice were examined in the dark–light test. The dark–light test consisted of a box that was partitioned into two compartments, a dark and light side. Mice were placed into the dark side for 2 min before a gate that separates the two compartments was lifted, thereby allowing mice to explore the dark or light sides for 10 min. The dependent variable was the time spent in the light side (Med Associates).
EPM
Mice were placed in the center of an EPM, a plus-shaped maze constructed of two open and two closed arms measuring 33 × 5 cm at 40 lux for 5 min. Time spent in the open arm versus time spent in closed arms was quantified by the Ethovision tracking software (Noldus Information Technology).
Rotarod
Each mouse was placed on a rotating rod (IITC Life Science), the speed of which gradually increased over 5 min. The test session ended when the animal fell off the rod or after 5 min. Mice were given four trials across 2 days for a total of eight trials.
Fear Conditioning
Training and testing were conducted under red light as previously described (Na et al, 2012). Briefly, mice were placed in individual fear conditioning chambers (Med Associates) and habituated to the chambers for 2 min. Two tone plus shock presentations were administered with an interstimulus interval of 1 min. White noise (90 dB) was played for 30 s co-terminating with a 1 s footshock (0.5 mA). A minute after the last tone plus shock pairing, mice were returned to their home cages. Chambers were cleaned between each trial. To test context-dependent fear conditioning, mice were placed in the same chamber 24 h later without tone or shock and freezing behavior was recorded for 5 min. Freezing behavior was defined as no movement except for respiration. After 4 h, the cue test was performed. To test cue-dependent fear conditioning, mice were placed in a novel environment scented with vanilla extract and lit with fluorescent lights for 6 min. Freezing behavior was scored every 5 s and assessed during a 3 min baseline period followed by 3 min of tone.
Cue/context Extinction Training
Extinction is considered an active form of learning (Robleto et al, 2004) thus, to test if Tau-Mecp2 mice can learn new contingencies, mice were trained to extinguish conditioned responses to cue and/or context. For cue extinction, mice were exposed to a novel environment (see above), and baseline freezing (3 min) and freezing during presentation of tone (3 min) were assessed. For context extinction training, mice were placed in the same context and freezing behavior was scored for 3 min. Sessions were terminated once WT controls reached baseline levels of freezing during tone and/or context presentation, which occurred after 8–9 days. A cohort of mice was tested for differences in nociception at the conclusion of the fear conditioning experiments. WT and Tau-Mecp2 mice were exposed to increasing footshock intensity (range 0.05–0.45 mA) to determine the footshock threshold at which animals responded by flinching or vocalizing. For all fear conditioning experiments, an observer blind to genotype and drug treatment scored freezing behavior. Percentage of freezing behavior during tone/context presentation was compared between groups.
Novel Object Recognition (NOR)
On day 1, mice were habituated to a rectangular box (88 × 17 cm) at 40 lux for 10 min. After 4 h, mice were reintroduced to the rectangular box containing two of the same objects (A and A), a metal pipe, for 10 min (familiarization phase). The following day, mice were placed in the rectangular box for 10 min and exposed to two objects: object A and a new object, B, a metal cone. An experimenter who was blind to treatment scored the amount of time that the animal spent investigating the objects and a difference score was calculated. A difference score was calculated by subtracting the time spent with ‘A’ from the time spent with ‘B’.
Hippocampal Slice Electrophysiology
Hippocampal slices were prepared as previously described (Akhtar et al, 2012; Kim et al, 2012; Na et al, 2012; Morris et al, 2013). Briefly, following pictrotoxin or saline treatment mice were anesthetized with Euthasol (30 mg/ml, 0.2 ml i.p.) and then decapitated. Brains were removed and quickly immersed in ice-cold artificial cerebral spinal fluid (ACSF) that was continuously bubbled with 95% O2 and 5% CO2, pH 7.4. A 350-μm thick transverse section of the hippocampus was sectioned and placed in ice-cold oxygenated ACSF for at least 1 h at 32 °C. The hippocampal slices were then transferred into a recording chamber and superfused with ACSF at a constant rate of 2.5 ml/min at 30 °C. Glass recording electrodes filled with ACSF (resistance, 1–2 MΩ) were used to record field excitatory post synaptic (fEPSP) potentials. Extracellular stimuli were delivered by placing a bipolar platinum–tungsten stimulating electrode to stimulate fibers of the Schaffer collateral pathway while the recording electrode was inserted into the CA1 just beneath the molecular layer. Input–output relationship was determined by providing an ascending series of stimulus input intensities (40–240 μA) until the maximum fEPSP response was determined. An input stimulus intensity that induced 40–50% of the maximum response was used for measuring paired pulse ratio (PPR) and LTP. An input intensity that induced the maximum response was used for high frequency stimulation-induced LTP. PPR was induced by giving two pulses at decreasing interpulse intervals (500, 400, 200, 100, 50, 30, and 20 ms) and analyzed by dividing the fEPSP slope of pulse 2 by that of pulse 1. Following 20 min of stable baseline fEPSP slope, LTP was induced with two 100 Hz trains of 100 pulses with an intertrain interval of 20 s.
Statistics
Locomotor activity, EPM, DL, and fear conditioning were analyzed using a two-way ANOVA with genotype and drug as between-group factors. LTP and rotarod data were analyzed using two-way repeated measures ANOVA. NOR, PPR, and input–output data were analyzed by one-way ANOVA. Fisher’s LSD was used following significant interaction/main effects. A p-value of <0.05 was required for statistical significance; however, an adjusted p-value of ⩽0.017 was used for Fisher’s LSD analyses that contained multiple comparisons.
RESULTS
PTX Treatment in Tau-Mecp2 Mice Ameliorates Impairments in Episodic Memory
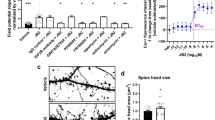
Individuals with MECP2 duplication often have moderate to severe intellectual disability. The Tau-Mecp2 mice display specific deficits in some learning and memory paradigms (Na et al, 2012). To assess whether PTX treatment has any impact on these learning and memory deficits, we first tested the mice in the novel object recognition paradigm. During familiarization, there was no significant difference in the amount of time that the WT or Tau-Mecp2 mice spent with the objects (data not shown). In agreement with previous data, Tau-Mecp2 mice had a significant deficit in performance in the NOR paradigm compared with WT mice (Figure 1a; Na et al, 2012). We found that chronic PTX did not significantly alter the performance of the WT mice compared with vehicle treatment (Figure 1a). However, PTX treatment ameliorated the deficits in NOR in Tau-Mecp2 mice with a substantial increase in the difference score compared with Tau-Mecp2 mice that were treated with a vehicle (Figure 1a).
PTX improved deficits in episodic memory and in motor performance. (a) PTX-treated Tau-Mecp2 mice showed an improved NOR compared with Veh-treated Tau-Mecp2 mice. Significant interaction effect (F(3,44)=3.62, p<0.05). Fisher’s LSD tests revealed significant differences between Tau-Mecp2 PTX and Tau-Mecp2 vehicle-treated mice. Difference score=time with novel object−time with familiar object. (b) Motor coordination was improved by PTX treatment in Tau-Mecp2 mice. Significant main effect of time and drug were seen in WT (F(7,255)=19.49, p<0.001; F(1,255)=4.27, p=0.047), respectively; post-hoc analyses did not reveal significant differences between groups. Significant main effects of drug and time were observed in Tau-Mecp2 mice (F(1,215)=5.65, p=0.03; F(7,215)=10.95, p<0.001, respectively). Post-hoc analyses indicated a significant difference between Tau-Mecp2 vehicle-treated mice and Tau-Mecp2 PTX mice during trial 8 with Tau-Mecp2 PTX spending more time on the rotarod compared with Tau-Mecp2 vehicle-treated mice. PTX, picrotoxin; Tau, Tau-Mecp2; Veh, vehicle; WT, wild type. Data are means±SEM. *p⩽0.017 compared with Tau-Mecp2 vehicle-treated mice.+p<0.05.
PTX Improves Motor Learning Deficits in Tau-Mecp2 Mice
Tau-Mecp2 mice have significant deficits in motor learning as assessed by the latency to fall off the rotarod compared with WT controls (Figure 1b). We found that chronic PTX treatment did not impact the performance of WT mice on the rotarod test (Figure 1b). However, PTX treatment improved the motor impairments of Tau-Mecp2 such that these mice now performed similar to WT mice (Figure 1b).
Chronic PTX Treatment Ameliorated Deficits in LTP and Short-Term Plasticity
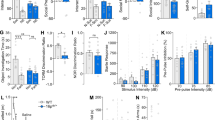
Tau-Mecp2 mice have deficits in hippocampal LTP that correlate with some of the learning and memory deficits (Na et al, 2012). We therefore examined whether PTX rescues the hippocampal LTP deficits in the Tau-Mecp2 mice. We confirmed that Tau-Mecp2 mice have deficits in LTP compared with WT controls (Figure 2a). We found that chronic PTX treatment had no effect on LTP from WT hippocampal slices (Figure 2b and c), however, it rescued decrements in LTP in Tau-Mecp2 hippocampal slices with the effect sustained for the full 2 h of the recording (Figure 2d).
LTP impairments were improved by PTX treatment in Tau-Mecp2 mice. (a) LTP was attenuated in Tau-Mecp2 vehicle-treated mice compared with WT vehicle-treated mice; significant interaction effect (F(68,2069)=4.57, p<0.001). Post-hoc analyses demonstrated significant differences between vehicle-treated groups at almost all time points. (b and c) LTP was not affected by PTX treatment in WT mice nor did it differentially affect LTP between Tau-Mecp2 and WT mice. (d) LTP deficits were rescued by chronic PTX treatment in Tau-Mecp2 mice compared with Tau-Mecp2 vehicle-treated mice. A significant interaction effect was seen (F(68,1931)=2.60, p<0.001). Post-hoc tests revealed that LTP was higher during the last hour of the test in Tau-Mecp2 PTX compared with Tau-Mecp2 vehicle-treated mice. (e) PPR was significantly enhanced at 20, 30, 50, 100, and 200 ms interstimulus intervals in Tau-Mecp2 hippocampal slices compared with WT slices as demonstrated by Fisher’s LSD post-hoc tests. A one-way ANOVA revealed significant differences at 20, 30, 50, 100, and 200 ms time intervals (F(3,64)=5.23, p=0.003; F(3,64)=3.53, p<0.02; F(3,64)=3.66, p<0.02; F(3,64)=4.06, p=0.01; F(3,64)=3.63, p=0.02, respectively). (f and g) PTX had no effect on PPR between WT groups and between WT and Tau-Mecp2 mice. (h) Chronic PTX treatment ameliorated the enhanced PPR at 100 m in Tau-Mecp2 mice as revealed by Fisher’s LSD tests. PTX, picrotoxin; Tau, Tau-Mecp2; Veh vehicle; WT, wild type. Data are means±SEM. *p<0.05. N=28 mice.
We next examined the effect of PTX on baseline transmission and short-term plasticity. We placed stimulating electrodes in the Schaffer collaterals and recorded field potentials in the CA1 region of the hippocampus to generate input–output curves. We found no significant differences as a result of genotype in WT or Tau-Mecp2 hippocampi or PTX treatment on input/output slopes as determined by the ratio of presynaptic volley amplitude to fEPSP slope (data not shown). We also examined PPR by recording the response to two pulses separated by varying interstimulus intervals and taking the ratio of the second response to first. We observed enhanced PPR at 20, 30, 50, 100, and 200 ms interstimulus intervals in Tau-Mecp2 hippocampal slices compared with WT controls (Figure 2e). Chronic PTX treatment did not alter any PPF responses in WT animals (Figure 2f and g), however, it normalized the enhanced PPR seen at the 100-ms interstimulus interval in Tau-Mecp2 mice (Figure 2g and h).
PTX Treatment had no Effect on Anxiety-Like Behavior in Tau-Mecp2 Mice
Tau-Mecp2 mice have a prevalent anxiety-like phenotype, therefore, we tested the effects of PTX treatment to ameliorate or attenuate the anxiety-related behavior in the dark–light and elevated plus maze tests. In the dark–light test, the Tau-Mecp2 mice spent significantly less time in the light side than WT control mice (main effect of genotype), suggesting an increase in anxiety-related behavior. We found that PTX treatment had no effect on the behavior of WT or Tau-Mecp2 mice in this paradigm (Figure 3a). In the elevated plus maze test, Tau-Mecp2 mice spent significantly less time in the open arm compared with WT mice (main effect of genotype), also suggestive of a heightened anxiety-like phenotype. We found that PTX treatment had no effect on the WT or Tau-Mecp2 mice in this paradigm (Figure 3b) consistent with the findings in the dark–light test (Figure 3a).
PTX did not impact the heightened anxiety-like phenotype in Tau-Mecp2 mice. (a and b) PTX did not alter the prevalent anxiety-like phenotype in Tau-Mecp2 mice in dark–light cycle (a) and elevated plus maze (EPM) (b) tests. Significant main effect of genotype (F(1,38)=25.72, p=0.001; F(1, 38)=5.79, p=0.02) for dark–light cycle and EPM, respectively. PTX, picrotoxin; Tau, Tau-Mecp2; Veh, vehicle; WT, wild type. Data are means±SEM. *p<0.05 compared with Tau-Mecp2 vehicle-treated mice.
PTX Treatment in Tau-Mecp2 Mice does not Ameliorate Impairments in Associative Learning
To assess whether PTX treatment has any impact on associative memory, we tested the mice in the fear conditioning paradigm. Baseline freezing between the Tau-Mecp2 and WT mice was indistinguishable (data not shown). After 24 h of training, the Tau-Mecp2 had a significant increase in context-dependent (Figure 4a) and in cue-dependent fear conditioning (Figure 4b). We found that chronic PTX treatment had no effect on the WT animals’ performance, and that it did not rescue or attenuate the context or cue fear conditioning deficits in the Tau-Mecp2 mice (Figure 4a and b). The increase in context and cue-dependent fear conditioning displayed by the Tau-Mecp2 mice indicates an inability to extinguish the conditioned responses (Figure 4c and d; see Na et al, 2012). Chronic PTX treatment did not impact the performance of WT mice in extinction learning, and also did not alter the context and cue-dependent extinction deficits in Tau-Mecp2 mice. The differences in freezing between genotypes are not due to differences in nociception as Tau-Mecp2 mice respond similarly to footshock as WT controls and were not altered by drug treatment (Figure 4e).
PTX does not rescue context- or cue-dependent fear conditioning deficts. (a and b) PTX did not affect freezing responses in Tau-Mecp2 or WT mice. Tau-Mecp2 mice froze significantly more than WT in contextual and cued fear conditioning, 24 h post-training, (significant main effect of genotype F(1,23)=56.89, p<0.001 and significant interaction effect between group and time F(24, 215)=2.93, p<0.001, for contextual and cued, respectively). (c and d) PTX did not improve extinction learning in Tau-Mecp2 mice in cued or contextual fear conditioning. A significant interaction effect indicated that WT mice extinguished freezing responses to cue faster than Tau-Mecp2 mice (F(24,215)=2.93, p<0.001). Significant main effects for context extinction were found for group and time (F(3, 247)=21.94; F(7, 247)=8.1), respectively. (e) There are no significant differences in nociception between WT and Tau-Mecp2 mice in flinching or jumping. There is a main effect of genotype in vocalization between WT and Tau-Mecp2 mice with Tau-Mecp2 mice receiving a higher shock intensity before vocalizing (F(1, 34)=14.94, p<0.01). PTX, picrotoxin; Tau, Tau-Mecp2; Veh, vehicle; WT, wild type. Data are means±SEM. *p<0.05 compared with WT vehicle-treated mice.
Body Weight and Locomotor Activity were not Adversely Affected by PTX Treatment
To ensure that PTX did not have any overt detrimental effects, body weight and locomotor activity were assessed in chronically treated mice. There were no significant differences between initial body weight and body weight at the end of experimentation for any of the treatment groups (Figure 5a). Locomotory activity was similar between Tau-Mecp2 mice and WT littermates. Chronic treatment with PTX had no significant effect on locomotor activity of either WT or Tau-Mecp2 mice over a 2-h testing period (Figure 5b, inset) or when plotted as 5-min epochs over the testing period (Figure 5b). Additionally, we did not observe any seizure activity at the dose of PTX used in either WT or Tau-Mecp2 mice (data not shown), similar to what has been previously reported (Fernandez et al, 2007; McGaugh et al, 1990).
PTX did not impact overt behavior or the heightened anxiety-like phenotype in Tau-Mecp2 mice. Chronic PTX treatment did not affect (a) body weight or (b) locomotor activity in Tau-Mecp2 or WT mice. Inset represents average over 2 h of locomotor activity. PTX, picrotoxin; Tau, Tau-Mecp2; Veh, vehicle; WT, wild type. Data are means±SEM. *p<0.05 compared with Tau-Mecp2 vehicle-treated mice.
DISCUSSION
The present study demonstrates that the GABAA receptor antagonist, PTX, ameliorates key behavioral and synaptic deficits observed in a mouse model of MECP2 duplication syndrome. Specifically, we demonstrate that chronic PTX treatment improves performance in the NOR paradigm, as well as motor coordination deficits, but did not impact the heightened anxiety-like phenotype or the associative learning deficits in Tau-Mecp2 mice. These data suggest that PTX’s effects, while beneficial for episodic memory in our mouse model, do not extend to learning processes that involve aversive elements (ie, aversive footshock). PTX treatment also rescued deficits in LTP with a partial rescue of short-term synaptic plasticity. Chronic PTX did not produce any overt detrimental effects and did not impact locomotory activity or body weight during the course of treatment in either the WT or Tau-Mecp2 mice. These results show that PTX ameliorates particular behavioral and synaptic impairments associated with MeCP2 overexpression and that GABAA receptors may have promise as a therapeutic target for the treatment of MECP2 duplication syndrome. Moreover, these data suggest that certain behavioral and synaptic aspects of MECP2 duplication syndrome stem from different biological alterations induced by MeCP2 overexpression.
Previous work has demonstrated specific behavioral phenotypes in Tau-Mecp2 mice (Na et al, 2012), including deficits in certain forms of learning and memory, thus, we focused on whether PTX could alleviate these impairments. Chronic PTX treatment ameliorated deficits in NOR but did not improve fear conditioning or extinction learning in the Tau-Mecp2 mice. One explanation for these findings is that PTX does not improve learning and memory processes associated with anxiety or aversion. This hypothesis is supported by the ineffectiveness of PTX on anxiety-like behavior in Tau-Mecp2 mice. A second possibility is that PTX only improves deficits in non-associative learning and memory tests. NOR is a test of episodic memory (Bevins and Besheer, 2006), whereas fear conditioning and extinction learning are both associative learning and memory paradigms (Abel and Lattal, 2001). In agreement with this possibility, we observe improved performance in PTX-treated Tau-Mecp2 mice in rotarod, another non-associative learning task. Taken together, these results support the premise that PTX is beneficial for non-associative or perhaps non-aversive forms of learning and memory in mice that overexpress MeCP2. However, we cannot exclude the possibility that the lack of an effect of PTX in the fear conditioning experiments could be related to the low drug dose used in these experiments. Future work will be necessary to further delineate the effect of PTX on other cognitive tasks.
The PTX-mediated behavioral improvements in the NOR test that were observed with the Tau-Mecp2 mice were similar to the PTX-mediated improvements in a mouse model of Down syndrome (Fernandez et al, 2007; Kleschevnikov et al, 2012). In the Down’s syndrome study, the impact of PTX on other forms of learning and memory were not assessed. It is conceivable that the improvement we observed with the Tau-MeCP2 mice in the NOR paradigm may be due to this test being more sensitive to the effects of PTX as outlined above. It is intriguing that animal models of Down syndrome, which is caused by trisomy 21, and MECP2 duplication syndrome, which is caused by duplication of chromosome Xq28, are responsive to PTX for the alleviation of similar learning and synaptic phenotypes. It is possible that the PTX rescue of hippocampal LTP decrements in these studies could account for the enhanced performance in the novel object recognition test. Past studies have shown the importance of the hippocampus in mediating novel object recognition as electrolytic and ibotenic acid lesions of the hippocampus significantly impair performance in this behavioral task (Broadbent et al, 2010). Interestingly, we do not observe an effect of chronic low-dose PTX on LTP in WT mice, a finding that is consistent with previous experiments (Fernandez et al, 2007; Pananceau et al, 1997). Our current findings support the premise that PTX treatment corrects alterations in excitation/inhibition to ameliorate the LTP deficits in the Tau-Mecp2 mice, which under normal conditions, are not affected by PTX treatment.
The PTX-mediated improvement of phenotypes in Tau-Mecp2 mice may arise in part from rebalancing the excitation–inhibition imbalance seen after overexpression of MeCP2 (Asaka et al, 2006; Calfa et al, 2011; Chao et al, 2007; Dani et al, 2005; Moretti et al, 2006; Nelson et al, 2011; Nelson et al, 2006; Zhang et al, 2008). Prolonged directional change in excitation or inhibition at a synapse results in compensatory alterations in baseline synaptic activity as shown by studies on homeostatic plasticity processes (Turrigiano and Nelson, 2000). One possibility is that chronic suppression of inhibitory neurotransmission corrects the excitation–inhibition imbalance seen in MeCP2 gain-of-function models via a process that is similar to homeostatic synaptic plasticity. Another non-mutually exclusive possibility is that increased activity leads to alterations in aberrant gene expression profiles elicited by MeCP2 overexpression, which could in turn counteract the behavioral and cellular deficits.
Collectively, our results demonstrate that PTX rescues specific learning and memory and synaptic impairments in a mouse model of MECP2 duplication syndrome. Our findings provide an important proof of principle that GABAA receptor antagonists may represent a potential treatment approach for patients with MECP2 duplication syndrome.
FUNDING AND DISCLOSURE
LMM reports having received lecture fees from F Hoffmann LaRoche. The remaining authors declare no conflict of interest.
References
Abel T, Lattal KM (2001). Molecular mechanisms of memory acquisition, consolidation and retrieval. Curr Opin Neurobiol 11: 180–187.
Akhtar MW, Kim MS, Adachi M, Morris MJ, Qi X, Richardson JA et al (2012). In vivo analysis of MEF2 transcription factors in synapse regulation and neuronal survival. PLoS One 7: e34863.
Amir RE, Van den Veyver IB, Wan M, Tran CQ, Francke U, Zoghbi HY (1999). Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat Genet 23: 185–188.
Asaka Y, Jugloff DG, Zhang L, Eubanks JH, Fitzsimonds RM (2006). Hippocampal synaptic plasticity is impaired in the Mecp2-null mouse model of Rett syndrome. Neurobiol Dis 21: 217–227.
Bevins RA, Besheer J (2006). Object recognition in rats and mice: a one-trial non-matching-to-sample learning task to study 'recognition memory'. Nat Protoc 1: 1306–1311.
Broadbent NJ, Gaskin S, Squire LR, Clark RE (2010). Object recognition memory and the rodent hippocampus. Learn Mem 17: 5–11.
Calfa G, Hablitz JJ, Pozzo-Miller L (2011). Network hyperexcitability in hippocampal slices from Mecp2 mutant mice revealed by voltage-sensitive dye imaging. J Neurophysiol 105: 1768–1784.
Chao HT, Zoghbi HY, Rosenmund C (2007). MeCP2 controls excitatory synaptic strength by regulating glutamatergic synapse number. Neuron 56: 58–65.
Chubykin AA, Atasoy D, Etherton MR, Brose N, Kavalali ET, Gibson JR et al (2007). Activity-dependent validation of excitatory versus inhibitory synapses by neuroligin-1 versus neuroligin-2. Neuron 54: 919–931.
Collins AL, Levenson JM, Vilaythong AP, Richman R, Armstrong DL, Noebels JL et al (2004). Mild overexpression of MeCP2 causes a progressive neurological disorder in mice. Hum Mol Genet 13: 2679–2689.
Dani VS, Chang Q, Maffei A, Turrigiano GG, Jaenisch R, Nelson SB (2005). Reduced cortical activity due to a shift in the balance between excitation and inhibition in a mouse model of Rett syndrome. Proc Natl Acad Sci USA 102: 12560–12565.
del Gaudio D, Fang P, Scaglia F, Ward PA, Craigen WJ, Glaze DG et al (2006). Increased MECP2 gene copy number as the result of genomic duplication in neurodevelopmentally delayed males. Genet Med 8: 784–792.
Fernandez F, Morishita W, Zuniga E, Nguyen J, Blank M, Malenka RC et al (2007). Pharmacotherapy for cognitive impairment in a mouse model of Down syndrome. Nat Neurosci 10: 411–413.
Friez MJ, Jones JR, Clarkson K, Lubs H, Abuelo D, Bier JA et al (2006). Recurrent infections, hypotonia, and mental retardation caused by duplication of MECP2 and adjacent region in Xq28. Pediatrics 118: e1687–e1695.
Hagberg B, Aicardi J, Dias K, Ramos O (1983). A progressive syndrome of autism, dementia, ataxia, and loss of purposeful hand use in girls: Rett's syndrome: report of 35 cases. Ann Neurol 14: 471–479.
Jiang M, Ash RT, Baker SA, Suter B, Ferguson A, Park J et al (2013). Dendritic arborization and spine dynamics are abnormal in the mouse model of MECP2 duplication syndrome. J Neurosci 33: 19518–19533.
Kim MS, Akhtar MW, Adachi M, Mahgoub M, Bassel-Duby R, Kavalali ET et al (2012). An essential role for histone deacetylase 4 in synaptic plasticity and memory formation. J Neurosci 32: 10879–10886.
Kleschevnikov AM, Belichenko PV, Faizi M, Jacobs LF, Htun K, Shamloo M et al (2012). Deficits in cognition and synaptic plasticity in a mouse model of Down’s syndrome ameliorated by GABAB receptor antagonists. J Neurosci 32: 9217–9227.
Lewis JD, Meehan RR, Henzel WJ, Maurer-Fogy I, Jeppesen P, Klein F et al (1992). Purification, sequence, and cellular localization of a novel chromosomal protein that binds to methylated DNA. Cell 69: 905–914.
Luikenhuis S, Giacometti E, Beard CF, Jaenisch R (2004). Expression of MeCP2 in postmitotic neurons rescues Rett syndrome in mice. Proc Natl Acad Sci USA 101: 6033–6038.
McGaugh JL, Castellano C, Brioni J (1990). Picrotoxin enhances latent extinction of conditioned fear. Behav Neurosci 104: 264–267.
Meins M, Lehmann J, Gerresheim F, Herchenbach J, Hagedorn M, Hameister K et al (2005). Submicroscopic duplication in Xq28 causes increased expression of the MECP2 gene in a boy with severe mental retardation and features of Rett syndrome. J Med Genet 42: e12.
Moretti P, Levenson JM, Battaglia F, Atkinson R, Teague R, Antalffy B et al (2006). Learning and memory and synaptic plasticity are impaired in a mouse model of Rett syndrome. J Neurosci 26: 319–327.
Morris MJ, Mahgoub M, Na ES, Pranav H, Monteggia LM (2013). Loss of histone deacetylase 2 improves working memory and accelerates extinction learning. J Neurosi 33: 6401–6411.
Na ES, Nelson ED, Adachi M, Autry AE, Mahgoub MA, Kavalali ET et al (2012). A mouse model for MeCP2 duplication syndrome: MeCP2 overexpression impairs learning and memory and synaptic transmission. J Neurosci 32: 3109–3117.
Nelson ED, Bal M, Kavalali ET, Monteggia LM (2011). Selective impact of MeCP2 and associated histone deacetylases on the dynamics of evoked excitatory neurotransmission. J Neurophysiol 106: 193–201.
Nelson ED, Kavalali ET, Monteggia LM (2006). MeCP2-dependent transcriptional repression regulates excitatory neurotransmission. Curr Biol 16: 710–716.
Pananceau M, Chen HX, Gustafsson B (1997). Long-term potentiation induced by single volley activation: a mechanism for bicuculline-induced enhancement of synaptic field potentials in the CA1 hippocampal region. Neuroscience 79: 95–101.
Ramocki MB, Peters SU, Tavyev YJ, Zhang F, Carvalho CM, Schaaf CP et al (2009). Autism and other neuropsychiatric symptoms are prevalent in individuals with MeCP2 duplication syndrome. Ann Neurol 66: 771–782.
Ramocki MB, Tavyev YJ, Peters SU (2010). The MECP2 duplication syndrome. Am J Med Genet A 152A: 1079–1088.
Ramwell PW, Shaw JE (1965). The effect of picrotoxin on motor activity and the electroencephalogram of mice. Br J Pharmacol Chemother 24: 651–658.
Robleto K, Poulos AM, Thompson RF (2004). Brain mechanisms of extinction of the classically conditioned eyeblink response. Learn Mem 11: 517–524.
Turrigiano G (2007). Homeostatic signaling: the positive side of negative feedback. Curr Opin Neurobiol 17: 318–324.
Turrigiano GG, Nelson SB (2000). Hebb and homeostasis in neuronal plasticity. Curr Opin Neurobiol 10: 358–364.
Van Esch H, Bauters M, Ignatius J, Jansen M, Raynaud M, Hollanders K et al (2005). Duplication of the MECP2 region is a frequent cause of severe mental retardation and progressive neurological symptoms in males. Am J Hum Genet 77: 442–453.
Zhang L, He J, Jugloff DG, Eubanks JH (2008). The MeCP2-null mouse hippocampus displays altered basal inhibitory rhythms and is prone to hyperexcitability. Hippocampus 18: 294–309.
Acknowledgements
This work was supported by National Institute of Health grant MH081060 (LMM) and a NARSAD Independent Investigator Award (ESN). We thank Dr Rudolf Jaenisch for generously providing the Tau-Mecp2 mice. We would also like to thank Elizabeth Gordon for assistance with breeding of the Tau-Mecp2 line. We would like to acknowledge Drs Ege Kavalali and Megumi Adachi for discussions and comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Na, E., Morris, M., Nelson, E. et al. GABAA Receptor Antagonism Ameliorates Behavioral and Synaptic Impairments Associated with MeCP2 Overexpression. Neuropsychopharmacol 39, 1946–1954 (2014). https://doi.org/10.1038/npp.2014.43
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2014.43
This article is cited by
-
Ketamine and rapid antidepressant action: new treatments and novel synaptic signaling mechanisms
Neuropsychopharmacology (2024)
-
Neonatal inflammation increases hippocampal KCC2 expression through methylation-mediated TGF-β1 downregulation leading to impaired hippocampal cognitive function and synaptic plasticity in adult mice
Journal of Neuroinflammation (2023)
-
Hippocampal Transcriptome Profile of Persistent Memory Rescue in a Mouse Model of THRA1 Mutation-Mediated Resistance to Thyroid Hormone
Scientific Reports (2016)
-
Imbalance of excitatory/inhibitory synaptic protein expression in iPSC-derived neurons from FOXG1+/− patients and in foxg1+/− mice
European Journal of Human Genetics (2016)
-
Dynamic methylation driven by neuronal activity in hippocampal neurons impacts complex behavior
Frontiers in Biology (2015)