Abstract
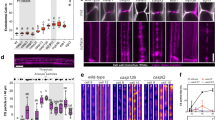
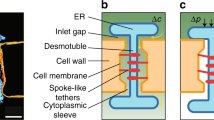
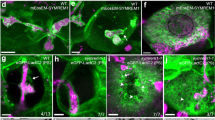
Plasmodesmata are remarkable cellular machines responsible for the controlled exchange of proteins, small RNAs and signalling molecules between cells. They are lined by the plasma membrane (PM), contain a strand of tubular endoplasmic reticulum (ER), and the space between these two membranes is thought to control plasmodesmata permeability. Here, we have reconstructed plasmodesmata three-dimensional (3D) ultrastructure with an unprecedented level of 3D information using electron tomography. We show that within plasmodesmata, ER–PM contact sites undergo substantial remodelling events during cell differentiation. Instead of being open pores, post-cytokinesis plasmodesmata present such intimate ER–PM contact along the entire length of the pores that no intermembrane gap is visible. Later on, during cell expansion, the plasmodesmata pore widens and the two membranes separate, leaving a cytosolic sleeve spanned by tethers whose presence correlates with the appearance of the intermembrane gap. Surprisingly, the post-cytokinesis plasmodesmata allow diffusion of macromolecules despite the apparent lack of an open cytoplasmic sleeve, forcing the reassessment of the mechanisms that control plant cell–cell communication.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Stahl, Y. & Faulkner, C. Receptor complex mediated regulation of symplastic traffic. Trends Plant Sci. 21, 450–459 (2016).
Otero, S., Helariutta, Y. & Benitez-alfonso, Y. Symplastic communication in organ formation and tissue patterning. Curr. Opin. Plant Biol. 29, 21–28 (2016).
Benkovics, A. H. & Timmermans, M. C. P. Developmental patterning by gradients of mobile small RNAs. Curr. Opin. Genet. Dev. 27, 83–91 (2014).
Vaddepalli, P. et al. The C2-domain protein QUIRKY and the receptor-like kinase STRUBBELIG localize to plasmodesmata and mediate tissue morphogenesis in Arabidopsis thaliana. Development 141, 4139–4148 (2014).
Daum, G., Medzihradszky, A., Suzaki, T. & Lohmann, J. U. A mechanistic framework for noncell autonomous stem cell induction in Arabidopsis. Proc. Natl Acad. Sci. USA 111, 14619–14624 (2014).
Stahl, Y. et al. Moderation of Arabidopsis root stemness by CLAVATA1 and ARABIDOPSIS CRINKLY4 receptor kinase complexes. Curr. Biol. 23, 362–371 (2013).
Benitez-alfonso, Y. et al. Symplastic intercellular connectivity regulates lateral root patterning. Dev. Cell 26, 136–147 (2013).
Faulkner, C. et al. LYM2-dependent chitin perception limits molecular flux via plasmodesmata. Proc. Natl Acad. Sci. USA 110, 9166–9170 (2013).
Wu, S. et al. Symplastic signaling instructs cell division, cell expansion, and cell polarity in the ground tissue of Arabidopsis thaliana roots. Proc. Natl Acad. Sci. USA 113, 11621–11626 (2016).
Lee, J.-Y. & Lu, H. Plasmodesmata: the battleground against intruders. Trends Plant Sci. 16, 201–210 (2011).
Vatén, A. et al. Callose biosynthesis regulates symplastic trafficking during root development. Dev. Cell 21, 1144–1155 (2011).
Tilsner, J. et al. Replication and trafficking of a plant virus are coupled at the entrances of plasmodesmata. J. Cell Biol. 201, 981–995 (2013).
Benitez-Alfonso, Y., Faulkner, C., Ritzenthaler, C. & Maule, A. J. Plasmodesmata: gateways to local and systemic virus infection. Mol. Plant. Microbe. Interact. 23, 1403–1412 (2010).
Ding, B., Turgeon, R. & Parthasarathy, M. V. Substructure of freeze-substituted plasmodesmata. Protoplasma 169, 28–41 (1992).
Tilsner, J., Amari, K. & Torrance, L. Plasmodesmata viewed as specialised membrane adhesion sites. Protoplasma 248, 39–60 (2011).
Knox, K. et al. Putting the squeeze on plasmodesmata: a role for RETICULONs in primary plasmodesmata formation. Plant Physiol. 168, 1563–1572 (2015).
Salmon, M. S. & Bayer, E. M. F. Dissecting plasmodesmata molecular composition by mass spectrometry-based proteomics. Front. Plant Sci. 3, 307 (2012).
Simpson, C., Thomas, C., Findlay, K., Bayer, E. & Maule, A. J. An Arabidopsis GPI-anchor plasmodesmal neck protein with callose binding activity and potential to regulate cell-to-cell trafficking. Plant Cell 21, 581–594 (2009).
Thomas, C. L., Bayer, E. M., Ritzenthaler, C., Fernandez-Calvino, L. & Maule, A. J. Specific targeting of a plasmodesmal protein affecting cell-to-cell communication. PLoS Biol. 6, e7 (2008).
Fernandez-Calvino, L. et al. Arabidopsis plasmodesmal proteome. PLoS ONE 6, e18880 (2011).
Lee, J.-Y. et al. A plasmodesmata-localized protein mediates crosstalk between cell-to-cell communication and innate immunity in Arabidopsis. Plant Cell 23, 3353–3373 (2011).
Grison, M. S. et al. Specific membrane lipid composition is important for plasmodesmata function in Arabidopsis. Plant Cell 27, 1228–1250 (2015).
Prinz, W. A. Bridging the gap: membrane contact sites in signaling, metabolism, and organelle dynamics. J. Cell Biol. 205, 759–769 (2014).
Pérez-Sancho, J. et al. Stitching organelles: organization and function of specialized membrane contact sites in plants. Trends Cell Biol. 26, 705–717 (2016).
Gallo, A., Vannier, C. & Galli, T. Endoplasmic reticulum–plasma membrane associations: structures and functions. Annu. Rev. Cell Dev. Biol. 32, 279–301 (2016).
von Filseck, J. M. et al. Phosphatidylserine transport by ORP/Osh proteins is driven by phosphatidylinositol 4-phosphate. Science 349, 432–436 (2015).
Chang, C.-L. et al. Feedback regulation of receptor-induced Ca2+ signaling mediated by e-syt1 and nir2 at endoplasmic reticulum-plasma membrane junctions. Cell Rep. 5, 813–825 (2013).
Giordano, F. et al. PI(4,5)P2-dependent and Ca2+-regulated ER–PM interactions mediated by the extended synaptotagmins. Cell 153, 1494–1509 (2013).
Chu, B.-B. et al. Cholesterol transport through lysosome-peroxisome membrane contacts. Cell 161, 291–306 (2015).
Tilsner, J., Nicolas, W., Rosado, A. & Bayer, E. M. Staying tight: plasmodesmata membrane contact sites and the control of cell-to-cell connectivity. Annu. Rev. Plant Biol. 67, 337–364 (2016).
Gisel, A., Barella, S., Hempel, F. D. & Zambryski, P. C. Temporal and spatial regulation of symplastic trafficking during development in Arabidopsis thaliana apices. Development 126, 1879–1889 (1999).
Sivaguru, M. et al. Aluminum-induced 1,3-β-D-glucan inhibits cell-to-cell trafficking of molecules through plasmodesmata. A new mechanism of aluminum toxicity in plants. Plant Physiol. 124, 991–1006 (2000).
Li, W. et al. Callose deposition at plasmodesmata is a critical factor in restricting the cell-to-cell movement of soybean mosaic virus. Plant Cell Rep. 31, 905–916 (2012).
Oparka, K. J. et al. Simple, but not branched, plasmodesmata allow the nonspecific trafficking of proteins in developing tobacco leaves. Cell 97, 743–754 (1999).
Kim, I. & Zambryski, P. C. Cell-to-cell communication via plasmodesmata during Arabidopsis embryogenesis. Curr. Opin. Plant Biol. 8, 593–599 (2005).
Schulz, A. Plasmodesmata widening accompanies the short-term increase in symplasmic phloem unloading in pea root tips under osmotic stress. Protoplasma 188, 22–37 (1995).
Brunkard, J. O., Runkel, A. M. & Zambryski, P. C. The cytosol must flow: intercellular transport through plasmodesmata. Curr. Opin. Cell Biol. 35, 13–20 (2015).
Beck, M. et al. Nuclear pore complex structure and dynamics revealed by cryoelectron tomography. Science 306, 1387–1390 (2004).
Overall, R. L., Wolfe, J. & Gunning, B. E. S. Intercellular communication in Azolla roots: I. Ultrastructure of plasmodesmata. Protoplasma 111, 134–150 (1982).
Tilney, L. G., Cooke, T. J., Connelly, P. S. & Tilney, M. S. The structure of plasmodesmata as revealed by plasmolysis, detergent extraction, and protease digestion. J. Cell Biol. 112, 739–747 (1991).
Moore, R., Fondren, W. M., Koon, E. C. & Wang, C. L. The influence of gravity on the formation of amyloplasts in columella cells of Zea mays L. Plant Physiol. 82, 867–868 (1986).
Bennett, T. et al. SOMBRERO, BEARSKIN1, and BEARSKIN2 regulate root cap maturation in Arabidopsis. Plant Cell 22, 640–654 (2010).
Staehelin, L. a., Zheng, H. Q., Yoder, T. L., Smith, J. D. & Todd, P. Columella cells revisited: novel structures, novel properties, and a novel gravisensing model. Gravit. Space Biol. Bull. 13, 95–100 (2000).
Dolan, L. et al. Cellular-organization of the Arabidopsis thaliana root. Development 119, 71–84 (1993).
Hamamoto, L., Hawes, M. C. & Rost, T. L. The production and release of living root cap border cells is a function of root apical meristem type in dicotyledonous angiosperm plants. Ann. Bot. 97, 917–923 (2006).
Overall, R. L. & Blackman, L. M. A model of the macromolecular structure of plasmodesmata. Trends Plant Sci. 1, 307–311 (1996).
Oparka, K. J. Getting the message across: how do plant cells exchange macromolecular complexes? Trends Plant Sci. 9, 33–41 (2004).
White, R. & Barton, D. The cytoskeleton in plasmodesmata: a role in intercellular transport? J. Exp. Bot. 62, 5249–5266 (2011).
Radford, J. E. & White, R. G. Inhibitors of myosin, but not actin, alter transport through Tradescantia plasmodesmata. Protoplasma 248, 205–216 (2011).
Mongrand, S., Stanislas, T., Bayer, E. M. F., Lherminier, J. & Simon-Plas, F. Membrane rafts in plant cells. Trends Plant Sci. 15, 656–663 (2010).
Sevilem, I., Yadav, S. R. & Helariutta, Y. Plasmodesmata: channels for intercellular signaling during plant growth and development. 1217, 3–24 (2015).
White, R. G. et al. Actin associated with plasmodesmata. Protoplasma 180, 169–184 (1994).
Radford, J. E. & White, R. G. Localization of a myosin-like protein to plasmodesmata. Plant J. 14, 743–750 (1998).
Faulkner, C. R., Blackman, L. M., Collings, D. A., Cordwell, S. J. & Overall, R. L. Anti-tropomyosin antibodies co-localise with actin microfilaments and label plasmodesmata. Eur. J. Cell Biol. 88, 357–369 (2009).
Ding, B., Kwon, M. & Warnberg, L. Evidence that actin filaments are involved in controlling the permeability of plasmodesmata in tobacco mesophyll. Plant J. 10, 157–164 (1996).
Sheahan, M. B., Staiger, C. J., Rose, R. J. & McCurdy, D. W. A green fluorescent protein fusion to actin-binding domain 2 of Arabidopsis fimbrin highlights new features of a dynamic actin cytoskeleton in live plant cells. Plant Physiol. 136, 3968–3978 (2004).
Boutté, Y. et al. Endocytosis restricts Arabidopsis KNOLLE syntaxin to the cell division plane during late cytokinesis. EMBO J. 29, 546–558 (2010).
Marais, C. et al. The Qb-SNARE Memb11 interacts specifically with Arf1 in the Golgi apparatus of Arabidopsis thaliana. J. Exp. Bot. 66, 6665–6678 (2015).
Bradley, M. O. Microfilaments and cytoplasmic streaming: inhibition of streaming with cytochalasin. J. Cell Sci. 12, 327–343 (1973).
Bayer, E., Thomas, C. L. & Maule, A. J. Plasmodesmata in Arabidopsis thaliana suspension cells. Protoplasma 223, 93–102 (2004).
Hawes, C. R., Juniper, B. E. & Horne, J. C. Low and high voltage electron microscopy of mitosis and cytokinesis in maize roots. Planta 152, 397–407 (1981).
Hepler, P. K. Endoplasmic reticulum in the formation of the cell plate and plasmodesmata. Protoplasma 111, 121–133 (1982).
Rutschow, H. L., Baskin, T. I. & Kramer, E. M. Regulation of solute flux through plasmodesmata in the root meristem. Plant Physiol. 155, 1817–1826 (2011).
Wang, N. & Fisher, D. B. The use of fluorescent tracers to characterize the post-phloem transport pathway in maternal tissues of developing wheat grains. Plant Physiol. 104, 17–27 (1994).
Terry, B. R., Matthews, E. K. & Haseloff, J. Molecular characterization of recombinant green fluorescent protein by fluorescence correlation microscopy. Biochem. Biophys. Res. Commun. 217, 21–27 (1995).
Imlau, A., Truernit, E. & Sauer, N. Cell-to-cell and long-distance trafficking of the green fluorescent protein in the phloem and symplastic unloading of the protein into sink tissues. Plant Cell 11, 309–322 (1999).
Blackman, L. M., Harper, J. D. I. & Overall, R. L. Localization of a centrin-like protein to higher plant plasmodesmata. Eur. J. Cell Biol. 78, 297–304 (1999).
Fernández-Busnadiego, R., Saheki, Y. & De Camilli, P. Three-dimensional architecture of extended synaptotagmin-mediated endoplasmic reticulum–plasma membrane contact sites. Proc. Natl Acad. Sci. USA 112, E2004–E2013 (2015).
Li, F. et al. Energetics and dynamics of SNAREpin folding across lipid bilayers. Nat. Struct. Mol. Biol. 14, 890–896 (2007).
Olesen, P. The neck constriction in plasmodesmata. Planta 144, 349–358 (1979).
Burch-Smith, T. M. & Zambryski, P. C. Loss of increased size exclusion limit (ise)1 or ise2 increases the formation of secondary plasmodesmata. Curr. Biol. 20, 989–993 (2010).
Bell, K. & Oparka, K. Imaging plasmodesmata. Protoplasma 248, 9–25 (2011).
Robinson-Beers, K. & Evert, R. F. Ultrastructure of and plasmodesmatal frequency in mature leaves of sugarcane. Planta 184, 291–306 (1991).
Eisenberg-Bord, M., Shai, N., Schuldiner, M. & Bohnert, M. A tether is a tether is a tether: tethering at membrane contact sites. Dev. Cell 39, 395–409 (2016).
Duckett, C. M., Oparka, K. J., Prior, D. a M., Dolan, L. & Roberts, K. Dye-coupling in the root epidermis of Arabidopsis is progressively reduced during development. Development 120, 3247–3255 (1994).
Badelt, K., White, R. G., Overall, R. L. & Vesk, M. Ultrastructural specializations of the cell wall sleeve around plasmodesmata. Am. J. 81, 1422–1427 (2016).
Kim, I., Kobayashi, K., Cho, E. & Zambryski, P. C. Subdomains for transport via plasmodesmata corresponding to the apical-basal axis are established during Arabidopsis embryogenesis. Proc. Natl Acad. Sci. USA 102, 11945–11950 (2005).
Kim, I., Cho, E., Crawford, K., Hempel, F. D. & Zambryski, P. C. Cell-to-cell movement of GFP during embryogenesis and early seedling development in Arabidopsis. Proc. Natl. Acad. Sci. USA 102, 2227–2231 (2005).
Messaoudi, C., Boudier, T., Sanchez Sorzano, C. O. & Marco, S. Tomoj: tomography software for three-dimensional reconstruction in transmission electron microscopy. BMC Bioinformatics 8, 288 (2007).
Sorzano, C. O. S. et al. Marker-free image registration of electron tomography tilt-series. BMC Bioinformatics 10, 124 (2009).
Lucocq, J. Unbiased 3-D quantitation of ultrastructure in cell biology. Trends Cell Biol. 3, 354–358 (1993).
Sterio, D. C. The unbiased estimation of number and sizes of arbitrary particles using the disector. J. Microsc. 134, 127–136 (1984).
Acknowledgements
This work was supported by the grants by the Region Aquitaine (to E.M.B) and PEPS (Initial Support for Exploratory Projects to E.M.B) and National Agency for Research (Grant ANR-14-CE19-0006-01 to E.M.B). Electron and fluorescence microscopy analyses were performed at the Bordeaux Imaging Centre (http://www.bic.u-bordeaux.fr/). The Region Aquitaine also supported the acquisition of the electron microscope (grant no. 2011 13 04 007 PFM). We thank Y. Benitez-Alfonso for providing the Arabidopsis PA–GFP lines and S. Mongrand, V. Arondel, Y. Boutté, Y. Jaillais and C. Hawes for critical review of the article before submission.
Author information
Authors and Affiliations
Contributions
Electron microscopy and associated-quantitative analyses were done by W.J.N. with the help of S.T. and L.B. M.S.G. performed the cell-to-cell connectivity essays with the help of W.J.N and L.B. F.P.C. and L.B. provided technical support for the FRAP experiments and with image quantification and acquisition. All statistical analyses were run by W.J.N. and M.S.G. A.G. and M.F. performed the control tests for latrunculin and cytochalasin treatments in the roots. The manuscript was written by E.M.B. and W.J.N. with contributions of L.B., K.O. and J.T. Research was designed by E.M.B.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figure 1, Supplementary Figure 3, Supplementary Figure 5, Supplementary Figure 8. (PDF 5630 kb)
Supplementary Video 2: Cytoplasmic streaming of Golgi vesicles is altered upon inhibition of actin polymerization
Timelapse of Mb12:YFP lines (a–c) and Ng1:GFP lines (d–f) taken at t = 0, 30 min and 1 h of treatment at 1 image every 5 seconds. (AVI 3575 kb)
Supplementary Video 4: Type I plasmodesmata in Arabidopsis cultured cells
Electron tomography volume showing a typical type I plasmodesmata pore with no visible cytoplasmic sleeve. (AVI 29289 kb)
Supplementary Video 6: Type II plasmodesmata in Arabidopsis cultured cells
Electron tomography volume of the type II plasmodesmata shown in Supplementary Fig. 5a,b. (AVI 27965 kb)
Supplementary Video 7: Type II plasmodesmata in Arabidopsis cultured cells
Electron tomography volume of the type II plasmodesmata exhibited in Supplementary Fig. 5c,d. (AVI 17357 kb)
Rights and permissions
About this article
Cite this article
Nicolas, W., Grison, M., Trépout, S. et al. Architecture and permeability of post-cytokinesis plasmodesmata lacking cytoplasmic sleeves. Nature Plants 3, 17082 (2017). https://doi.org/10.1038/nplants.2017.82
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2017.82
This article is cited by
-
Accelerated remodeling of the mesophyll-bundle sheath interface in the maize C4 cycle mutant leaves
Scientific Reports (2022)
-
Analysis of plasmodesmata permeability using cultured tobacco BY-2 cells entrapped in microfluidic chips
Journal of Plant Research (2022)