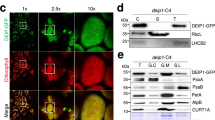
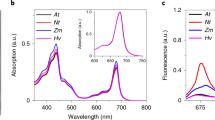
Abstract
Efficient photosynthesis depends on maintaining balance between the rate of light-driven electron transport occurring in photosystem I (PSI) and photosystem II (PSII), located in the chloroplast thylakoid membranes. Balance is achieved through a process of ‘state transitions’ that increases energy transfer towards PSI when PSII is overexcited (state II), and towards PSII when PSI is overexcited (state I). This is achieved through redox control of the phosphorylation state of light-harvesting antenna complex II (LHCII). PSI is served by both LHCII and four light-harvesting antenna complex I (LHCI) subunits, Lhca1, 2, 3 and 4. Here we demonstrate that despite unchanged levels of LHCII phosphorylation, absence of specific Lhca subunits reduces state transitions in Arabidopsis. The severest phenotype—observed in a mutant lacking Lhca4 (ΔLhca4)—displayed a 69% reduction compared with the wild type. Yet, surprisingly, the amounts of the PSI–LHCI–LHCII supercomplex isolated by blue native polyacrylamide gel electrophoresis (BN–PAGE) from digitonin-solubilized thylakoids were similar in the wild type and ΔLhca mutants. Fluorescence excitation spectroscopy revealed that in the wild type this PSI–LHCI–LHCII supercomplex is supplemented by energy transfer from additional LHCII trimers in state II, whose binding is sensitive to digitonin, and which are absent in ΔLhca4. The grana margins of the thylakoid membrane were found to be the primary site of interaction between this ‘extra’ LHCII and the PSI–LHCI–LHCII supercomplex in state II. The results suggest that the LHCI complexes mediate energetic interactions between LHCII and PSI in the intact membrane.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Haldrup, A., Jensen, P. E., Lunde, C. & Scheller, H. V. Balance of power: a view of the mechanism of photosynthetic state transitions. Trends in Plant Sci. 6, 301–305 (2001).
Ruban, A. V. & Johnson, M. P. Dynamics of higher plant photosystem cross-section associated with state transitions. Photosynth. Res. 99, 173–183 (2009).
Horton, P. & Black, M. T. Activation of adenosine 5′-triphosphate induced quenching of chlorophyll fluorescence by reduced plastoquinone. The basis of state I–state II transitions in chloroplasts. FEBS Lett. 119, 141–144 (1980).
Bellafiore, S., Barneche, F., Peltier, G. & Rochaix, J. D. State transitions and light adaptation require chloroplast thylakoid protein kinase STN7. Nature 433, 892–895 (2005).
Pribil, M., Pesaresi, P., Hertle, A., Barbato, R. & Leister, D. Role of plastid protein phosphatase TAP38 in LHCII dephosphorylation and thylakoid electron flow. PLoS Biol. 8, e1000288 (2010).
Shapiguzov, A., et al. The PPH1 phosphatase is specifically involved in LHCII dephosphorylation and state transitions in Arabidopsis. Proc. Natl Acad. Sci. USA 107, 4782–4787 (2010).
Frenkel, M., Bellafiore, S., Rochaix, J. D. & Jansson, S. Hierarchy amongst photosynthetic acclimation responses for plant fitness. Physiol. Plantarum 129, 455–459 (2007).
Tikkanen, M., Grieco, M., Kangasjärvi, S. & Aro, E. M. Thylakoid protein phosphorylation in higher plant chloroplasts optimizes electron transfer under fluctuating light. Plant Physiol. 152, 723–735 (2010).
Albertsson, P. Å. A quantitative model of the domain structure of the photosynthetic membrane. Trends Plant Sci. 6, 349–354 (2001).
Dekker, J. P. & Boekema, E. J. Supramolecular organization of thylakoid membrane proteins in green plants. Biochim. Biophys. Acta 1706, 12–39 (2005).
Galka, P., et al. Functional analyses of the plant photosystem I-light-harvesting complex II supercomplex reveal that light-harvesting complex II loosely bound to photosystem II is a very efficient antenna for photosystem I in state II. Plant Cell 24, 2963–2978 (2012).
Wientjes, E., Drop, B., Kouril, R., Boekema, E. J. & Croce, R. During state 1 to state 2 transition in Arabidopsis thaliana, the photosystem II supercomplex gets phosphorylated but does not disassemble. J. Biol. Chem. 288, 32821–32826 (2013).
Kyle, D. J., Staehelin, L. A. & Arntzen, C. J. Lateral mobility of the light-harvesting complex in chloroplast membranes controls excitation energy distribution in higher plants. Arch. Biochem. Biophys. 222, 527–541 (1983).
Chuartzman, S. G., et al. Thylakoid membrane remodeling during state transitions in Arabidopsis. Plant Cell 20, 1029–1039 (2008).
Pietrzykowska, M., et al. The light-harvesting chlorophyll a/b binding proteins Lhcb1 and Lhcb2 play complementary roles during state transitions in Arabidopsis. Plant Cell. 26, 3646–3660 (2014).
Bassi, R., Rigoni, F., Barbato, R. & Giacometti, G. M. Light-harvesting chlorophyll a/b proteins (LHCII) populations in phosphorylated membranes. Biochim. Biophys. Acta 936, 29–38 (1988).
Tikkanen, M., et al. Phosphorylation-dependent regulation of excitation energy distribution between the two photosystems in higher plants. Biochim. Biophys. Acta 1777, 425–432 (2008).
Lunde, C., Jensen, P. E., Haldrup, A., Knoetzel, J. & Scheller, H. V. The PSI-H subunit of photosystem I is essential for state transitions in plant photosynthesis. Nature 408, 613–615 (2000).
Zhang, S. & Scheller, H. V. Light-harvesting complex II binds to several small subunits of photosystem I. J. Biol. Chem. 279, 3180–3187 (2004).
Kouril, R., et al. Structural characterization of a complex of photosystem I and light-harvesting complex II of Arabidopsis thaliana. Biochemistry 44, 10935–10940 (2005).
Boekema, E. J., et al. Green plant photosystem I binds light-harvesting complex I on one side of the complex. Biochem. 40, 1029–1036 (2001).
Mazor, Y., Borovikova, A. & Nelson, N. The structure of plant photosystem I super-complex at 2.8 Å resolution. eLife 4, e07433 (2015).
Qin, X., Suga, M., Kuang, T. & Shen, J.-R. Structural basis for energy transfer pathways in the plant PSI-LHCI supercomplex. Science. 348, 989–995 (2015).
Klimmek, F. et al. Structure of the higher plant light harvesting complex I: in vivo characterization and structural interdependence of the Lhca proteins. Biochemistry 44, 3065–3073 (2005).
Morosinotto, T., Ballotarri, M., Klimmek, F., Jansson, S. & Bassi, R. The association of the antenna system to photosystem I in higher plants. J. Biol. Chem. 280, 31050–31058 (2005).
Wientjes, E., Oostergetel, G. T., Jansson, S., Boekema, E. J. & Croce, R. The role of Lhca complexes in the supramolecular organization of higher plant photosystem I. J. Biol. Chem. 284, 7803–7810 (2009).
Zhang, H., Goodman, H. M. & Jansson, S. Antisense inhibition of the photosystem I antenna protein Lhca4 in Arabidopsis thaliana. Plant Physiol. 115, 1525–1531 (1997).
Ganeteg, U., Strand, Å., Gustafsson, P. & Jansson, S. The properties of the chlorophyll a/b-binding proteins Lhca2 and Lhca3 studied in vivo using antisense inhibition. Plant Physiol. 127, 150–158 (2001).
Ganeteg, U., Külheim, C., Andersson, J. & Jansson, S. Is each light-harvesting complex protein important for plant fitness? Plant Physiol. 134, 502–509 (2004).
Ihalainen, J. A., et al. Excitation energy trapping in photosystem I complexes depleted in Lhca1 and Lhca4. FEBS Lett. 579, 4787–4791 (2005).
Järvi, S., Suorsa, M., Paakkarinen, V. & Aro, E. M. Optimized native gel systems for separation of thylakoid protein complexes: novel super- and mega-complexes. Biochem J. 439, 207–214 (2011).
Liu, Z. F., et al. Crystal structure of spinach major light-harvesting complex at 2.72 Å resolution. Nature 428, 287–292 (2004).
Grieco, M., Suorsa, M., Jajoo, A., Tikkanen, M. & Aro, E. M. Light-harvesting II antenna trimers connect energetically the entire photosynthetic machinery — including both photosystems II and I. Biochim. Biophys. Acta 1847, 607–619 (2015).
Jensen, P. E., Gilpin, M., Knoetzel, J. & Scheller, H. V. The PSI-K subunit of photosystem I is involved in the interaction between light-harvesting complex I and the photosystem I reaction center core. J. Biol. Chem. 275, 24701–24708 (2000).
Varotto, C., et al. Single and double knockouts of the genes for photosystem I subunits G, K, and H of Arabidopsis. effects on photosystem I composition, photosynthetic electron flow, and state transitions. Plant Physiol. 129, 616–624 (2002).
Bassi, R. & Simpson, D. Chlorophyll-protein complexes of barley photosystem I. Eur. J. Biochem. 163, 221–230 (1987).
Kim, E., Ahn, T. K. & Kumazak, S. Changes in antenna sizes of photosystems during state transitions in granal and stroma-exposed thylakoid membrane of intact chloroplasts in Arabidopsis mesophyll protoplasts. Plant Cell Physiol. 56, 759–768 (2015).
Bell, A. J., Frankel, L. K. & Bricker, T. M. High yield non-detergent isolation of photosystem I-light harvesting chlorophyll II membranes from spinach thylakoids. J. Biol. Chem. 290, 18429–18437 (2015).
Williams, R. S., Allen, J. F., Brain, A. P. R. & Ellis, R. J. Effect of Mg2+ on excitation energy transfer between LHCII and LHCI in a chlorophyll-protein complex. FEBS Lett. 225, 59–66 (1987).
Andreasson, E. & Albertsson, P. Å. Heterogeneity in Photosystem I - the larger antenna of Photosystem Iα is due to functional connection to a special pool of LHCII. Biochim. Biophys. Acta 1141, 175–182 (1993).
Jansson, S., Stefansson, H., Nystrom, U., Gustafsson, P. & Albertsson, P. Å. Antenna protein composition of PS I and PS II in thylakoid sub-domains. Biochim. Biophys. Acta 1320, 297–309 (1997).
Kouril, R., Wientjes, E., Bultema, J. B., Croce, R. & Boekema, E. J. High-light vs. low-light: Effect of light acclimation on photosystem II composition and organization in Arabidopsis thaliana. Biochim. Biophys. Acta 1827, 411–419 (2013).
Melis, A. Kinetic analysis of P-700 photoconversion: effect of secondary electron donation and plastocyanin inhibition. Arch. Biochem. Biophys. 217, 536–545 (1982).
Fristedt, R. et al. Phosphorylation of photosystem II controls functional macroscopic folding of photosynthetic membranes in Arabidopsis. Plant Cell. 21, 3950–3964 (2009).
Ruban, A. V. et al. Plasticity in the composition of the light harvesting antenna of higher plants preserves structural integrity and biological function. J. Biol. Chem. 281, 14981–14990 (2006).
Kiss, A., Crouchman, S., Ruban, A. V. & Horton, P. The PsbS protein controls the organisation of the photosystem II antenna in higher plant thylakoid membranes. J. Biol. Chem. 283, 3972–3978 (2008).
Acknowledgements
M.P.J. acknowledges funding from the Leverhulme Trust (U.K.) and the Krebs Institute and Project Sunshine at the University of Sheffield. A.V.R. gratefully acknowledges funding from the Biotechnology and Biological Sciences Research Council (U.K.), Leverhulme Trust and The Royal Society Wolfson Research Merit Award. C.N.H. and M.P.J. acknowledge research grant BB/M000265/1 from the Biotechnology and Biological Sciences Research Council (UK). C.N.H. was also supported by an Advanced Award 338895 from the European Research Council. This work was also supported as part of the Photosynthetic Antenna Research Center (PARC), an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, and Office of Basic Energy Sciences under Award Number DE-SC0001035. PARC's role was to provide partial support for C.N.H.
Author information
Authors and Affiliations
Contributions
S.L.B., P.M., M.A.W. and M.P.J. performed experiments. M.P.J. analysed the data. M.P.J, C.N.H., P.H., S.J. and A.V.R. designed the study and M.P.J. wrote the manuscript. All authors discussed the results and commented on the manuscript. P.H. and A.V.R. together carried out the preliminary experiments on which this study is based.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Benson, S., Maheswaran, P., Ware, M. et al. An intact light harvesting complex I antenna system is required for complete state transitions in Arabidopsis. Nature Plants 1, 15176 (2015). https://doi.org/10.1038/nplants.2015.176
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2015.176
This article is cited by
-
Analysis of state 1—state 2 transitions by genome editing and complementation reveals a quenching component independent from the formation of PSI-LHCI-LHCII supercomplex in Arabidopsis thaliana
Biology Direct (2023)
-
Characterizing compensatory mechanisms in the absence of photoprotective qE in Chlamydomonas reinhardtii
Photosynthesis Research (2023)
-
Spectral diversity of photosystem I from flowering plants
Photosynthesis Research (2023)
-
Generation and physiological characterization of genome-edited Nicotiana benthamiana plants containing zeaxanthin as the only leaf xanthophyll
Planta (2023)
-
Qualitative and quantitative evaluation of thylakoid complexes separated by Blue Native PAGE
Plant Methods (2022)