Abstract
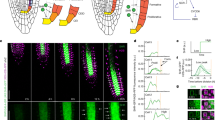
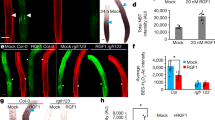
Cell size is determined by the duration and rate of growth and plays a central role in cell function. Root hairs are tip-growing cellular projections that emerge from the root epidermis and explore the soil to acquire nutrients and water1. Previously we demonstrated that the basic helix–loop–helix transcription factor root hair defective 6-like 4 (RSL4) is necessary and sufficient for root hair growth2. Here we show that RSL4 is synthesized in a 4-h pulse at the initiation of hair elongation and is gradually degraded by the 26S proteasome. The amount of RSL4 synthesis during this pulse is modulated as part of a root hair growth response to low phosphate. RSL4 synthesis increases in low phosphate and this increase prolongs the growth phase, resulting in the development of long root hairs. Our data demonstrate that the amount of RSL4 synthesized during the pulse directly determines the final size of the differentiated root hair cell. We propose that the modulation of growth-promoting transcription factors by external cues could be a general mechanism for the regulation of cell growth by environmental factors during development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Datta, S. et al. Root hairs: development, growth and evolution at the plant-soil interface. Plant Soil 346, 1–14 (2011).
Yi, K., Menand, B., Bell, E. & Dolan, L. A basic helix-loop-helix transcription factor controls cell growth and size in root hairs. Nature Genet. 42, 264–267 (2010).
Pires, N. D. et al. Recruitment and remodeling of an ancient gene regulatory network during land plant evolution. Proc. Natl Acad. Sci. USA 110, 9571–9576 (2013).
Genschik, P., Criqui, M. C., Parmentier, Y., Derevier, A. & Fleck, J. Cell cycle-dependent proteolysis in plants. Identification of the destruction box pathway and metaphase arrest produced by the proteasome inhibitor mg132. Plant Cell 10, 2063–2076 (1998).
Colon-Carmona, A., You, R., Haimovitch-Gal, T. & Doerner, P. Technical advance: spatio-temporal analysis of mitotic activity with a labile cyclin-GUS fusion protein. Plant J. 20, 503–508 (1999).
Capron, A., Okresz, L. & Genschik, P. First glance at the plant APC/C, a highly conserved ubiquitin-protein ligase. Trends Plant Sci. 8, 83–89 (2003).
Bates, T. R. & Lynch, J. P. Stimulation of root hair elongation in Arabidopsis thaliana by low phosphorus availability. Plant Cell Environ. 19, 529–538 (1996).
Brown, L. K., George, T. S., Dupuy, L. X. & White, P. J. A conceptual model of root hair ideotypes for future agricultural environments: what combination of traits should be targeted to cope with limited P availability? Ann. Bot. 112, 317–330 (2013).
Jang, G., Yi, K., Pires, N. D., Menand, B. & Dolan, L. RSL genes are sufficient for rhizoid system development in early diverging land plants. Development 138, 2273–2281 (2011).
Menand, B. et al. An ancient mechanism controls the development of cells with a rooting function in land plants. Science 316, 1477–1480 (2007).
Svistoonoff, S. et al. Root tip contact with low-phosphate media reprograms plant root architecture. Nature Genet. 39, 792–796 (2007).
Rubio, V. et al. A conserved MYB transcription factor involved in phosphate starvation signaling both in vascular plants and in unicellular algae. Genes Dev. 15, 2122–2133 (2001).
Jain, A., Nagarajan, V. K. & Raghothama, K. G. Transcriptional regulation of phosphate acquisition by higher plants. Cell Mol. Life Sci. 69, 3207–3224 (2012).
Ma, Z., Bielenberg, D. G., Brown, K. M. & Lynch, J. P. Regulation of root hair density by phosphorus availability in Arabidopsis thaliana. Plant Cell Environ. 24, 459–467 (2001).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the EquationSource math mrow msup mn2 mrow mo− miΔ miΔ msub miC mtextT method. Methods 25, 402–408 (2001).
Barber, R. D., Harmer, D. W., Coleman, R. A. & Clark, B. J. GAPDH as a housekeeping gene: analysis of GAPDH mRNA expression in a panel of 72 human tissues. Physiol. Genomics 21, 389–395 (2005).
Foreman, J. et al. Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 422, 442–446 (2003).
Acknowledgements
We thank M. Pernas-Ochoa, B. Catarino, S. Honkanen, V. Jones, H. Breuninger, T. Tam and J. Agusti for critically reading the manuscript. We acknowledge the help of I. Moore and M. Fricker with confocal microscopy. This research was funded by EMBO LTF (ALTF 817-2008) and EU Marie Curie FP7-PEOPLE-IEF-2008 (Project number-236802) grants (to S.D.) and ERC (European Research Council Advanced Grant EVO500, Project No: 25028) and BBSRC (BBG0198941) grants (to L.D.).
Author information
Authors and Affiliations
Contributions
S.D. performed the experiments with the help of H.P. S.D. and L.D. analysed and interpreted the data. L.D. and S.D. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Datta, S., Prescott, H. & Dolan, L. Intensity of a pulse of RSL4 transcription factor synthesis determines Arabidopsis root hair cell size. Nature Plants 1, 15138 (2015). https://doi.org/10.1038/nplants.2015.138
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2015.138
This article is cited by
-
OsUGE1 is directly targeted by OsGRF6 to regulate root hair length in rice
Theoretical and Applied Genetics (2023)
-
Apoplastic class III peroxidases PRX62 and PRX69 promote Arabidopsis root hair growth at low temperature
Nature Communications (2022)
-
Mechanistic Insights into Strigolactone Biosynthesis, Signaling, and Regulation During Plant Growth and Development
Journal of Plant Growth Regulation (2021)
-
Differences in nutrient foraging among Trifolium subterraneum cultivars deliver improved P-acquisition efficiency
Plant and Soil (2018)
-
A mechanistic framework for auxin dependent Arabidopsis root hair elongation to low external phosphate
Nature Communications (2018)