Abstract
Regular marijuana use during adolescence, but not adulthood, may permanently impair cognition and increase the risk for psychiatric diseases, such as schizophrenia. Cortical oscillations are integral for cognitive processes and are abnormal in patients with schizophrenia. We test the hypothesis that adolescence is a sensitive period because of the active development of cortical oscillations and neuromodulatory systems that underlie them. The endocannabinoid system upon which marijuana acts is one such system. Here we test the prediction that adolescent cannabinoid exposure alters cortical oscillations in adults. Using in vitro local field potential, in vivo electrocorticogram recordings and cognitive behavioral testing in adult mice, we demonstrate that chronic adolescent, but not adult, cannabinoid exposure suppresses pharmacologically evoked cortical oscillations and impairs working memory performance in adults. The later-maturing prefrontal cortex is more sensitive to adolescent exposure than the earlier-maturing, primary somatosensory cortex. These data establish a link between chronic adolescent cannabinoid exposure and alterations in adult cortical network activity that underlie cognitive processes.
Similar content being viewed by others
INTRODUCTION
Marijuana is the most commonly used illicit drug among adolescents and young adults in the United States (Substance Abuse and Mental Health Services Administration (SAMHSA), 2011). Persistent marijuana use before adulthood may permanently impair cognitive functioning (Solowij et al, 2002; Meier et al, 2012) and confer a higher risk of developing psychiatric diseases, such as schizophrenia, in susceptible individuals (Arseneault et al, 2004). Chronic adolescent, but not adult, cannabinoid exposure produces lasting working memory impairments and recapitulates other schizophrenia endophenotypes in rodents, including impaired sensorimotor gating, social avoidance, and anhedonia/avolition (Schneider and Koch, 2003; O’Shea et al, 2004; Quinn et al, 2008).
Adolescents are especially sensitive to repeated marijuana use, presumably because marijuana interferes with ongoing anatomical and physiological maturation of the brain, particularly the cerebral cortex (Andersen, 2003). The cortex matures along a caudal-to-rostral gradient, with primary sensory cortical areas maturing earlier, and prefrontal regions developing into early adulthood (Gogtay et al, 2004). The cortical endocannabinoid (eCB) system, in which marijuana acts, develops along the same gradient, with changes in cannabinoid-1 receptor (CB1R) density and distribution (Heng et al, 2011) and eCB metabolism (Long et al, 2012).
Coincident with eCB development is the maturation of cortical oscillations (Uhlhaas et al, 2009). Oscillations reflect the synchronous activity of neural networks (Buzsaki and Draguhn, 2004) and are integral to sensory processing, working memory, and nearly all cognitive functions, as they may provide a mechanism by which the brain ‘binds’ the activity of distributed neural ensembles into a coherent representation of cognitive or sensory content (Singer and Gray, 1995; Fries, 2005; Buzsaki, 2006). Cortical oscillations are abnormal in diseases in which these functions are impaired, such as schizophrenia (Gonzalez-Burgos and Lewis, 2008; Uhlhaas and Singer, 2010). Furthermore, oscillations underlie efficient cortical network processing (Buzsaki and Draguhn, 2004) and are dependent on the anatomical and physiological processes that mature throughout adolescence (Uhlhaas et al, 2009).
Acute cannabinoid administration attenuates the power of oscillations (Hajós et al., 2000; Robbe et al, 2006; Kucewicz et al, 2011) by acting at CB1Rs to suppress glutamate release from pyramidal neurons (Holderith et al, 2011; Sales-Carbonell et al, 2013). Furthermore, oscillations are suppressed in chronic marijuana users, particularly in those with an earlier age of onset of use (Skosnik et al, 2012). The adolescent maturation of cortical oscillations and the ongoing development of the eCB system that modulates this network activity, suggest that oscillations may be vulnerable to adolescent marijuana use. Here we test the hypothesis that chronic adolescent, but not adult, cannabinoid exposure alters cortical oscillations, particularly in less developed rostral cortical areas, and impairs cognitive behavioral performance in adults.
MATERIALS AND METHODS
Animals
All experiments were performed according to University of Maryland School of Medicine Institutional Animal Use and Care Committee protocols. Data were collected and analyzed by experimenters ‘blind’ to animals’ treatment condition. Male CD-1 mice (Harlan Laboratories, Frederick, MD) were obtained at P30 (adolescent treatment) or P65 (adult treatment) and injected once daily i.p. from P35 to P55 or P70 to P90. Mice were group housed with cage mates exposed to both vehicle and WIN/Δ-9 tetrahydrocannabinol (THC). After the last injection, adolescent-treated mice were left undisturbed until adulthood (Figure 1a); adult-treated mice were left undisturbed for at least 10 days after the final injection to allow WIN to wash out of tissue. For electrocorticogram (ECoG) experiments, mice were injected once per day i.p. for 3 days with saline, 10 or 20 mg/kg ketamine; the order of injections was alternated between animals.
Robust oscillations are pharmacologically induced in vitro in adult mouse neocortex in adolescent vehicle but not WIN-treated mice. (a) Experimental time course: comparisons of human and rodent development are modified from (Andersen, 2003). The CB1R agonist WIN55,212-2 (WIN; 0.25 or 1 mg/kg) or vehicle were administered to adolescent (P35–P55) or adult mice (P70–P90) once daily for 20 days. LFPs were recorded in brain slices from adult mice (>P100). (b, c) 1-s epoch of in vitro LFP from mPFC slice from an adult mouse administered vehicle (b) or 1 mg/kg WIN (c) during adolescence. LFPs were recorded before (baseline) and during kainic acid (KA; 400 nM) and carbachol (CCh; 20 μM) perfusion (KA+CCh). (d, e) Fourier transform of 10-s LFP recordings in (b) and (c), respectively. KA+CCh (black trace) markedly increases power at all frequencies compared with baseline conditions (grey trace). Frequency ranges: θ=4–7 Hz; α=8–12 Hz; β=13–29 Hz; γ=30–80 Hz. Inset in (e) shows a magnified view of the FFT in (e).
Drugs
WIN55-212,2 (0.25 or 1 mg/kg; Sigma Aldrich, St Louis, MO) and THC (5 mg/kg; National Institute on Drug Abuse Drug Supply Program, Bethesda, MD) were dissolved in 100% ethanol and injected in a 1 : 1 : 18 solution of ethanol:castor oil (Alkamuls EL-620, Rhodia Chemicals): 0.9% saline (1 ml/kg). Control animals were injected vehicle (1 : 1 : 18 ethanol:oil:saline). Ketamine (10 or 20 mg/kg; Bioniche Pharma, IL) was dissolved 1 : 5 in 0.9% saline and injected i.p. (0.1 ml).
In Vitro Slice Preparation and Local Field Potential (LFP) Recordings
Adult mice (>P100) were anesthetized with ketamine (100 mg/kg) and decapitated. Two or three 400 μm coronal sections were cut from each cortical region (mPFC or SCx) from one hemisphere using a microtome (Integraslice 7550 MM, Campden Instruments, IN). During cutting, tissue was immersed in ice-cold ACSF containing (in mM): 26 NaHCO3, 5 BES, 15 glucose, 200 sucrose, 3 KCl, 1.5 MgSO4, and 1 CaCl2. Next, slices were incubated for 30 min at 36 °C and 30 min at 22 °C in ACSF containing (in mM): 120 NaCl, 25 NaHCO3, 5 BES, 15 glucose, 3 KCl, 1.3 MgSO4, and 2 CaCl2 (normal ACSF). Slices were continually saturated with 95% O2–5% CO2. Slices were maintained at 36 °C in an interface-type recoding chamber and were perfused at 0.7 ml/min with ACSF containing (in mM): 120 NaCl, 25 NaHCO3, 5 BES, 15 glucose, 5 KCl, 1.3 MgSO4, 2 CaCl2 (high K+ ACSF), kainic acid (KA; 400 nM) and carbachol (CCh; 20 μM). LFP recordings were made through a glass pipette filled with normal ACSF (resistance∼0.5–1 MΩ) with an ER-1 amplifier (Cygnus Technology), sampled at 5 kHz, filtered between 0.1 Hz and 1 KHz, and stored on a Mac computer using Igor Pro (Version 6.1, Wavemetrics, Portland, OR).
In Vitro Data Analysis
We analyzed in vitro LFP data with custom-written Igor Pro scripts and the Time-Frequency Toolkit (http://www.igorexchange.com/project/TFPlot). Discrete fast Fourier transforms (FFTs) were performed on 10 s of LFP data and oscillation power (area under the curve) was integrated at different frequencies (θ=4–7 Hz; α=8–12 Hz; β=13–29 Hz; γ=30–80 Hz). Frequency bandwidth boundaries were based on Buhl et al, 1998 and Uhlhaas and Singer, 2010. Statistical analyses were performed with STATA (Version 12, StataCorp, College Station, TX). Data were tested for normality and extreme outliers were identified using the fourth-spread outlier detection test and removed (Hoaglin et al, 1986). Kruskal–Wallis (KW) or Mann–Whitney U (MWU) tests were used to determine a significant main effect of treatment (significant P<0.05). Pairwise comparisons were performed with MWU tests (significant P<0.05).
In Vivo Surgical Procedures and ECoG Recordings
Adult mice (>P100) were anesthetized with ketamine (100 mg/kg) and xylazine (20 mg/kg). A F20-EET radio-telemetric transmitter (Data Sciences International, Minneapolis, MN) was implanted subcutaneously and its leads implanted over the dura above the frontal cortex (1.7 mm anterior to bregma) and the cerebellum (6.4 mm posterior to bregma). Animals were individually housed and recovered from surgery for 48 h before recording.
Mice were acclimated to the behavior testing room for 1 h before ECoG recordings. ECoGs were recorded with the Dataquest A.R.T. acquisition system (Data Sciences International) with frontal ECoG recordings referenced to the cerebellum. Baseline ECoG (10 min) recordings were followed by an i.p. injection of saline, 10 or 20 mg/kg ketamine, and 40 min of post-injection recordings.
In Vivo Data Analysis
We analyzed in vivo ECoGs with custom-written MATLAB scripts (Version 2012a, Mathworks, MA) and the mtspecgramc routine in the Chronux Toolbox (http://chronux.org; Mitra and Bokil, 2008). Oscillation power in each bandwidth (δ=1–3 Hz; θ=4–7 Hz; α=8–12 Hz; β=13–29 Hz; γ=30–80 Hz) was computed in 10 s bins from spectrograms for each animal, and averaged into 10 m bins. Post-injection power was compared with the baseline period with MWU tests (significant P<0.05) using STATA.
Novel Object Recognition Behavior
Adult mice (>P100) were habituated to a black, plastic behavioral arena (12” × 9” × 11”) for 1 h in low light conditions. On the next day, mice were introduced to two identical objects (two shot glasses, 1.5” wide × 3” tall, or two white glass vases, 3” wide × 4” tall ) for 10 min in the arena, returned to their home-cage for 60 min, and then exposed to one object from the first session (vase or shot glass) and one novel object (shot glass or vase) for 10 min. The objects and the arena were cleaned thoroughly with 70% ethanol between sessions to eliminate olfactory cues. Object identity (shot glass or vase) and the location of novel/familiar objects (left or right) were counterbalanced between animals. Behavior was videotaped during these sessions and the time spent interacting with each object during the first 3 min of the test session was manually scored offline by an experimenter blind to animals’ treatment condition and to the novel/familiar nature of the objects. Object interaction was said to occur when the animal’s snout was oriented within 2 cm of the object. Object interaction data were tested for normality and analyzed with two-tailed Student’s t-tests (significant P<0.05).
RESULTS
Adolescent, but Not Adult, WIN Administration Suppresses In Vitro Cortical Oscillations Preferentially in Rostral Cortical Areas
In adolescent vehicle or WIN-treated mice (0.25 or 1 mg/kg), we assessed the ability of the adult neocortex to produce synchronous activity by pharmacologically evoking oscillations. The method we used—KA (400 nM) and CCh (20 μM)—reliably produces β and γ oscillations in cortical slices (Buhl et al, 1998; Oke et al, 2010) that strongly resemble network synchrony in the intact neocortex (Steriade et al, 1996). KA and CCh promote robust LFP oscillations in vitro by enhancing excitatory drive (Buhl et al, 1998; Cunningham et al, 2003) and activating cholinergic receptors on GABAergic interneurons (Fisahn et al, 1998; Gulyás et al., 2010).
Coronal brain slices containing medial prefrontal cortex (mPFC) or primary somatosensory cortex (SCx) were prepared from adult mice administered WIN or vehicle during adolescence (Figure 1a). An example LFP recorded in vitro from adult mPFC of an adolescent vehicle-treated mouse before and during KA+CCh perfusion (Figure 1b) shows robust β and γ oscillations and increased power in θ and α bandwidths (θ=4–7 Hz; α=8–12 Hz; β=13–29 Hz; γ=30–80 Hz) evoked by KA+CCh. This is quantified in the accompanying FFTs of the LFPs (Figure 1d) and in the spectrogram and power spectral density (PSD) in Figure 2a.
Chronic adolescent WIN administration attenuates pharmacologically evoked oscillations in vitro in mPFC and SCx. (a, b) Spectrogram (left) and power spectral density (right) of a 1-s representative KA+CCh LFP recorded in mPFC of an adult mouse administered vehicle (a) or 1 mg/kg WIN (b) during adolescence. Minimum–maximum scales are the same in a and b. (c–f) Box and whisker plots (box: 25th percentile, median, 75th percentile; whiskers: adjacent value to 25% or 75% values) of power from FFTs of LFPs in mPFC of adult mice with KA+CCh perfusion. Mice were administered WIN (0.25 or 1 mg/kg) or vehicle during adolescence (Figure 1a). KW tests determined significant differences between the three treatment conditions and pairwise comparisons were performed using MWU tests (significant P< 0.05). (g, h) Spectrograms and power spectral densities of 1-s example KA+CCh LFP recorded in SCx of adult mouse administered vehicle (g) or 1 mg/kg WIN (h) during adolescence. Minimum–maximum scales are the same in g and h. (i–l) Box and whisker plots of power from FFTs of LFPs in SCx of adult mice in the presence of KA+CCh. Mice were treated and statistics were performed as described for (c–f).
In mPFC slices from adolescent WIN-treated mice, pharmacologically evoked oscillations were profoundly suppressed. Figure 1c shows an example in vitro LFP from mPFC of an adolescent 1 mg/kg WIN-treated adult before and during KA+CCh perfusion. Note the markedly attenuated power of these oscillations as quantified in the FFT (Figure 1e) and spectrogram and PSD (Figure 2b) compared with oscillation power in an adolescent vehicle-treated mouse (Figures 1b and d and Figure 2a).
In slices of adult mPFC, power in γ, β, and α bandwidths was significantly and dose-dependently attenuated by adolescent WIN exposure (Figures 2c–e, Table 1a). Although it approached statistical significance, there was no significant main effect on θ power (Figure 2f, Table 1a).
We next tested the prediction that the caudally located SCx would be less sensitive to adolescent cannabinoid exposure than the rostral mPFC. In SCx of a vehicle-treated mouse, KA+CCh evoked robust γ oscillations (Figure 2g), in contrast to oscillations recorded in an adolescent 1 mg/kg WIN-administered mouse (Figure 2h). Adolescent WIN administration significantly suppressed γ, β, α, and θ oscillations in adult SCx (Figures 2 i–l, Table 1b). However, SCx was less sensitive than mPFC to adolescent WIN exposure as evidenced by the relatively weak effects of adolescent 0.25 mg/kg WIN and the markedly smaller effects of 1 mg/kg (Table 1b).
Long-term cognitive impairments and elevated risk of psychiatric disorders in regular marijuana users are less pronounced when use is initiated in adulthood, instead of in adolescence (Arsenault et al, 2004; Meier et al, 2012). Our hypothesis predicts that the long-term effects of WIN on cortical oscillations would be restricted to adolescent exposure. Adult mice were administered WIN (1 mg/kg) or vehicle for 20 days (P70–P90; Figure 1a) and oscillations were evoked in vitro with KA+CCh. We found that adult treatment had no significant effect on oscillation power in any frequency examined in mPFC (Table 1a) or SCx (Table 1b). The persistent suppression of pharmacologically evoked oscillations recorded in mPFC and SCx of adult mice after WIN exposure during adolescence, but not adulthood, supports our hypothesis that cortical oscillations are markedly sensitive to repeated adolescent cannabinoid exposure.
Adolescent THC Administration Suppresses In Vitro Cortical Oscillations Preferentially in Rostral Cortical Areas
Although WIN is used experimentally to induce cannabinoid-mediated effects, treating mice with the primary active ingredient in marijuana, THC, allowed us to directly test our hypothesis that repeated adolescent marijuana use persistently disrupts cortical oscillations. We administered THC (5 mg/kg) to adolescent mice and recorded oscillations in vitro as above (Figure 3a).
Chronic adolescent THC administration suppresses oscillations in vitro in mouse mPFC but not SCx. (a) THC (5 mg/kg) or vehicle was administered to adolescent mice (P35–P55) and LFPs were recorded in slices from adult mice (>P100). (b–e) Box and whisker plots of power from FFTs of LFPs in mPFC of adult mice with KA+CCh perfusion. Power from adolescent THC or vehicle-treated mice were compared using MWU tests (significant P<0.05). (f–i) Box and whisker plots of power extracted from FFTs of LFPs in SCx of adult mice in the presence of KA+CCh. Mice were treated and statistics were performed as in (b–e).
In adult mPFC, LFP power in γ, β, α, and θ bandwidths was significantly attenuated by adolescent THC administration (Figures 3b–e, Table 1a). SCx was less sensitive than mPFC to adolescent THC exposure (Figures 3f–i, Table 1b) as evidenced in a lack of significant effect on γ, β, or α oscillations and the mild attenuation of θ power by adolescent THC.
Adolescent WIN Administration Attenuates In Vivo Cortical Oscillations
To test whether adolescent cannabinoid administration impairs cortical oscillations in the intact adult brain, we recorded ECoGs in vivo from freely moving adult mice administered WIN (1 mg/kg) or vehicle during adolescence (Figure 1a). Robust oscillations were pharmacologically evoked with the non-competitive NMDA receptor antagonist ketamine as sub-anesthetic doses evoke robust cortical oscillations in rodents and humans (Plourde et al, 1997; Hakami et al, 2009) that are independent of animals’ behavioral states (Hakami et al, 2009).
Ten-minute baseline ECoG recordings were followed by an i.p. injection of saline, 10 or 20 mg/kg ketamine, and 40 min of post-injection recordings (Figure 4a). Immediately after ketamine injection, all mice displayed characteristic movement abnormalities (tottering, falling over, and turning in circles). In vehicle-treated mice, ketamine (10 or 20 mg/kg) markedly increased γ and α power (Figures 4a and c) but did not increase power at other frequencies (data not shown). Ketamine-evoked γ (Figures 4a and b) and α power (Figures 4c and d) were normalized to power before and after saline injection, which did not produce changes in power at either frequency (data not shown).
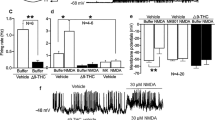
Chronic adolescent administration of WIN attenuates cortical oscillations in vivo. (a) Representative time course of γ power in frontal ECoG before and after injection of 20 mg/kg ketamine. Mice were treated with 1 mg/kg WIN (grey trace) or vehicle (black trace) from P35 to P55 and ECoGs were recorded from adults (>P100). γ Power after ketamine injection was normalized to γ power after saline injection. (b) Box and whisker plots of ECoG γ power in adolescent vehicle (n=5) or WIN-treated (n=7) mice after injection of 10 mg/kg (light grey) or 20 mg/kg (dark grey) ketamine, normalized as above. B=minutes 0–10 of recording; 1=minutes 10–20 of recording; 2=minutes 20–30 of recording; 3=minutes 30–40 of recording; 4=minutes 40–50 of recording. Post-injection power was compared with baseline power with MWU tests (significant P<0.05). (c) Representative time course of α power in frontal ECoG before and after injection of 20 mg/kg in an adolescent vehicle (black trace) or WIN-treated (grey trace) adult mouse. Data are presented as in (a). (d) Box and whisker plots of ECoG α power in adolescent vehicle (n=5) or WIN-treated (n=7) mice after injection of 10 mg/kg (light grey) or 20 mg/kg (dark grey) ketamine, normalized to power before and after saline injection. Time segments are indicated and statistics were performed as in (b).
γ Oscillations
An example time course from a vehicle-treated mouse demonstrates that γ power increased approximately 10 min after 20 mg/kg ketamine and peaked at nearly 800% of saline levels from 20 to 30 min before it returned to baseline (Figure 4a). Group data analysis (Figure 4b) revealed that 10 or 20 mg/kg ketamine significantly increased γ power above baseline from 10 to 30 min after injection in adolescent vehicle-treated mice. In contrast, neither 10 or 20 mg/kg ketamine had a significant effect on γ power in ECoGs of adolescent WIN-treated mice, as shown in the example time course of γ power (Figure 4a) and in group data (Figure 4b).
α Oscillations
Acute administration of 10 or 20 mg/kg ketamine to adolescent vehicle-treated mice also dose-dependently increased α power, as shown in the example time course (Figure 4c) and in group data (Figure 4d). In adolescent WIN-treated mice, ketamine failed to elevate α power above baseline levels (Figures 4c and d). Therefore, while both 10 and 20 mg/kg ketamine significantly increased γ and α power in ECoGs of freely moving adolescent vehicle-treated mice, adolescent WIN treatment prevented this ketamine-induced increase in oscillatory power.
Adolescent WIN Administration Impairs Cognitive Behavioral Performance
To test whether chronic adolescent cannabinoid exposure produces lasting cognitive impairments, we tested adult mice that had been treated in adolescence with WIN (1 mg/kg) or vehicle in the novel object recognition test of working memory. Although vehicle-treated mice spend significantly more time exploring a novel object, WIN-treated animals spend equal time exploring familiar and novel objects ((vehicle (n=7 mice): mean % time with novel object=70.4± 5.1%; mean % time with familiar object=29.6±5.1%; P<0.001, two-tailed t-test) and (WIN (n=8 mice): mean % time with novel object=51.2±4.5%; mean % time with familiar object=48.8±4.54%; P=0.710, two-tailed t-test)). Total object interaction time did not differ significantly between vehicle and WIN-treated mice (vehicle: mean=15.4±3.9 s; WIN: mean=23.5±4.4 s; P=0.198, two-tailed t-test), suggesting that the above results are not biased by differences in locomotion or object engagement during the task.
DISCUSSION
We present evidence that 20-day adolescent, but not adult, cannabinoid exposure attenuates adult cortical oscillations predominantly in cortical areas that are less developed in adolescence. These findings are consistent with reports that regular marijuana users have suppressed evoked γ oscillations with those who initiated use as adolescents showing the greatest subsequent effects (Skosnik et al, 2012). However, as these studies were conducted in adults who still regularly used marijuana, a link between marijuana use specifically during adolescence and suppressed oscillations in adulthood could not be determined. Our findings are also consistent with reports of impaired ketamine-evoked γ synchrony in animal models of psychiatric illnesses (Phillips et al, 2012) that are characterized by abnormal network activity (Uhlhaas and Singer, 2010) and more likely in those that regularly used marijuana as adolescents (Arseneault et al, 2004).
We also report impaired novel object recognition behavior in adolescent WIN-treated mice, which is consistent with previous reports of cognitive impairments after persistent adolescent cannabis exposure in both rodents (Schneider and Koch, 2003; O’Shea et al, 2004; Quinn et al, 2008) and humans (Solowij et al, 2002; Meier et al 2012). Impairments in working memory, processing speed, perceptual reasoning, and other executive functions have been documented in long-term adolescent cannabis users (Solowij et al, 2002; Meier et al 2012). Poor cognitive performance in persistent marijuana users may be related to the cortical oscillation attenuation that we report, as synchronous cortical neural activity plays a role in selective attention (Fries et al, 2001), integration of sensory information (Singer and Gray, 1995), working memory (Roux et al, 2012), and other cognitive functions (Buzsaki, 2006; Wang, 2010). Chronic adolescent, but not adult, cannabinoid-induced oscillation suppression suggests that attenuated network synchrony is not merely a consequence of repeated cannabinoid exposure, but reflects a unique sensitivity of the adolescent brain to modification by cannabinoids. Our data are consistent with findings that adolescent, but not adult, cannabinoid exposure produces lasting cognitive impairments in rodents (Schneider and Koch, 2003; O’Shea et al, 2004; Quinn et al, 2008) and humans (Solowij et al, 2002; Meier et al, 2012).
The heightened sensitivity of the adolescent cortex is probably attributable to anatomical and neurochemical development that occurs during this period (Andersen, 2003). Postnatal maturation of cortical grey and white matter progresses along a caudal-to-rostral gradient (Giedd et al, 1999; Gogtay et al, 2004). As more caudal, sensory cortical areas develop earlier than rostral prefrontal regions (Luna, 2009), prefrontal cortical circuitry is particularly vulnerable to adolescent drug use (Andersen, 2003). Our data confirm this vulnerability, as the magnitude of oscillation suppression seen after adolescent cannabinoid exposure is greater in the rostral mPFC than the caudal SCx.
Cortical glutamatergic and GABAergic networks that synchronize the firing of pyramidal neurons and sculpt the temporal profile of cortical oscillations (Whittington et al, 2000; Cardin et al, 2009) also undergo substantial adolescent maturation (Cao et al, 2000; Hashimoto et al, 2009). Coincident adolescent development of the cortical eCB system includes reduced CB1R expression (Deshmukh et al, 2007; Heng et al, 2011) and changes in eCB metabolism (Long et al, 2012). The highly dynamic nature of glutamatergic, GABAergic, and eCB system development parallels the maturation of cortical oscillations that are suppressed by cannabinoids, and may underlie the attenuation of cortical oscillations that we observe after adolescent cannabinoid exposure.
Acute cannabinoid exposure attenuates the power of neural oscillations recorded in vitro (Hajós et al., 2000) and in vivo, in freely moving animals (Robbe et al, 2006; Hajós et al., 2008; Kucewicz et al, 2011) and during working memory tasks (Kucewicz et al, 2011) by CB1R-mediated suppression of glutamate release from pyramidal neurons (Holderith et al, 2011; Sales-Carbonell et al, 2013). This CB1R-mediated attenuation of excitatory transmission preferentially suppresses the frequency and firing precision of fast-spiking GABAergic interneurons, resulting in smaller and less synchronized field potentials (Holderith et al, 2011). As we have found similar oscillation suppression after chronic exposure to either WIN or THC—two structurally different cannabinoids that both act as CB1R agonists—we suggest that CB1Rs may be responsible for the chronic effects that we observe. However, as WIN and THC have additional targets including CB2Rs (Showalter et al, 1996) and other GPCRs (Breivogel et al, 2001; Ryberg et al, 2007), non-CB1R receptors may underlie oscillation suppression by chronic adolescent WIN or THC administration. Elucidating the receptor mechanism responsible for the effects of adolescent WIN and THC exposure is beyond the scope of this report and related studies are currently underway in our laboratory.
To our knowledge, ours is the first study to demonstrate a direct link between cannabinoid exposure specifically during adolescence and abnormal electrophysiological activity in the adult neocortex, as well as to report a differential vulnerability of cortical regions that parallels their maturational state at the time of drug exposure.
FUNDING AND DISCLOSURE
This work was funded by NIH grant F31DA031547 awarded to SMR. All reagents and materials used for this work are commercially available except THC, which was obtained from the National Institute on Drug Abuse Drug Supply Program in Bethesda, MD. The authors declare no conflict of interest.
References
Andersen SL (2003). Trajectories of brain development: point of vulnerability or window of opportunity? Neurosci Biobehav Rev 27: 3–18.
Arseneault L, Canno M, Whitton J, Murray RM (2004). Causal association between cannabis and psychosis: an examination of the evidence. Br J Psychiatry 325: 1212–1213.
Breivogel CS, Griffin G, Di Marzo V, Martin BR (2001). Evidence for a new G protein-coupled cannabinoid receptor in mouse brain. Mol Pharm 60: 155–163.
Buhl EH, Tamas G, Fisahn A (1998). Cholinergic activation and tonic excitation induce persistent gamma oscillations in mouse somatosensory cortex in vitro. J Physiol 513: 117–126.
Buzsaki G, Draguhn A (2004). Neuronal oscillations in cortical networks. Science 304: 1926–1929.
Buzsaki G (2006) Rhythms of the Brain. Oxford University Press: New York, NY, USA.
Cao Z, Lickey ME, Liu L, Kirk E, Gordon B (2000). Postnatal development of NR1, NR2A and NR2B immunoreactivity in the visual cortex of the rat. Brain Res 859: 26–37.
Cardin JA, Carlén M, Meletis K, Knoblich U, Zhang F, Deisseroth K et al (2009). Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459: 663–667.
Cunningham MO, Davies CH, Buhl EH, Kopell N, Whittington MA (2003). Gamma oscillations induced by kainate receptor activation in the entorhinal cortex in vitro. J Neurosci 23: 9761–9769.
Deshmukh S, Onozuka K, Bender KJ, Bender VA, Lutz B, Mackie K et al (2007). Postnatal development of cannabinoid receptor type 1 expression in rodent somatosensory cortex. Neuroscience 145: 279–287.
Fisahn A, Pike FG, Buh EH, Paulsen O (1998). Cholinergic induction of network oscillations at 40 Hz in the hippocampus in vitro. Nature 394: 186–189.
Fries P (2005). A mechanism for cognitive dynamics: neuronal communication through neuronal coherence. Trends Cogn 9: 474–480.
Fries P, Reynolds JH, Rorie AE, Desimone R (2001). Modulation of oscillatory neuronal synchronization by selective visual attention. Science 291: 1560–1563.
Giedd JN, Blumenthal J, Jeffries NO, Rajapakse JC, Vaituzis AC, Liu U et al (1999). Development of the human corpus callosum during childhood and adolescence: a longitudinal MRI study. Prog Neuropsychopharmacol Biol Psychiatry 23: 571–588.
Gogtay N, Giedd JN, Lusk L, Hayashi KM, Greenstein D, Vaituzis AC et al (2004). Dynamic mapping of human cortical development during childhood through early adulthood. Proc Natl Acad Sci USA 101: 8174–8179.
Gonzalez-Burgos G, Lewis DA (2008). GABA neurons and the mechanisms of network oscillations: implications for understanding cortical dysfunction in schizophrenia. Schizophr Bull 34: 944–961.
Gulyás AI, Szabó GG, Ulbert I, Holderith H, Monyer H, Erdélyi F et al (2010). Parvalbumin-containing fast-spiking basket cells generate the field potential oscillations induced by cholinergic receptor activation in the hippocampus. J Neurosci 30: 15134–15145.
Hajós M, Hoffmann WE, Kocsis B (2008). Activation of cannabinoid-1 receptors disrupts sensory gating and neuronal oscillation: relevance to schizophrenia. Biol Psychiatry 63: 1075–1083.
Hajós N, Katona I, Naiem SS, Mackie K, Ledent C, Mody I et al (2000). Cannabinoids inhibit hippocampal GABAergic transmission and network oscillations. Eur J Neurosci 12: 3239–3249.
Hakami T, Jones NC, Tolmacheva EA, Gaudias J, Chaumont J, Salzberg M et al (2009). NMDA receptor hypofunction leads to generalized and persistent aberrant gamma oscillations independent of hyperlocomotion and the state of consciousness. PLoS One 4: e6755.
Hashimoto T, Nguyen QL, Rotaru D, Keenan T, Arion D, Beneyto M et al (2009). Protracted developmental trajectories of GABAA receptor alpha1 and alpha2 subunit expression in primate prefrontal cortex. Biol Psychiatry 65: 1015–1023.
Heng L, Beverley JA, Steiner H, Tseng KY (2011). Differential developmental trajectories for CB1 cannabinoid receptor expression in limbic/associative and sensorimotor cortical areas. Synapse 65: 278–286.
Hoaglin DC, Iglewicz B, Tukey JW (1986). Performance of some resistant rules for outlier labeling. J Am Stat Assoc 81: 991–999.
Holderith N, Nemeth B, Papp OI, Veres JM, Nagy GA, Hajós N (2011). Cannabinoids attenuate hippocampal gamma oscillations by suppressing excitatory synaptic input onto CA3 pyramidal neurons and fast spiking basket cells. J Physiol 589: 4921–4934.
Kucewicz MT, Tricklebank MD, Bogacz R, Jones MW (2011). Dysfunctional prefrontal cortical network activity and interactions following cannabinoid receptor activation. J Neurosci 31: 15560–15568.
Long LE, Lind J, Webster M, Weickert CS (2012). Developmental trajectory of the endocannabinoid system in human dorsolateral prefrontal cortex. BMC Neurosci 13: 87.
Luna B (2009). Developmental changes in cognitive control through adolescence. Adv Child Dev Behav 37: 233–278.
Meier MH, Caspi A, Ambler A, Harrington H, Houts R, Keefe RSE et al (2012). Persistent cannabis users show neuropsychological decline from childhood to midlife. Proc Natl Acad Sci USA 109: E2657–E2664.
Mitra P, Bokil H (2008) Observed Brain Dynamics. Oxford University Press: New York, NY, USA.
Oke OO, Magony A, Anver H, Ward PD, Jiruska P, Jeffrys JGR et al (2010). High-frequency gamma oscillations coexist with low-frequency gamma oscillations in the rat visual cortex in vitro. Eur J Neurosci 31: 1435–1445.
O’Shea M, Singh ME, McGregor IS, Mallet PE (2004). Chronic cannabinoid exposure produces lasting memory impairment and increased anxiety in adolescent but not adult rats. J Psychopharmacol 18: 502–508.
Phillips KG, Cotel MC, McCarthy AP, Edgar DM, Tricklebank M, O’Neill MJ et al (2012). Differential effects of NMDA antagonists on high frequency and gamma EEG oscillations in a neurodevelopmental model of schizophrenia. Neuropharmacology 62: 1359–1370.
Plourde G, Baribeau J, Bonhomme V (1997). Ketamine increases the amplitude of the 40-Hz auditory steady-state response in humans. Br J Anaesthesia 78: 524–529.
Quinn HR, Matsumoto I, Callaghan PD, Long LE, Arnold JC, Gunasekaran N et al (2008). Adolescent rats find repeated Δ9-THC less aversive than adult rats but display greater residual cognitive deficits and changes in hippocampal protein exposure following exposure. Neuropsychopharmacology 33: 1113–1126.
Robbe D, Montgomery SM, Thome A, Rueda-Orozco PE, McNaughton BL, Buzsaki G (2006). Cannabinoids reveal importance of spike timing coordination in hippocampal function. Nat Neurosci 9: 1526–1533.
Roux F, Wibral M, Mohn HM, Singer W, Uhlhaas PJ (2012). Gamma-band activity in human prefrontal cortex codes for the number of relevant items maintained in working memory. J Neurosci 32: 12411–12420.
Ryberg E, Larsson N, Sjögren S, Hjorth S, Hermannson N-O, Leonova J et al (2007). The orphan receptor GPR55 is a novel cannabinoid receptor. Br J Pharmacol 152: 1092–1101.
Sales-Carbonell C, Rueda-Orozco PE, Soria-Gómez E, Buzsáki G, Marsicano G, Robbe D (2013). Striatal GABAergic and cortical glutamatergic neurons mediate contrasting effects of cannabinoids on cortical network synchrony. Proc Natl Acad Sci USA 110: 719–724.
Schneider M, Koch M (2003). Chronic pubertal, but not adult chronic cannabinoid treatment impairs sensorimotor gating, recognition memory, and the performance in a progressive ratio task in adult rats. Neuropsychopharmacology 28: 1760–1769.
Showalter VM, Compton DR, Martin BR, Abood ME (1996). Evaluation of binding in a transfected cell line expressing a peripheral cannabinoid receptor (CB2): identification of cannabinoid receptor subtype selective ligands. J Pharmacol Exp Ther 278: 989–999.
Singer W, Gray CM (1995). Visual feature integration and the temporal correlation hypothesis. Annu Rev Neurosci 18: 555–586.
Skosnik PD, D’Souza DC, Steinmetz AB, Edwards CR, Vollmer JM, Hetrick WP et al (2012). The effect of chronic cannabinoids on broadband EEG neural oscillations in humans. Neuropsychopharmacology 37: 2184–2193.
Steriade M, Amzica F, Contreras D (1996). Synchronization of fast (30-40 Hz) spontaneous cortical rhythms during brain activation. J Neurosci 16: 392–417.
Solowij N, Stephens RS, Roffman RA, Babor T, Kadden R, Miller M et al (2002). Cognitive functioning of long-term heavy cannabis users seeking treatment. JAMA 287: 1123–1131.
Substance Abuse and Mental Health Services Administration (SAMHSA) (2011). Results from the 2011 National Survey on Drug Use and Health: Summary of National Findings http://www.samhsa.gov/data/nsduh/2k11results/nsduhresults2011.pdf.
Uhlhaas PJ, Roux F, Rodriguez E, Rotarska-Jagiela A, Singer W (2010). Neural synchrony and the development of cortical networks. Trends Cogn Sci 14: 72–80.
Uhlhaas PJ, Roux F, Singer W, Haenschel C, Sireteanu R, Rodriguez E (2009). The development of neural synchrony reflects late maturation and restructuring of functional networks in humans. Proc Natl Acad Sci USA 106: 9866–9871.
Uhlhaas PJ, Singer W (2010). Abnormal neural oscillations and synchrony in schizophrenia. Nat Rev Neurosci 11: 100–113.
Wang XJ (2010). Neurophysiological and computational principles of cortical rhythms in cognition. Physiol Rev 90: 1195–1268.
Whittington MA, Traub RD, Kopell N, Ermentrout B, Buhl EH (2000). Inhibition-based rhythms: experimental and mathematical observations on network dynamics. Int J Psychophysiol 38: 315–336.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Neuropsychopharmacology website
Supplementary information
Rights and permissions
About this article
Cite this article
Raver, S., Haughwout, S. & Keller, A. Adolescent Cannabinoid Exposure Permanently Suppresses Cortical Oscillations in Adult Mice. Neuropsychopharmacol 38, 2338–2347 (2013). https://doi.org/10.1038/npp.2013.164
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2013.164
Keywords
This article is cited by
-
Endocannabinoids at the synapse and beyond: implications for neuropsychiatric disease pathophysiology and treatment
Neuropsychopharmacology (2023)
-
Intravenous self-administration of delta-9-THC in adolescent rats produces long-lasting alterations in behavior and receptor protein expression
Psychopharmacology (2021)
-
The Role of the Endocannabinoid System and Genetic Variation in Adolescent Brain Development
Neuropsychopharmacology (2018)
-
Molecular Basis of Cannabis-Induced Schizophrenia-Relevant Behaviours: Insights from Animal Models
Current Behavioral Neuroscience Reports (2017)
-
NMDAR inhibition-independent antidepressant actions of ketamine metabolites
Nature (2016)