Abstract
Addiction is a chronic relapsing disorder hypothesized to be produced by drug-induced plasticity that renders individuals vulnerable to craving-inducing stimuli such as re-exposure to the drug of abuse. Drug-induced plasticity that may result in the addiction phenotype includes increased excitatory signaling within corticostriatal pathways that correlates with craving in humans and is necessary for reinstatement in rodents. Reduced cystine–glutamate exchange by system xc– appears to contribute to heightened excitatory signaling within the striatum, thereby posing this as a novel target in the treatment of addiction. In the present report, we examined the impact of repeated N-acetyl cysteine, which is commonly used to activate cystine–glutamate exchange, on reinstatement in rodents in a preclinical study and on craving in cocaine-dependent humans in a preliminary, proof-of-concept clinical experiment. Interestingly, repeated administration (7 days) of N-acetyl cysteine (60 mg/kg, IP) produced a significant reduction in cocaine (10 mg/kg, IP)-induced reinstatement, even though rats (N=10–12/group) were tested 24 h after the last administration of N-acetyl cysteine. The reduction in behavior despite the absence of the N-acetyl cysteine indicates that repeated N-acetyl cysteine may have altered drug-induced plasticity that underlies drug-seeking behavior. In parallel, our preliminary clinical data indicate that repeated administration (4 days) of N-acetyl cysteine (1200–2400 mg/day) to cocaine-dependent human subjects (N=4 per group) produced a significant reduction in craving following an experimenter-delivered IV injection of cocaine (20 mg/70 kg/60 s). Collectively, these data demonstrate that N-acetyl cysteine diminishes the motivational qualities of a cocaine challenge injection possibly by altering pathogenic drug-induced plasticity.
Similar content being viewed by others
INTRODUCTION
The treatment of addiction to psychomotor stimulants is hindered by the lack of an FDA-approved pharmacotherapy. Obstacles to drug development include the limited number of therapeutic targets, the identification of which will likely require a more complete understanding of the neural basis of drug addiction. In particular, it is critical to understand how drug-induced plasticity gives rise to persistent drug craving when an individual is exposed to relapse-inducing stimuli, such as acute exposure to the abused drug.
Abnormal glutamate signaling within corticostriatal pathways represents an example of drug-induced neuroplasticity that may be pathological for compulsive drug craving and use, as it has been linked to craving in humans (Volkow et al, 2005) and reinstatement of extinguished drug seeking in rodents (Cornish and Kalivas, 2000; McFarland et al, 2003; Li et al, 2010; Xi et al, 2010). Extant data indicate that reduced cystine–glutamate exchange by system xc– may contribute to abnormal glutamate signaling within corticostriatal pathways and that cocaine-induced changes in system xc– are necessary for cocaine-induced reinstatement in rodents (Baker et al, 2003; Madayag et al, 2007; Kau et al, 2008). Thus, system xc– has been proposed as a potential therapeutic target in the treatment of drug addiction.
A second potential obstacle to the development of effective treatments for drug addiction is the predictive validity of existing animal models (Epstein and Preston, 2003; Katz and Higgins, 2003; Epstein et al, 2006; Yahyavi-Firouz-Abadi and See, 2009). A commonly used model is the reinstatement paradigm, whereby the capacity of discrete stimuli to reinstate extinguished operant responding on a lever that had been used to self-administer the abused substance is assessed. For example, N-acetyl cysteine, a cysteine prodrug used to drive system xc– was identified as a potential novel treatment for addiction, as acute administration blocked the capacity of an acute cocaine injection to reinstate extinguished cocaine seeking in rodents (Baker et al, 2003; Madayag et al, 2007; Kau et al, 2008). What is unclear, however, is the impact of repeated N-acetyl cysteine on cocaine-induced reinstatement and cocaine-induced craving in drug-dependent humans. This is important as repeated administration of N-acetyl cysteine results in reduced bioavailability (Pendyala and Creaven, 1995), yet would be the most likely method by which the drug would be delivered clinically. Moreover, the utility of this paradigm as a preclinical screen rests on the assumption that a reduction in the capacity of a discrete stimulus, such as acute exposure to the self-administered drug, to reinstate extinguished drug seeking in rodents reflects a reduced motivation to obtain the abused drug that would be predictive of diminished drug craving in humans. The extent to which this is not the case raises questions regarding the translational utility as well as predictive validity of the reinstatement paradigm as a tool for preclinical screening of potential antiaddiction therapeutics.
The primary goals of this study were to further evaluate the therapeutic potential of subchronic N-acetyl cysteine treatment and, in doing so, examine the predictive validity of the reinstatement paradigm. Specifically, we examined the impact of repeated administration of N-acetyl cysteine on cocaine intake and cocaine-primed reinstatement in rodents and on self-reports of rush, high, and craving by nontreatment-seeking, cocaine-dependent human participants.
MATERIALS AND METHODS
Rat Subjects
Male Sprague Dawley rats (Harlan, Indianapolis, IN) weighing 275–325 g upon arrival were individually housed in a temperature-controlled colony room with a 12-h reversed light/dark cycle. Housing conditions and experimental protocols were approved by the institutional animal care and use committee of the Marquette University, and carried out according to the NIH Guide for the Care and Use of Laboratory Animals (1996).
Rat Surgeries
Rats were implanted with indwelling catheters under ketamine HCl (100 mg/kg, IP; Fort Dodge Animal Health, Fort Dodge, IA) and xylazine (2 mg/kg, IP; Lloyd Laboratories, Shenandoah, IA) anesthesia as previously described (Madayag et al, 2007; Kau et al, 2008). Following surgery, rats were given at least 5 days to recover before testing.
Rat Cocaine Self-Administration Training
Daily cocaine self-administration sessions occurred in operant conditioning chambers (ENV-008CT; MED-Associates, St Albans, VT) equipped with two retractable levers, two stimulus lights, and a water bottle and housed in sound-attenuating cubicles (ENV-016M, MED-Associates) as previously described (Madayag et al, 2007; Kau et al, 2008). In brief, rats underwent drug self-administration training during daily 2-h sessions in which operant responses on the active lever were reinforced with a cocaine infusion (0.5 mg/kg/200 μl IV; NIDA, Bethesda, MD) under an FR1 schedule of reinforcement. Each reinforced response resulted in the illumination of the stimulus light located above the active lever and was followed by a 25-s timeout period. Responding on a second lever designated as inactive and located on the back wall was recorded but had no programmed consequences. Once rats displayed stable rates of drug self-administration, operationally defined <10% variation in daily responding over at least three consecutive sessions, they underwent 12 daily maintenance sessions.
Rat Extinction Training and Reinstatement Testing
Following self-administration, rats remained in their home cages for 7 days before extinction training to ensure an adequate drug-free period before reinstatement, even if rats quickly extinguished. Extinction training involved placing rats into the operant chambers for 2-h/day as described above, except that each active lever press now resulted in a saline infusion. This continued until the mean number of lever presses was ⩽10 responses across at least three out of four sessions, at which point rats were tested for drug-primed reinstatement. Each rat underwent two reinstatement tests separated by 7 days. Each test was identical to the extinction sessions described above except that subjects received a cocaine injection (10 mg/kg, IP) immediately before being placed into the chamber.
Rat N-Acetyl Cysteine Treatment
Rats received daily N-acetyl cysteine (0 or 60 mg/kg, IP; NAC) 60 min before daily maintenance self-administration sessions or during the 7 days separating the two reinstatement test days.
Human Subjects
A total of six right-handed (Oldfield, 1971) participants (4 males) meeting DSM-IV criteria for cocaine dependence, but otherwise healthy, were recruited from the general population via local advertisement. Subjects were excluded if they had an IQ <80, were positive for pregnancy, HIV, hepatitis, coagulopathy, neurological or hepatic illness, sulfa allergy, or concomitant or historical drug dependence other than cocaine and nicotine. Participants were 41.8±7.4 (mean±SD; range 28–49) years of age, had 18.3±4.0 (12–20) years experience smoking crack cocaine with an age of first use of 21.0±4.2 (15–25) years, and were 8–48 h abstinent from cocaine by self-report and positive only for cocaine upon admission. All were exclusively crack cocaine users, four were one-pack/day cigarette smokers, and none met criteria for any other Axis I or II psychiatric disorder. Subjects underwent thorough medical and psychiatric screenings that included full blood and urine toxicology batteries and a 12-lead electrocardiogram (ECG) to exclude cardiac abnormalities. Participants were counseled regarding the dangers of cocaine use by a study clinician and were offered the opportunity for cocaine detoxification and rehabilitation at each study visit; all declined treatment and continued to decline detoxification/rehabilitation offers throughout the study period. After a complete description of the study and viewing a video of General Clinical Research Center (GCRC) study procedures, subjects provided written informed consent to the Medical College of Wisconsin (MCW) IRB-approved protocol.
Participants first underwent a drug ‘run-up’ procedure to become acquainted with the procedure, experienced a 20 mg/70 kg/60 s IV dose of clinician-delivered cocaine in a controlled setting where their individual responses could be closely monitored and where any emergency interventions could be immediately applied. An Advanced Cardiac Life Support (ACLS)-certified clinician, support staff, and emergency equipment were present and available at all experimental sessions. This dose of cocaine has been used safely in other studies and approximates, or is slightly less, than the ‘high’ induced by a single use of crack cocaine reported by participants in our previous studies (Breiter et al, 1997; Li et al, 2000; Kufahl et al, 2005). ECG, heart rate, and pulse oximetry were monitored continuously and an auto-inflatable cuff monitored blood pressure at 2-min intervals. Cocaine delivery was withheld if the heart rate exceeded 120 beats/min, if systolic blood pressure was >160 mm Hg, or if diastolic blood pressure was >100 mm Hg.
Human Drug Treatment
Participants remained inpatient for 1 to 2 separate 4-day/3-night stays, each with one blinded, oral study medication: N-acetyl cysteine (400 or 800 mg TID, total daily dose 1200 or 2400 mg; N=4) or baclofen (20 mg TID, total daily dose 60 mg; N=4) (Panorama Compounding, Van Nuys, CA). There were no differences in the responses of the participants receiving 400 or 800 mg N-acetyl cysteine; thus, these data were pooled to increase the power of the statistics. Participants received nine doses of the study medications with TID dosing on days 2 and 3 and 1 to 2 doses on day 1 (depending on when they completed the predrug test session) and day 4 (2–4 h before the postdrug test session; see Figure 1).
Participants underwent phone and clinical interview/laboratory screening (S) before study enrollment (E). After 1 to 8 weeks, participants underwent a run-up procedure (R) to determine the safety of delivering cocaine (20 mg per 70 kg IV over 60 s). Afterwards, participants remained inpatient for 1 to 2 separate 4-day/3-night stays, each with one blinded, oral study medication: treatment A (oral N-acetyl cysteine; 400 or 800 mg TID) or B (oral baclofen; 20 mg TID). During each inpatient visit, the participant underwent a 1-h test session before and after drug treatment (pretest and posttest, respectively) while receiving a total of nine doses of the test medication during the 3 days between the two tests. Participants were discharged home on day 4, with some returning after a 7- to 14-day treatment washout period (W) for the second treatment arm of the study. After 12 to 16 days of the end of the final arm, participants returned for a discharge interview (DC) before study termination.
Human Test Sessions
During each 4-day visit, participants underwent separate test sessions on days 1 and 4 (pre- and post-drug treatment). Each test consisted of a 1-h session with three stimulus epochs, each starting with a 3- to 5-min baseline period designed to permit the determination of the capacity of each of the stimuli to induce high, rush, and craving. Cue-induction of high, rush, and craving was assessed during the first two epochs in which participants were exposed to a 4-min ‘neutral’ video depicting nonaffective, household scenes and a 4-min cocaine video depicting two cocaine-experienced males seated at a table simulating the use of crack cocaine. Drug induction of high, rush, and craving was assessed during the third epoch, in which participants received a cocaine infusion (20 mg/70 kg, IV) delivered in a volume of 10 ml/60 s in order to minimize the acute hedonic effect while maximizing drug craving (Abreu et al, 2001; Nelson et al, 2006). Subjects had an IV catheter inserted into the left forearm for drug delivery and the right forearm for drawing blood samples and as an alternate access site if needed. During the baseline and stimulus periods, participants completed visual analog scale (VAS) ratings of high, rush, and craving (Kufahl et al, 2005) resulting in 3–5 1-min ratings during each baseline, five 1-min ratings during the neutral and cocaine videos, and 10 1-min ratings following the cocaine infusion, as described below. A 5-min, two-back, visuospatial working memory (VSWM; see below) task was completed after each video with the goal of engaging working memory in order to reduce residual drug euphoria and craving.
Human Behavioral Ratings
Subjects were instructed to rate their current level of high (while viewing the word ‘HIGH’ through prism glasses on a back-projected screen), rush (‘RUSH’), and craving (‘CRAVING’) once every minute throughout the entire experiment, except during two 5-min VSWM periods. Participants were instructed to use their common ‘street’ definitions for the constructs above. Questions cycled in a fixed order and remained on screen until answered or for a maximum of 15 s. Subjects recorded their behavioral VAS ratings by using a joystick manipulandum to move a tab along a horizontal bar with anchors ‘Least Ever’ and ‘Most Ever’ at extrema and pushing a button at the desired rating.
Human VSWM Task
To cognitively engage the participant and distract from the internal hedonic or craving state in an attempt to return craving to baseline, a two-back VSWM task was incorporated into the session following each of the neutral and cocaine-related videos. In brief, the ‘Bouncing Boxes’ task consisted of 30 s on/30 s off blocks for 5 min that requires a constant update of the working memory register and constant engagement. This task was targeted at ∼80% correct responses, is amenable to training, and shows minimal practice effect; however, it is not based on fund of knowledge.
Statistical Methods
The SPSS statistics package (v17) was used to perform the statistical analyses. The data were analyzed using ANOVA with drug treatment (eg, dose of N-acetyl cysteine) as a between-subject factor, and time (time, day, or phase of the experiment) as a repeated factor. Significant main effects and interactions were further evaluated using Student's t-tests as comparisons involved only two groups. Because our goal was to examine the impact of N-acetyl cysteine on evoked craving, rush, and high, basal values for these measures collected immediately before each phase of the experiment were subtracted from the values obtained during/after the neutral video, drug video, and cocaine infusion (see Table 1 for baseline values). Furthermore, deconstruction of interactions obtained in the analysis of the human data was restricted to reveal an effect of day (pre- or post-drug treatment) or drug treatment to minimize the number of comparisons.
RESULTS
Daily N-Acetyl Cysteine Treatment Does Not Alter the Reinforcing or Euphoric Properties of Cocaine
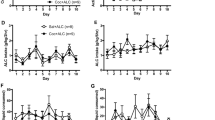
Figure 2 illustrates that N-acetyl cysteine administered before daily sessions of cocaine self-administration does not alter cocaine intake in rodents. An ANOVA comparing daily cocaine intake with treatment (saline or N-acetyl cysteine) as a between-subject variable and infusion number during each daily session as a within-subject variable did not yield a significant interaction (F11, 132=0.644, p=0.79), main effect of time (F11, 132=0.505, p=0.90), or main effect of treatment (F1, 12=0.43, p=0.525).
Daily N-acetyl cysteine does not alter the amount of cocaine self-administered by rats. Data depict the mean±SEM number of infusions across 12 maintenance self-administration sessions. Rats received N-acetyl cysteine (0 or 60 mg/kg, IP) 60 min before each session of cocaine self-administration (0.5 mg/kg, IV/infusion; 2 h/day; N=7/group).
In cocaine-dependent human participants, repeated N-acetyl cysteine administration did not alter self-reports of cocaine-induced rush or high. Figure 3 illustrates evoked high or rush (difference from respective baseline that preceded each stimulus) reported during the presentation of the neutral video, drug video, or following an infusion of cocaine on test day 1 (predrug test) and test day 2 (postdrug test). An ANOVA comparing evoked rush with drug treatment (baclofen or N-acetyl cysteine) as a between-subject factor and test day and phase of the experiment as repeated-measures variables failed to produce a significant three-way interaction (F2, 12=0.054, p=0.947), but yielded an interaction between drug treatment and experimental phase (F2, 12=4.3, p=0.039). Deconstruction of the two-way interaction failed to produce a significant effect involving treatment, indicating a lack of an effect of N-acetyl cysteine or baclofen on self-reports of rush (see Figure 3a). An ANOVA comparing evoked high with drug treatment as a between-subject factor and test day and phase of the experiment as repeated-measures variables also failed to produce a significant interaction or main effect involving test day (F1, 6=0.043, p=0.984) or treatment (F1, 6=0.706, p=0.433). Physiological measures (heart rate and blood pressure) did not differ in peak or baseline measures across treatment groups.
Daily N-acetyl cysteine does not alter cocaine-induced rush (a) or high (b) in cocaine-dependent human participants. Data depict a change in self-reports (mean±SEM) of rush and high from baseline values collected immediately before values obtained during exposure to a neutral video and cocaine video, or following a cocaine injection (20 mg/70 kg/60 s, IV). Measures of rush and high were obtained before and following daily baclofen (20 mg; TID; N=4) or N-acetyl cysteine (400 or 800 mg; TID; N=4) treatment.
Daily N-Acetyl Cysteine Treatment Reduces Cocaine-Induced Reinstatement in Rodents and Craving in Humans
Figure 4 illustrates nonreinforced operant responding on the active lever during the last extinction session, reinstatement test 1 (before N-acetyl cysteine treatment), or reinstatement test 2 (24 h after the last of seven daily N-acetyl cysteine treatments). An ANOVA comparing cocaine seeking with treatment (saline or N-acetyl cysteine) as a between-subject variable and experimental phase (extinction, reinstatement 1, or reinstatement 2) as a within-subject variable resulted in a significant interaction (F2, 46=4.08, p=0.023). Post hoc comparisons indicated that cocaine seeking by the two groups of rats did not differ before N-acetyl cysteine treatment (eg, extinction responding or first reinstatement test); however, responding was significantly reduced during the second reinstatement test in rats that had received daily N-acetyl cysteine treatment (t-test, p<0.05).
Daily N-acetyl cysteine decreases cocaine-induced reinstatement in rodents. Data depict the mean (±SEM) number of lever presses over during the last extinction test, or during two reinstatement tests. Rats self-administered cocaine (0.5 mg/kg/200 μl IV; 2-h/day; N=10–12/group) over 12 daily sessions. After 7 days of the last self-administration session, rats underwent daily extinction training in the absence of any drug treatment. This was followed by a test for cocaine-induced (10 mg/kg, IP) reinstatement. The day after the first reinstatement test, rats received seven daily injections of saline or N-acetyl cysteine (60 mg/kg, IP). The day after the seventh and final injection of saline or N-acetyl cysteine, rats underwent a second test for cocaine-induced reinstatement. #A significant difference from the first reinstatement test; t-test, p<0.05. *A significant difference from extinction responding, p<0.05.
In cocaine-dependent human participants, N-acetyl cysteine significantly decreased craving reported after an injection of cocaine. An ANOVA comparing evoked craving with drug treatment as a between-subject factor and test day and phase of the experiment as repeated-measures variables produced a significant three-way interaction (F2, 12=14.5, p=0.001). A post hoc comparison of the magnitude of craving reported following a cocaine injection on the first test day (before daily treatment with N-acetyl cysteine or baclofen) and the second test day (after daily drug treatment) indicated that daily N-acetyl cysteine, but not baclofen, produced a significant reduction in cocaine-induced craving (Figure 5; t-test, p<0.05).
N-acetyl cysteine reduces craving reported after a cocaine injection by human cocaine-dependent participants. Data depict a change in self-reports (mean±SEM) of craving from baseline values collected immediately before values obtained during exposure to a neutral video and cocaine video, or following a cocaine injection (20 mg/70 kg/60 s, IV). Measures of craving were obtained before and after daily baclofen (20 mg; TID; N=4) or N-acetyl cysteine (400 or 800 mg; TID; N=4) treatment. *A significant difference from craving reported following a cocaine injection on test day 1 (eg, pre-baclofen or N-acetyl cysteine treatment), p<0.05, t-test.
DISCUSSION
The goals of this study were to further evaluate the therapeutic potential of N-acetyl cysteine treatment and, in doing so, examine the predictive validity of the reinstatement paradigm. Specifically, we studied the impact of repeated administration of N-acetyl cysteine on cocaine intake and cocaine-primed reinstatement in rodents and on self-reports of rush, high, and craving by nontreatment-seeking, cocaine-dependent human participants. The primary findings were that repeated N-acetyl cysteine lessened cocaine-induced increases in drug seeking in rats and craving in humans without altering the reinforcing effects of cocaine in rats and the euphoric properties of cocaine in humans. These findings further establish system xc– as a novel treatment for addiction, lend support for the use of the self-administration/reinstatement paradigm as a model of aspects of addiction, and provide insight into why N-acetyl cysteine reduces drug use as revealed by earlier studies (Mardikian et al, 2007).
Daily N-acetyl cysteine administration in this study significantly blunted cocaine-induced increases in extinguished drug seeking in rodents. There are several key points to emphasize from this novel data set. Specifically, neither repeated administration of N-acetyl cysteine nor a 24-h delay between the last dosing of N-acetyl cysteine and behavioral testing diminished the effectiveness of this drug in blocking cocaine-induced reinstatement that has been established in earlier studies utilizing acute administration immediately before testing (Baker et al, 2003, 2008; Kau et al, 2008). Furthermore, these data may indicate that reinstatement can be blocked by very low levels of N-acetyl cysteine or the accumulation of an active metabolite, such as cystine, as the half-life of systemic N-acetyl cysteine has been estimated to be 6.25 h (Olsson et al, 1988), although this was conducted in humans. Alternatively, repeated N-acetyl cysteine may have resulted in reduced reinstatement by masking or reversing cocaine-induced plasticity that is necessary for cocaine reinstatement. This would extend our earlier observation that repeated N-acetyl cysteine treatment during self-administration training prevents the development of plasticity that is required for cocaine-induced reinstatement (Madayag et al, 2007). In support of the latter conclusion, repeated N-acetyl cysteine has been shown to produce an enduring suppression of cue- and heroin-induced reinstatement that persists for at least 40 days (Zhou and Kalivas, 2008).
Daily N-acetyl cysteine administered to nontreatment seeking, cocaine-dependent participants reduced craving produced by a cocaine infusion. There are at least two interpretations of the data. First, the reduced craving observed on the second test session (eg, after N-acetyl cysteine treatment) may have been because of an order effect as we lack a placebo group. However, we did not observe a change in cocaine-evoked craving in participants receiving baclofen. A more likely interpretation is that N-acetyl cysteine reduced craving produced by a cocaine injection. We also observed a reduction in craving produced by a cocaine video in subjects treated with N-acetyl cysteine, although the effect was not significant because of insufficient power. The lack of a significant change may be because of the preliminary nature of the clinical data arising from a lack of power. In support, an earlier report has observed that N-acetyl cysteine significantly reduces desire to use cocaine generated by a cocaine video (LaRowe et al, 2007). Thus, the capacity of N-acetyl cysteine to reduce craving or desire to use cocaine following exposure to cocaine or cocaine-paired cues may provide insight into the observation of reduced cocaine use following repeated N-acetyl cysteine treatment (Mardikian et al, 2007). In addition to cocaine, N-acetyl cysteine has been shown to decrease tobacco use by smokers, reduce pathological gambling, and lessen other types of compulsive behaviors such as trichotillomania (Grant et al, 2007, 2009; Knackstedt et al, 2009). Although additional studies are needed to confirm these preliminary findings, the extant data support the potential for N-acetyl cysteine to reduce multiple types of addiction and other disorders of compulsive behavior.
Repeated administration of N-acetyl cysteine in this study failed to alter the euphoric or reinforcing properties of cocaine as assessed in humans and rats, respectively. Because of the preliminary nature of the clinical experiment, it is possible that we lacked sufficient power to detect an impact of N-acetyl cysteine on the euphoric properties of cocaine in humans or that a higher dose of N-acetyl cysteine may have produced a more robust effect on these measures. However, at the very least, the human data indicate that cocaine-induced craving is more sensitive to the effects of repeated N-acetyl cysteine than the euphoric properties of cocaine. Furthermore, the rodent data are consistent with our earlier data demonstrating that N-acetyl cysteine does not alter the acute effects of cocaine, such as drug self-administration or cocaine-induced locomotor activity (Madayag et al, 2007). Thus, the existing data pose an intriguing question regarding the use of N-acetyl cysteine as a treatment for addiction that merits further study. Specifically, is the value of a pharmacotherapy for addiction significantly diminished if the acute, euphoric properties of the previously abused drug persist in a manner that could maintain recreational drug use, provided there is a reduction in compulsive, uncontrollable drug use?
An important goal of this translational study was to provide a preliminary assessment of the predictive validity of the reinstatement paradigm. There have been concerns raised regarding the number of apparent false positives whereby potential medications that block reinstatement fail to produce a positive effect in clinical trials. However, as has been noted in several excellent reviews, it is currently difficult to draw conclusions regarding the predictive validity of the reinstatement paradigm (Epstein and Preston, 2003; Epstein et al, 2006; Yahyavi-Firouz-Abadi and See, 2009), in part because of the discrepancies of what is actually measured in preclinical reinstatement studies and clinical trials. Preclinical studies typically examine the capacity of acute administration of a putative medication to alter evoked drug seeking following exposure to a discrete stimulus, such as stress or a drug injection. In contrast, clinical trials often assess drug use following chronic administration of the medication. Thus, the key assumptions being made are that: (1) the effectiveness of a compound is not diminished following repeated administration, (2) the stimuli used to reinstate responding in rodents model aspects of addiction, such as craving, and (3) the phenomena being modeled, such as craving, actually contribute to continued drug use. In this study, we sought to deconstruct the numerous leaps between typical preclinical reinstatement studies and a clinical trial by more closely matching the outcomes assessed in rodents and humans. Specifically, we examined the capacity of repeated N-acetyl cysteine to alter evoked increases in reinstatement and craving (eg, change from baseline following a cocaine injection). Although the human data are preliminary and may lack sufficient power, our data indicate that N-acetyl cysteine failed to alter cocaine reinforcement in rodents and reward-based measures in humans, but significantly reduced cocaine-induced increases in drug seeking in rodents and craving in humans. Thus, the data appear to support the use of the reinstatement model as a drug screen for the development of medications to treat drug addiction.
References
Abreu ME, Bigelow GE, Fleisher L, Walsh SL (2001). Effect of intravenous injection speed on responses to cocaine and hydromorphone in humans. Psychopharmacology (Berl) 154: 76–84.
Baker DA, Madayag A, Kristiansen LV, Meador-Woodruff JH, Haroutunian V, Raju I (2008). Contribution of cystine-glutamate antiporters to the psychotomimetic effects of phencyclidine. Neuropsychopharmacology 33: 1760–1772.
Baker DA, McFarland K, Lake RW, Shen H, Tang XC, Toda S et al (2003). Neuroadaptations in cystine-glutamate exchange underlie cocaine relapse. Nat Neurosci 6: 743–749.
Breiter HC, Gollub RL, Weisskoff RM, Kennedy DN, Makris N, Berke JD et al (1997). Acute effects of cocaine on human brain activity and emotion. Neuron 19: 591–611.
Cornish JL, Kalivas PW (2000). Glutamate transmission in the nucleus accumbens mediates relapse in cocaine addiction. J Neurosci 20: RC89.
Epstein DH, Preston KL (2003). The reinstatement model and relapse prevention: a clinical perspective. Psychopharmacology (Berl) 168: 31–41.
Epstein DH, Preston KL, Stewart J, Shaham Y (2006). Toward a model of drug relapse: an assessment of the validity of the reinstatement procedure. Psychopharmacology (Berl) 189: 1–16.
Grant JE, Kim SW, Odlaug BL (2007). N-acetyl cysteine, a glutamate-modulating agent, in the treatment of pathological gambling: a pilot study. Biol Psychiatry 62: 652–657.
Grant JE, Odlaug BL, Kim SW (2009). N-acetylcysteine, a glutamate modulator, in the treatment of trichotillomania: a double-blind, placebo-controlled study. Arch Gen Psychiatry 66: 756–763.
Katz JL, Higgins ST (2003). The validity of the reinstatement model of craving and relapse to drug use. Psychopharmacology (Berl) 168: 21–30.
Kau KS, Madayag A, Mantsch JR, Grier MD, Abdulhameed O, Baker DA (2008). Blunted cystine-glutamate antiporter function in the nucleus accumbens promotes cocaine-induced drug seeking. Neuroscience 155: 530–537.
Knackstedt LA, LaRowe S, Mardikian P, Malcolm R, Upadhyaya H, Hedden S et al (2009). The role of cystine-glutamate exchange in nicotine dependence in rats and humans. Biol Psychiatry 65: 841–845.
Kufahl PR, Li Z, Risinger RC, Rainey CJ, Wu G, Bloom AS et al (2005). Neural responses to acute cocaine administration in the human brain detected by fMRI. Neuroimage 28: 904–914.
LaRowe SD, Myrick H, Hedden S, Mardikian P, Saladin M, McRae A et al (2007). Is cocaine desire reduced by N-acetylcysteine? Am J Psychiatry 164: 1115–1117.
Li SJ, Biswal B, Li Z, Risinger R, Rainey C, Cho JK et al (2000). Cocaine administration decreases functional connectivity in human primary visual and motor cortex as detected by functional MRI. Magn Reson Med 43: 45–51.
Li X, Li J, Gardner EL, Xi ZX (2010). Activation of mGluR7s inhibits cocaine-induced reinstatement of drug-seeking behavior by a nucleus accumbens glutamate-mGluR2/3 mechanism in rats. J Neurochem 114: 1368–1380.
Madayag A, Lobner D, Kau KS, Mantsch JR, Abdulhameed O, Hearing M et al (2007). Repeated N-acetylcysteine administration alters plasticity-dependent effects of cocaine. J Neurosci 27: 13968–13976.
Mardikian PN, LaRowe SD, Hedden S, Kalivas PW, Malcolm RJ (2007). An open-label trial of N-acetylcysteine for the treatment of cocaine dependence: a pilot study. Prog Neuropsychopharmacol Biol Psychiatry 31: 389–394.
McFarland K, Lapish CC, Kalivas PW (2003). Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci 23: 3531–3537.
National_Research_Council (1996). NIH Guide for the Care and Use of Laboratory Animals. National Academy Press: Washington, DC.
Nelson RA, Boyd SJ, Ziegelstein RC, Herning R, Cadet JL, Henningfield JE et al (2006). Effect of rate of administration on subjective and physiological effects of intravenous cocaine in humans. Drug Alcohol Depend 82: 19–24.
Oldfield RC (1971). The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9: 97–113.
Olsson B, Johansson M, Gabrielsson J, Bolme P (1988). Pharmacokinetics and bioavailability of reduced and oxidized N-acetylcysteine. Eur J Clin Pharmacol 34: 77–82.
Pendyala L, Creaven PJ (1995). Pharmacokinetic and pharmacodynamic studies of N-acetylcysteine, a potential chemopreventive agent during a phase I trial. Cancer Epidemiol Biomarkers Prev 4: 245–251.
Volkow ND, Wang GJ, Ma Y, Fowler JS, Wong C, Ding YS et al (2005). Activation of orbital and medial prefrontal cortex by methylphenidate in cocaine-addicted subjects but not in controls: relevance to addiction. J Neurosci 25: 3932–3939.
Xi ZX, Li X, Peng XQ, Li J, Chun L, Gardner EL et al (2010). Inhibition of NAALADase by 2-PMPA attenuates cocaine-induced relapse in rats: a NAAG-mGluR2/3-mediated mechanism. J Neurochem 112: 564–576.
Yahyavi-Firouz-Abadi N, See RE (2009). Anti-relapse medications: preclinical models for drug addiction treatment. Pharmacol Ther 124: 235–247.
Zhou W, Kalivas PW (2008). N-acetylcysteine reduces extinction responding and induces enduring reductions in cue- and heroin-induced drug-seeking. Biol Psychiatry 63: 338–340.
Acknowledgements
We gratefully acknowledge the generous support provided by the following grants: DA017328 (to DAB), DA025617 (to DAB and JRM), DA015758 (to JRM), DA010214 (to SJL), DA00486 (to RCR), DA09465 (to RCR), DA019754 (to SLA), and MCW GCRC RR00058.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Baker co-founded and receives consulting fees and research funding from Promentis Pharmaceuticals. He currently serves on the Board of Directors for Promentis Pharmaceuticals. He is an inventor on patents currently under review regarding the use of cysteine prodrugs for the treatment of addiction disorders and schizophrenia. Dr John Mantsch co-founded and receives consultant fees and research funding from Promentis Pharmaceuticals. Dr Robert Risinger is currently employed by Brisol-Myers Squibb, and has previously received compensation for employment by Ortho-McNeil Janssen Scientific Affairs, LLC; however, neither company has any involvement with this work, which was completed when he was an employee of the Medical College of Wisconsin. The other authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Amen, S., Piacentine, L., Ahmad, M. et al. Repeated N-Acetyl Cysteine Reduces Cocaine Seeking in Rodents and Craving in Cocaine-Dependent Humans. Neuropsychopharmacol 36, 871–878 (2011). https://doi.org/10.1038/npp.2010.226
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2010.226
Keywords
This article is cited by
-
Cannabinoid use is enhanced by stress and changes conditioned stress responses
Neuropsychopharmacology (2022)
-
Mechanistic Effects and Use of N-acetylcysteine in Substance Use Disorders
Current Behavioral Neuroscience Reports (2022)
-
The Potential of N-Acetyl-L-Cysteine (NAC) in the Treatment of Psychiatric Disorders
CNS Drugs (2022)
-
N-acetylcysteine in substance use disorder: a lesson from preclinical and clinical research
Pharmacological Reports (2021)
-
Potential of Glial Cell Modulators in the Management of Substance Use Disorders
CNS Drugs (2020)