Abstract
The benefit of deep brain stimulation (DBS) in controlling the motor symptoms of Parkinson’s disease is well established, however, the impact on the non-motor symptoms (NMS) remains to be elucidated, although the growing investigative efforts are promising. This article reviews the reported data and considers the level of evidence available with regard to the effect of DBS on NMS total burden and on the cognitive, neuropsychiatric, sleep, pain, dysautonomic, and weight domains. Multiple case series suggest that DBS improves the burden of NMS by reducing prevalence, intensity, and non-motor fluctuations. There is level I evidence on the effect of DBS on cognition and mood. Slight cognitive decline has been reported in most class I studies, although the functional effect is probably minimal. Two randomized prospective studies reported no change in depression while improvement of anxiety has been reported by a class I trial. Prospective cohort studies point to improvement of hyperdopaminergic behaviors, such as impulse control disorders, while others report that hypodopaminergic states, like apathy, can appear after DBS. There is only class III evidence supporting the benefit of DBS on other NMS such as nocturnal sleep, pain, dysautonomia (urinary, gastrointestinal, cardiovascular, and sweating), and weight loss. Although preliminary results are promising, randomized prospectively controlled trials with NMS as primary end points are necessary to further explore the effect of DBS on these often invalidating symptoms and offer conclusions about efficacy.
Similar content being viewed by others
Introduction
The efficacy of deep brain stimulation (DBS) on the control of motor symptoms in Parkinson’s disease (PD) has been consistently demonstrated in Class I clinical trials and is thus well established.1–3 In the past two decades there is a growing recognition that non-motor symptoms (NMS) are fundamental to the concept of PD as they dominate the premotor stage and are prevalent throughout disease progression.4 A single NMS, like pain or depression, can overshadow the clinical picture in some patients, and NMS burden (NMSB) as a whole has been recently identified as the most important factor in determining the quality of life of PD patients.5 The importance of NMS makes the need for effective therapies increasingly evident and thus behooves the scientific community to investigate the effect of DBS on these symptoms. There are previous reviews concerning this topic,6–8 but the need for an updated critical review of the current data is warranted as the number of publications regarding NMS and DBS has grown exponentially in the past years. To give the reader an analytical overview, studies are reviewed in depth and classified according to the quality of evidence they provide: level I (large randomized controlled trials and meta-analysis), level II (small randomized trials and controlled prospective trials), and level III (prospective uncontrolled case series), following parameters established by the evidence-based task force commissioned by the Movement Disorders Society.9
This review includes clinical studies considering NMS as a primary or secondary outcome from DBS intervention and focusing on NMSB, non-motor fluctuations (NMF), cognition, neuropsychiatric symptoms (depression and anxiety, suicide attempts, apathy, and impulse control disorders (ICDs)), sleep, pain, autonomic (urinary, gastrointestinal, cardiovascular, and sweating), and weight gain.
The pathopysiology of NMS and DBS
The pathophysiology underlying cognitive and neuropsychiatric manifestations in PD is certainly complex and individually variable. Multiples studies corroborate widespread Lewy type-α synucleinopathy (LTS) in the neocortex as an important factor, and limbic and brainstem involvement may also have a role, as well as coexisting Alzheimer’s disease pathology, cerebrovascular disease, and amyloid angiopathy.10 The implication of cortico-basal ganglia (BG) circuitry disorders in the development of these NMS is also gaining attention as understanding of these networks progresses.
On the basis of the model developed by Alexander et al.11 in 1986, it is generally accepted that the BG constitute a part of three distinct functional and anatomical loops involving sensorimotor, associative, and limbic processing.
These networks have a role in weeding out relevant information from noise for the selection of movement patterns, actions, and goal-directed behaviors, while neurophysiological and neuroimaging studies have delineated their segregated topographical organization into parallel circuits. It is the ventral striatum and caudate output pathways that project, through the ventromedial globus pallidus, subthalamic nucleus (STN), substantia nigra, and thalamus, to the anterior cingulate and dorsolateral prefrontal and lateral orbitofrontal cortices (which send return pathways to these structures) thus regulating cognitive and behavioral processes. The well-known motor circuit involves the direct and indirect pathways and connects the dorsolateral putamen, globus pallidus interna (GPi), STN, and thalamus to the motor and premotor cortical areas.12,13 The dopamine depletion seen in PD leads to a reduction of selectivity and spacial focalization of this cortico-BG circuitry and thus may be the pathological substrate of, not only motor symptoms such as brdykinesia and rigidity, but also non-motor symptoms such as apathy and dysexecutive functions.
Dopamine replacement therapy may restores the balance. However, because striatal dopamine depletion is heterogeneous, treatment may lead to hypersensitivity of postsynaptic dopamine receptors in selective subterritories of the striatum. If the dorsolateral striatal territory expresses this sensitization, the result can be augmented movements (dyskinesias). If more ventromedial regions are implicated, ICDs may arise.12 It is well established that DBS surgery in the main relay centers of the cortico-BG circuitry, such as STN or GPi, has similar clinical effects to dopaminergic therapy. It is thought that DBS high frequency stimulation in the dorsolateral region of the STN facilitates movement by releasing the ‘No Go’ signal normally exerted by this nucleus on the motor BG circuitry. It is possible that an anteromedial electrode placement or current spread to this area may also release the ‘No Go’ signal on the limbic and associative circuits13 and thus produce cognitive and affective disinhibition, explaining some of the neuropsychiatric NMS effects seen after DBS.14,15
The pathophysiological basis for DBS effect on other non-motor symptoms such as sleep disorders, pain, and dysautonomia is complex and different mechanisms may have a role. First, adjacent regions near the STN, for example, the pedunculopontine nucleus, could be modulated by DBS, thus resulting in beneficial effects on sleep.16 Second, a modulation of BG circuitry may result in effects on autonomic centers of the thalamus, projecting to the anterior cingulate and lateral frontal cortex with beneficial effects of symptoms like sweating17 and bladder control.18
DBS effect of NMSB as a whole
NMSB can be evaluated by the NMS Questionnaire (NMSQuest),19 a qualitative patient completed tool, or measured in a quantitative fashion by the NMS Scale (NMSS).20 However, there have only been a few studies that have specifically used validated tools for measuring NMS in relation to DBS in PD (see Table 1). In a small open-label study including 10 PD patients, a 36% overall improvement in the mean NMSS score after STN DBS surgery was reported.21 However, the sample was too small to be able to report any meaningful subgroup/domain analysis. Using the NMSQuest-based scoring system, Nazzarro et al.22 studied 24 patients who underwent bilateral STN DBS and showed that the NMSQuest score decreased from 12 (severe NMSB) pre-surgery to 7 (moderate NMSB) post surgery at 1-year follow-up.22 Subsequently, Dafsari et al.23 have reported the first multicentre European DBS study of STN stimulation where NMS were the primary outcome.23 This was an open prospective observational study including 60 patients who underwent STN DBS and were evaluated at baseline and at 6-month follow-up with the NMSS. The authors reported a 42% improvement in NMSB, underpinned by a range of significant improvements in specific NMS (sleep, urinary, and perceptual problems). The immediate effect of DBS on NMSB has also been studied with nonspecific tools. Wolz et al.24 reported on 10 NMS in 34 patients with a median DBS time of 13 months. In this cohort, DBS did not have an immediate effect on the frequencies of a wide range of autonomic, sensory, cognitive, and neuropsychiatric NMS as assessed by a semi-structured interview, and only a significant improvement of the frequency of ‘inner restlessness’ was reported.24 However, the severity of most NMS, as measured by Visual Analog Scales, was significantly improved; particularly fatigue, and inner restlessness in a subset of patients. The methodological differences in these studies including objectives, measuring tools, and follow-up times may account for the variability of results.
DBS effect on non-motor fluctuations
The NMF accompany motor fluctuations in most cases and are common in PD.25 NMF may exist as NMS symptoms that typically worsen during motor-off periods, that are only present during motor-off periods, or that fluctuate independently from the motor state.25 Witjas et al.26 studied NMF in 40 patients who underwent STN DBS surgery and were followed for 1 year.26 They classified NMF into four main groups: cognitive, psychiatric, autonomic, and sensory fluctuations. The NMF that improved the most after STN DBS were sensory symptoms and pain, with 84.2% improvement. Other NMF which improved were in the dysautonomic and cognitive domains with 60 and 70% decrease in severity. However, psychiatric fluctuations seemed to respond less consistently postoperatively. Ortega-Cubero et al.27 studied 20 patients and reported reduced frequency and severity after 2-year follow-up of both psychiatric and autonomic fluctuations after STN DBS surgery, partially supporting the previous observations.27 These open-label data (see Table 1) implying the benefit of DBS on NMSB and NMF need to be confirmed in controlled studies. These small sample studies probably included subjects with a range of baseline characteristics regarding their NMF that may explain the differing results. For example, pre-surgical NMF response to levodopa therapy, duration, severity, and frequency may influence response to DBS.
DBS effect on cognition
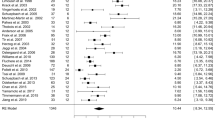
Cognitive decline is far-reaching in PD and seems an inevitable consequence of disease progression as more than 80% of patients develop dementia after 20-year follow-up.28 A large meta-analysis including more than 600 patients showed that DBS of the STN generates a statistically significant small decrease in the executive functions and working memory.29 Similarly, a class I study by Witt et al.30 found mild decrease in executive functions in the STN group when compared with the best medical therapy group (BMT) after a 6-month follow-up.30 However, another subsequent meta-analysis including more than 10 000 patients reported that 57% of the included studies did not find significant cognitive changes after DBS.31 This was corroborated by a large randomized prospective study by Willams et al.32 comparing a BMT group with an STN surgery group during a 1-year follow-up, and showing no differences in cognitive decline.32 Other prospective uncontrolled studies have found none or clinically irrelevant cognitive decline,33–37 while some report a decline in lexical fluency.33,36–38 Two large randomized studies compared the classic surgical targets for PD and showed no difference between the groups.3,39
These variable results may be partly attributed to patient cohort differences with respect to baseline demographic characteristics, PD symptomatology, surgical techniques, and postoperative management. The main risk factors associated with cognitive decline and surgery have been subdivided into (1) preoperative risk factors: impaired attention, higher levodopa equivalent dosages and motor impairment, older age, higher motor, and severe axial symptoms; (2) intra-operative risk factors such as surgical electrode trajectories (through the frontal lobe and head of the caudate nucleus); and (3) postoperative factors including stimulation parameters, electrode location, and medication changes.14,40
In conclusion, there is level I and II type evidence (see Table 2) consistently reporting a moderate decrease in verbal fluency after STN DBS, as well as a mild reduction in abstract reasoning, working memory and executive functions. However, this mild cognitive deterioration is generally not clinically relevant.
Dbs effect on neuropsychiatric symptoms
Depression
Patients with PD may manifest neuropsychiatric symptoms such as depression, anxiety, apathy, impulsiveness, introversion, hopelessness, and suicidal behavior at any stage of the disease. Depression is considered one of the most frequent mood state changes in PD with a prevalence of about 40–50%, with major depression occurring in 5 to 10% of cases.8 Two large randomized controlled trials showed that there was no difference between the STN DBS and BMT groups with respect to depression after a 6-month follow-up (See Table 2).1,2 One randomized study comparing the difference in depressive symptoms between the two classic targets, did not show differences,39 while Follet et al.3 reported worsening of depression after STN DBS when compared with GPi.3 Smaller controlled trials on the immediate effect of DBS41 and prospective case series with up to 11-year follow-up have reported no change in depression,38,42 or mild improvement.37,43 In summary, there is level I evidence demonstrating that DBS is not detrimental for depression, and some class II and III studies suggesting it may improve slightly. Differing results may be due to population differences, methodological variations (i.e., measuring tools, follow-up time) and postoperative motor control and medical treatment changes.
Anxiety
Anxiety is another frequent NMS, usually coexisting with depression and with motor fluctuations, occurring in about 40% of PD patients.8 There is one large randomized controlled study including 156 patients who underwent STN DBS or BMT providing class I data of improvement in anxiety scales at 6-month follow-up after DBS (see Table 2).30 Similarly to depression results, some prospective series suggest that anxiety may benefit from STN DBS.41,43
Impulse control disorders
ICDs are behavioral addictions that include hypersexuality, hyperphagia, pathologic gambling and compulsive shopping. These ICDs affect up to 40% of PD patients with dopamine agonist therapy and approximately 15% of PD patients overall.44 There are conflictive results as far as their response to surgery (see Table 3). One of the recognized causes of ICDs relates to a hyperdopaminergic state, related to dopamine replacement therapy. Various large prospective studies suggest that when ICDs are caused by hyperdopaminergic states, they improve after surgery due to drug reduction.43,45,46 Lhommée et al.43 go as far as proposing that disabling dopaminergic treatment abuse and drug-induced behavioral addictions in PD may be considered a new indication for subthalamic stimulation.43 However, the verdict is still not settled since some cases of de novo impulsivity have been reported after surgery.47–49 In a large retrospective study including 89 patients, Kim et al.49 reported improvement in 13 of 20 patients with ICDs, however nine cases developed de novo ICDs.49 In a survey directed to the Parkinson Study Group centers with the objective of evaluating the prevalence and screening of ICDs pre and post DBS, 67% of the centers observed at least one case of de novo ICD occurrence after surgery.50 These seemingly contradictory results may be due to differing underlying pathophysiological mechanisms. ICDs that are related to a hyperdopaminergic state clearly improve with medication adjustments after DBS. However, in a subset of patients with greater mesolimbic degeneration, DBS itself may produce alterations in the limbic BG-loop and lead to hyperactivation of the ventral striatum and development of ICDs.
Apathy
Apathy is a behavioral disorder characterized by decreased motivation, emotional involvement, and goal-directed and self-driven behaviors. It has been described in about 40% of patients with PD51 and has recently been classified into four subtypes relating to a (1) reward deficiency syndrome, (2) emotional distress, (3) executive dysfunction, and (4) autoactivation deficit.52 Improvement in apathy has been reported after starting dopaminergic therapy and after turning on STN DBS.41 However, worsening of apathy after surgery has also been reported, more than a decade ago,36 and in a more recent prospective study including 63 patients, Thobois et al.51 described how 34 patients developed apathy in the first months (mean 4.7 months, 3.3–8.2 months) post surgery (see Table 3). In this seminal study, the authors related the de novo apathy cases to a rapid withdrawal of dopaminergic drugs after surgery. However, medication changes do not explain the whole story in postsurgical apathy and pre-existing risk factors such as greater dopaminergic mesolimbic degeneration,12 NMF,51 and dyskinesias may have a role.53 Similar to the post-DBS results for ICDs, these seemingly contradictory results probably depend on the subtype of apathy and its pathophysiological basis.
Suicide risk
The increased risk of suicidal behaviors after DBS remains an open-ended question. Attention was drawn to this subject by a large meta-analysis reviewing the first decade of DBS results, which described a completed suicide rate of 0.16–0.32% after DBS.31 This issue was further explored by Voon et al.54 in a multicenter retrospective case series, including 5311 patients, that reported an attempted suicide rate of 0.9% and a completed suicide rate of 0.45% in the first postoperative.54 These alarming results were not corroborated in a later randomized controlled trial by Weintraub and colleagues55 comparing a BMT group including 299 patients and a DBS group (including both STN and GPi targets) of 255 patients.55 However, this trial was possibly underpowered owing to the rarity of suicidal events. The risk factors for suicidal ideation and behaviors proposed by this group included selection bias of personality traits in surgical patients, depression, previous history of ICDs, and unrealistic expectations of surgery.
DBS effect on sleep
PD patients commonly report sleep problems, suffering from insomnia, excessive daytime sleepiness, restless legs syndrome, and REM sleep behavior disorder (RBD).56,57 Several studies have investigated the effect of DBS on sleep dysfunction in PD (see Table 4). Iranzo and colleagues58 were pioneers in showing objective improvement in sleep quality studied by polysomnography after STN DBS. They reported increased continuous sleep time and a decreased arousal index in 11 patients at 6-month follow-up.58 Hjort et al.59 reported improvement of overall sleep quality after STN DBS as they found significant improvement in the mean total Parkinson Disease Sleep Scale after 3 months.59 They did not observe change in nocturia or excessive daytime sleepiness despite medication reduction. In another study in a larger cohort, Amara et al.60 reported subjective sleep quality improvement after unilateral STN DBS as measured by the Pittsburgh Sleep Quality Index.60 Dafsari et al.23 also provide significant evidence of subjective sleep improvement after DBS.23 The long-term benefit of sleep after DBS is supported by an early study by Lyons and Pahwa61 that found consistent improvement for up to 2 years.61 As previously reported, this group did not find change in excessive daytime sleepiness.61 In summary, there is level III type evidence suggesting that STN DBS may improve nocturnal sleep in PD patients, particularly sleep quality. Most authors hypothesize that sleep benefit after DBS could result from improved nocturnal mobility and reduced sleep fragmentation. Current data do not report any reduction in REM sleep behavior disorder or improvement of excessive daytime sleepiness.
DBS effect on pain
There have been several open label studies addressing the issue of pain, a common NMS of PD and its response to DBS, using either STN or the GPi as targets (see Table 4). Loher et al.62 reported on the effect of unilateral and bilateral GPi DBS in 16 patients: evaluations documented at 3 months indicated pain relief, which was sustained at the 12-month follow-up.62 In another study, Kim et al.63 reported a positive outcome of pain after STN DBS that persisted after 2 years, although some patients developed de novo musculoskeletal pain.63 In their recently published study, the IPMDS NMS study group showed that after 6 months of STN DBS, there was significant reduction in pain scores as measured by the NMSS.23 It is worth noting that these studies do not specifically address the classification of pain in PD. On the other hand, Cury et al.64 investigated a cohort of 41 patients and tackled this issue specifically, reporting significant improvement in pain intensity after 1 year of STN DBS.64 The prevalence of pain decreased from 70 to 21% and the most improved types were dystonic and musculoskeletal pain while central and neuropathic pain did not change. This supports previous findings with quantitative sensory testing describing that sensitivity to pain was not influenced by STN DBS and suggesting that DBS may have no direct modulation of central pain processing.65
Pain and DBS studies to date suffer from methodological limitations due to their open label nature and lack of a coherent tool to address the multifactorial nature of pain in PD that is currently possible with the recently validated King’s PD Pain Scale.66 There is only class III evidence supporting the benefit of DBS on pain and future randomized controlled studies should take into account the various types of PD-related pain as they may be influenced differently by DBS.
DBS effect on the autonomic domain
Urinary symptoms
Urinary symptoms are important constituents of autonomic dysfunction and occur in 38–71% of PD patients and are therefore of the most frequent NMS.57 Another review has reported improvement in detrusor hyperreflexia and increased bladder capacity after DBS.7 A recent study reported significant improvement in urinary symptoms from baseline in the urinary domain of the NMSS, which addresses frequency, urgency, and nocturia.23 In other open label studies studying the immediate effects of turning DBS ON, Seif et al.67 found improved bladder function as measured by objective urodynamic parameters67 and Wolz et al.24 found that patients reported a positive direct effect on urinary symptoms with DBS on.24 The underlying physiological mechanism may be related to improved cortical control,68 and improved sensory gating.18 However, these data need to be corroborated in further larger studies as some have reported subjective benefit but no change in urodynamic parameters (see Table 5).69
Gastrointestinal symptoms
Not many studies have specifically looked at the effect of DBS on the gastrointestinal system (see Table 5). The first study to document evidence was conducted by Arai et al.70 who studied 16 PD patients and measured gastric emptying by the excretion of CO2 in the 13C-acetate breath test.70 They reported improvement of gastric emptying after turning DBS on and speculated on a modulatory role of the dysfunctional vagal neural control in PD. The study by Dafsari et al.23 supports the above-mentioned findings of improved gastrointestinal emptying after STN DBS.23 Another study by Zibetti et al.71 reported the positive effects of STN DBS in improving salivation and constipation in a 1- and 3-year follow-up of 36 patients.71 There is therefore class III evidence suggesting some benefit in gastrointestinal symptoms after DBS. Future studies should take into account the complexity of underlying pathological mechanisms. For example, constipation may depend on a central component (mediated by neural loss and LTS in the dorsal motor nucleus of the vagus nerve and spinal cord) and a peripheral component (mediated by a rostral–caudal gastrointestinal gradient of LTS). Patients with more brainstem pathology, may obtain a benefit not seen in others.
Troche et al.72 reviewed nine studies that specifically addressed the effect of STN DBS on dysphagia and found no effect.72 However, in a recent manometric study in 16 PD patients, Derrey et al.73 found significant improvement in esophageal body contractions and enhancement of lower esophageal sphincter opening. Again, dysphagia in PD is a complex symptom that may be caused by alterations in the oral, pharyngeal, or esophageal phases of swallowing. On the basis of a small series of autopsy studies, dysphagia probably depends on LTS pathology of the pharyngeal nerves and localized muscle atrophy10 and thus one would not expect to see an effect by DBS.10 However, some patients may also show abnormal esophageal peristaltic movements, which could be modulated by DBS through the vagal nerve, but whether this translates into clinical benefit remains to be elucidated.74
Orthostatic hypotension
An early study in a small cohort indicated instant improvement of orthostatic hypotension after turning STN DBS on,75 but failed to report any change in cardiovascular control (see Table 5). These findings were supported by a more recent controlled study that reported improved orthostatic hypotension in PD patients after DBS.76 Another controlled study including 28 PD patients proposed that the vagal component of the arterial-cardiac baroreflex improved postoperatively.77 Dafsari et al.23 suggested that STN DBS may have a long-term effect on cardiovascular symptoms, and opine that further studies are needed.23
Hyperhidrosis
There have been contradictory outcomes on the effect of STN DBS on sweating function, where some studies show positive results and others fail to observe differences (see Table 5). Wolz et al.24 found no change after immediate stimulation;24 whereas Dafsari et al.23 found significant improvement in hyperhidrosis at 6-month follow-up.23 In another 6-month study including 19 patients, Trachani et al.17 indicated that there was subjective improvement in sweating, however, no objective reduction in hyperhidrosis was demonstrated.17 These studies were limited by the small number of included subjects, and offer class III evidence that DBS probably does not worsen sweating spells. Contradictory results can partially be explained by differences in baseline characteristics (e.g., sweating spells clearly related with dyskinesias, off periods, or nighttime akinesia may benefit from DBS due to motor improvement), measuring tools (there is no validated tool that takes into account sweating fluctuations, subjective perception, and objective findings) and postsurgical medication changes.
Olfaction
Although odor detection78 and identification79 seems to not be influenced by DBS, odor discrimination seems to be improved as a result of DBS,78 possibly owing to improved cognitive odor information processing.80 In a recent open-label, prospective study including 60 patients with PD, patients experienced a subjective improvement of the ability to smell and taste.23
DBS effect on body weight
Generally, patients with PD lose weight gradually as the disease progresses.81 The effect of DBS on body weight is still a matter under discussion; however, in recent years, many observational studies (see Table 6) have reported that patients gain weight after surgery and this may adversely affect their metabolic status.82 In a retrospective review including 182 patients with a range of movement disorders and DBS targets, Strowd et al.83 described a mean weight gain of about 1 kg per year for 2 years following surgery.83 In a recent retrospective case–control study, this same group reported a mean weight gain of 2.9 kg in the STN DBS group, significantly more than in the control group on BMT, with mean loss of 1.8 kg.84 An early prospective study showed a 4.7 kg. increase in PD patients after surgery at 1-year follow-up.85 Other groups have reported weight gain ranging from 4.3 to 14 kg.86 One study explored this weight gain by gender and found that men gained lean body mass, whereas women gained weight at the expense of fat, but mean weight gain was similar.87 Another study evaluated the difference between STN DBS patients and BMT, showing greater weight gain in the surgical group.88 There are different hypotheses attempting to explain postsurgical weight gain, relating it to dopaminergic therapy modifications, increased food intake related to changes in the hypothalamus,89 decreased energy expenditure by better control of dyskinesias,90 and improvement of motor symptoms.85 In summary, there is Class III evidence suggesting that PD patients gain weight after STN DBS.
Discussion
This review of the current data investigating the impact of DBS on the NMS suggests that some symptoms may improve, others remain unchanged and some worsen, corroborating previous findings.6–8 There is high-quality evidence demonstrating that DBS is generally a safe procedure with regard to cognition and behavioral morbidity. Data support the improvement of anxiety, and possibly also depression and ICDs. The benefit on sleep, dystonic, and musculoskeletal pain, urinary and gastrointestinal symptoms, and weight loss has also been suggested by prospective uncontrolled studies. Overall, cognitive function generally remains stable, while some small studies suggest that REM sleep behavior disorder, excessive daytime sleepiness, and dysphagia also remain unchanged by DBS. On the other hand, after surgery, decreased verbal fluency is consistently reported and apathy possibly worsens, as suggested by prospective series.
In interpreting postsurgical results, the reader must bear in mind the limitations of each type of study (Table 1,2,3,4,5,6). Even in high-quality studies, it is difficult to extrapolate the direct impact of DBS from the possible indirect effects that may be modulated by changes in medication, motor symptom improvement, or management of therapeutic expectations, among many other variables.
Substantial progress has been made in recent years to further our understanding of the NMS. Development of disease-specific patient-reported questionnaires, such as the NMSQuest19 or questionnaire for impulsive-compulsive disorders in Parkinson's disease,91 and disease-specific scales, like the NMSS,20 the REM Sleep Behavior Disorder Severity Scale,92 and King’s PD Pain Scale,66 have provided the necessary tools to measure NMS, thus enabling an increasing number of clinical trials to include specific NMS and NMS as a whole as primary end points. DBS multidisciplinary investigative efforts combining functional neuroimaging, neurophysiology, and clinical data provide a unique opportunity in advancing toward understanding the neurophysiological mechanisms behind NMS. A key point in the future may be to understand how the somatotopy of the BG loops influences various non-motor outcomes. Neuropsychiatric and cognitive results probably depend more highly on cortico-BG circuitry than other non-motor symptoms, such as pain and dysautonomia, that may have a central and peripheral component, as suggested by neuropathologic findings of LTS at both levels.10
Of the classic DBS targets, most of the data pertains to the subthalamic nucleus (STN), with a paucity of studies regarding the GPi, and null investigations specifically looking at the effect of thalamic ventroimtermediate stimulation. There are few comparative studies available for non-motor effects of STN and GPi DBS. Although some data suggest, advantages of GPi DBS on mood and behavior,55,93,94 little is known on other non-motor symptoms such as autonomic dysfunctions, pain or sleep symptoms. Future studies comparing STN and GPi DBS effects should not only focus on the quality of life and motor symptoms but also on a wide range of non-motor symptoms. This requires the utilization of scores covering a wide range of NMS such as the NMS Scale, the NMS Questionnaire, and laboratory-assisted evaluation of NMS with, e.g., cognitive tests such as the verbal fluency for executive dysfunctions, polysomnography for sleep, CO2 excretion measurement for gastrointestinal functions, urometric tests for urinary symptoms, etc.
DBS was initially developed to target medical refractory motor symptoms such as severe dyskinesias and fluctuations. However, we have since learned that NMS in PD may affect patient quality of life to a higher degree than motor symptoms5 and current effective medical treatments are lacking. Recent DBS technology developments have provided directional stimulation systems (Boston Scientific and Medtronic development center Eindhoven), which allow current steering towards specific subareas of anatomic target structures. Case reports95 and smaller scope96 studies pioneering this technology in patients with PD have provided evidence of its clinical usefulness to achieve optimized motor effects and avoid side effects of DBS. On the basis of the anatomy and functional circuitry of the BG, there is a rationale to examine the non-motor effects of DBS in subareas of the target region in a similar experimental design as in these studies on motor effects of directional stimulation.
We advocate that future randomized controlled studies in DBS should (1) include NMS as primary end points, (2) involve large cohorts that can be divided into subtypes,97 and (3) specifically analyze volumes of tissue activated in context of patients’ individual BG anatomy/somatotopy and thus advance towards therapies tailored personally, based on motor and non-motor symptom profile.
References
Deuschl, G. et al. A randomized trial of deep-brain stimulation for Parkinson’s disease. N. Engl. J. Med. 355, 896–908 (2006).
Weaver, F. M. et al. Bilateral deep brain stimulation vs best medical therapy for patients with advanced Parkinson disease: a randomized controlled trial. JAMA 301, 63–73 (2009).
Follett, K. A. et al. Pallidal versus subthalamic deep-brain stimulation for Parkinson’s disease. N. Engl. J. Med. 362, 2077–2091 (2010).
Chaudhuri, K. R. & Schapira, A. H. V. Non-motor symptoms of Parkinson’s disease: dopaminergic pathophysiology and treatment. Lancet Neurol. 8, 464–474 (2009).
Martinez-Martin P., Rodriguez-Blazquez C., Kurtis M. M., Chaudhuri K. R., NMSS Validation Group. The impact of non-motor symptoms on health-related quality of life of patients with Parkinson’s disease. Mov. Disord. 26: 399–406 (2011).
Klingelhoefer, L., Samuel, M., Chaudhuri, K. R. & Ashkan, K. An update of the impact of deep brain stimulation on non motor symptoms in Parkinson’s disease. J. Parkinsons Dis. 4, 289–300 (2014).
Wang, X.-H. et al. Target selection recommendations based on impact of deep brain stimulation surgeries on nonmotor symptoms of Parkinson’s disease. Chin. Med. J. (Engl) 128, 3371–3380 (2015).
Kim, H., Jeon, B. S. & Paek, S. H. Nonmotor symptoms and subthalamic deep brain stimulation in Parkinson’s disease. Mov. Disord. 8, 83–91 (2015).
Lang, A. E. & Lees, A. Management of Parkinson’s disease: an evidence-based review. Mov. Disord. 17, S1–S6 (2002).
Adler, C. H. & Beach, T. G. Neuropathological basis of nonmotor manifestations of Parkinson’s disease. Mov. Disord. 31, 1114–1119 (2016).
Alexander, G. E., DeLong, M. R. & Strick, P. L. Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu. Rev. Neurosci. 9, 357–381 (1986).
Tremblay, L., Worbe, Y., Thobois, S., Sgambato-Faure, V. & Féger, J. Selective dysfunction of basal ganglia subterritories: from movement to behavioral disorders. Mov. Disord. 30, 1155–1170 (2015).
Obeso, J. A. et al. Functional organization of the basal ganglia: therapeutic implications for Parkinson’s disease. Mov. Disord. 23, 548–559 (2008).
Volkmann, J., Daniels, C. & Witt, K. Neuropsychiatric effects of subthalamic neurostimulation in Parkinson disease. Nat. Rev. Neurol. 6, 487–498 (2010).
Castrioto, A., Lhommée, E., Moro, E. & Krack, P. Mood and behavioural effects of subthalamic stimulation in Parkinson’s disease. Lancet Neurol. 13, 287–305 (2014).
Alessandro, S. et al. Non-motor functions in parkinsonian patients implanted in the pedunculopontine nucleus: focus on sleep and cognitive domains. J. Neurol. Sci. 289, 44–48 (2010).
Trachani, E. et al. Effects of subthalamic nucleus deep brain stimulation on sweating function in Parkinson’s disease. Clin. Neurol. Neurosurg. 112, 213–217 (2010).
Herzog, J. et al. Improved sensory gating of urinary bladder afferents in Parkinson’s Disease following subthalamic stimulation. Brain 131, 132–145 (2008).
Chaudhuri, K. R. et al. International multicenter pilot study of the first comprehensive self-completed nonmotor symptoms questionnaire for Parkinson’s disease: the NMSQuest study. Mov. Disord. 21, 916–923 (2006).
Chaudhuri, K. R. et al. The metric properties of a novel non-motor symptoms scale for Parkinson’s disease: results from an international pilot study. Mov. Disord. 22, 1901–1911 (2007).
Reich, M. M. et al. Changes in the non-motor symptom scale in Parkinson’s disease after deep brain stimulation. Basal Ganglia 1, 131–133 (2011).
Nazzaro, J. M., Pahwa, R. & Lyons, K. E. The impact of bilateral subthalamic stimulation on non-motor symptoms of Parkinson’s disease. Parkinsonism Relat. Disord. 17, 606–609 (2011).
Dafsari, H. S. et al. Beneficial effects of bilateral subthalamic stimulation on non-motor symptoms in Parkinson’s disease. Brain Stimul. 9, 78–85 (2016).
Wolz, M. et al. Immediate effects of deep brain stimulation of the subthalamic nucleus on nonmotor symptoms in Parkinson’s disease. Park. Relat. Disord. 18, 994–997 (2012).
Storch, A. et al. Nonmotor fluctuations in Parkinson disease: severity and correlation with motor complications. Neurology 80, 800–809 (2013).
Witjas, T. et al. Effects of chronic subthalamic stimulation on nonmotor fluctuations in Parkinson’s disease. Mov. Disord. 22, 1729–1734 (2007).
Ortega-Cubero, S. et al. Effect of deep brain stimulation of the subthalamic nucleus on non-motor fluctuations in Parkinson’s disease: two-years' follow-up. Parkinsonism Relat. Disord. 19, 543–547 (2013).
Hely, M. A., Reid, W. G. J., Adena, M. A., Halliday, G. M. & Morris, J. G. L. The Sydney multicenter study of Parkinson’s disease: the inevitability of dementia at 20 years. Mov. Disord. 23, 837–844 (2008).
Parsons, T. D., Rogers, S. A., Braaten, A. J., Woods, S. P. & Tröster, A. I. Cognitive sequelae of subthalamic nucleus deep brain stimulation in Parkinson’s disease: a meta-analysis. Lancet Neurol. 5, 578–588 (2006).
Witt, K. et al. Neuropsychological and psychiatric changes after deep brain stimulation for Parkinson’s disease: a randomised, multicentre study. Lancet Neurol. 7, 605–614 (2008).
Appleby, B. S., Duggan, P. S., Regenberg, A. & Rabins, P. V. Psychiatric and neuropsychiatric adverse events associated with deep brain stimulation: a meta-analysis of ten years’ experience. Mov. Disord. 22, 1722–1728 (2007).
Williams, A. et al. Deep brain stimulation plus best medical therapy versus best medical therapy alone for advanced Parkinson’s disease (PD SURG trial): a randomised, open-label trial. Lancet Neurol. 9, 581–591 (2010).
Pillon, B. et al. Neuropsychological changes between ‘off’ and ‘on’ STN or GPi stimulation in Parkinson’s disease. Neurology 55, 411–418 (2000).
Krack, P. et al. Five-year follow-up of bilateral stimulation of the subthalamic nucleus in advanced Parkinson’s disease. N. Engl. J. Med. 349, 1925–1934 (2003).
Daniele, A. et al. Cognitive and behavioural effects of chronic stimulation of the subthalamic nucleus in patients with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 74, 175–182 (2003).
Funkiewiez, A. et al. Long term effects of bilateral subthalamic nucleus stimulation on cognitive function, mood, and behaviour in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 75, 834–839 (2004).
Castelli, L. et al. Chronic deep brain stimulation of the subthalamic nucleus for Parkinson’s disease: effects on cognition, mood, anxiety and personality traits. Eur. Neurol. 55, 136–144 (2006).
Rizzone, M. G. et al. Long-term outcome of subthalamic nucleus DBS in Parkinson’s disease: from the advanced phase towards the late stage of the disease? Parkinsonism Relat. Disord. 20, 376–381 (2014).
Odekerken, V. J. et al. Subthalamic nucleus versus globus pallidus bilateral deep brain stimulation for advanced Parkinson’s disease (NSTAPS study): a randomised controlled trial. Lancet Neurol. 12, 37–44 (2013).
Witt, K. et al. Relation of lead trajectory and electrode position to neuropsychological outcomes of subthalamic neurostimulation in Parkinson’s disease: results from a randomized trial. Brain 136, 2109–2119 (2013).
McDonald, L. M., Page, D., Wilkinson, L. & Jahanshahi, M. Deep brain stimulation of the subthalamic nucleus improves sense of well-being in Parkinson’s disease. Mov. Disord. 27, 372–378 (2012).
Kaiser, I., Kryspin-Exner, I., Brücke, T., Volc, D. & Alesch, F. Long-term effects of STN DBS on mood: psychosocial profiles remain stable in a 3-year follow-up. BMC Neurol. 8, 43 (2008).
Lhommée, E. et al. Subthalamic stimulation in Parkinson’s disease: restoring the balance of motivated behaviours. Brain 135, 1463–1477 (2012).
Rossi, P. J., Gunduz, A. & Okun, M. S. The subthalamic nucleus, limbic function, and impulse control. Neuropsychol. Rev. 25, 398–410 (2015).
Ardouin, C. et al. Pathological gambling in Parkinson’s disease improves on chronic subthalamic nucleus stimulation. Mov. Disord. 21, 1941–1946 (2006).
Shotbolt, P. et al. Relationships between deep brain stimulation and impulse control disorders in Parkinson’s disease, with a literature review. Parkinsonism Relat. Disord. 18, 10–16 (2012).
Demetriades, P., Rickards, H. & Cavanna, A. E. Impulse control disorders following deep brain stimulation of the subthalamic nucleus in Parkinson’s disease: clinical aspects. Parkinsons Dis. 2011, 658415 (2011).
Moum, S. J. et al. Effects of STN and GPi deep brain stimulation on impulse control disorders and dopamine dysregulation syndrome. PLoS One 7, e29768 (2012).
Kim, Y. E. et al. Impulse control and related behaviors after bilateral subthalamic stimulation in patients with Parkinson’s disease. J. Clin. Neurosci. 20, 964–969 (2013).
Hack, N. et al. Impulsive and compulsive behaviors in Parkinson Study Group (PSG) centers performing deep brain stimulation surgery. J. Parkinsons Dis. 4, 591–598 (2014).
Thobois, S. et al. Non-motor dopamine withdrawal syndrome after surgery for Parkinson’s disease: predictors and underlying mesolimbic denervation. Brain 133, 1111–1127 (2010).
Pagonabarraga, J., Kulisevsky, J., Strafella, A. P. & Krack, P. Apathy in Parkinson’s disease: Clinical features, neural substrates, diagnosis, and treatment. Lancet Neurol. 14, 518–531 (2015).
Higuchi, M. et al. Predictors of the emergence of apathy after bilateral stimulation of the subthalamic nucleus in patients with Parkinson’s disease. Neuromodulation 18, 113–117 (2015).
Voon, V. et al. A multicentre study on suicide outcomes following subthalamic stimulation for Parkinson’s disease. Brain 131, 2720–2728 (2008).
Weintraub, D. et al. Suicide ideation and behaviours after STN and GPi DBS surgery for Parkinson’s disease: results from a randomised, controlled trial. J. Neurol. Neurosurg. Psychiatry 84, 1113–1118 (2013).
Chaudhuri, K. R. & Martinez-Martin, P. Clinical assessment of nocturnal disability in Parkinson’s disease: the Parkinson's Disease Sleep Scale. Neurology 63, S17–S20 (2004).
Chaudhuri, K. R. & Healy, D. G. Schapira AHV. Non-motor symptoms of Parkinson’s disease: diagnosis and management. Lancet Neurol. 5, 235–245 (2006).
Iranzo, A., Valldeoriola, F., Santamaría, J., Tolosa, E. & Rumià, J. Sleep symptoms and polysomnographic architecture in advanced Parkinson’s disease after chronic bilateral subthalamic stimulation. J. Neurol. Neurosurg. Psychiatry 72, 661–664 (2002).
Hjort, N., Østergaard, K. & Dupont, E. Improvement of sleep quality in patients with advanced Parkinson’s disease treated with deep brain stimulation of the subthalamic nucleus. Mov. Disord. 19, 196–199 (2004).
Amara, A. W. et al. Unilateral subthalamic nucleus deep brain stimulation improves sleep quality in Parkinson’s disease. Parkinsonism Relat. Disord. 18, 63–68 (2012).
Lyons, K. E. & Pahwa, R. Effects of bilateral subthalamic nucleus stimulation on sleep, daytime sleepiness, and early morning dystonia in patients with Parkinson disease. J. Neurosurg. 104, 502–505 (2006).
Loher, T. J., Burgunder, J.-M., Weber, S., Sommerhalder, R. & Krauss, J. K. Effect of chronic pallidal deep brain stimulation on off period dystonia and sensory symptoms in advanced Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 73, 395–399 (2002).
Kim, H.-J., Jeon, B. S., Lee, J.-Y., Paek, S. H. & Kim, D. G. The benefit of subthalamic deep brain stimulation for pain in Parkinson disease: a 2-year follow-up study. Neurosurgery 70, 18–23 (2012).
Cury, R. G. et al. Effects of deep brain stimulation on pain and other nonmotor symptoms in Parkinson disease. Neurology 83, 1403–1409 (2014).
Gierthmühlen, J. et al. Influence of deep brain stimulation and levodopa on sensory signs in Parkinson’s disease. Mov. Disord. 25, 1195–1202 (2010).
Chaudhuri, K. R. et al. King’s Parkinson's disease pain scale, the first scale for pain in PD: an international validation. Mov. Disord. 30, 1623–1631 (2015).
Seif, C. et al. Effect of subthalamic deep brain stimulation on the function of the urinary bladder. Ann. Neurol. 55, 118–120 (2004).
Herzog, J. et al. Subthalamic stimulation modulates cortical control of urinary bladder in Parkinson’s disease. Brain 129, 3366–3375 (2006).
Winge, K. et al. Lower urinary tract symptoms and bladder control in advanced Parkinson’s disease: effects of deep brain stimulation in the subthalamic nucleus. Mov. Disord. 22, 220–225 (2007).
Arai, E. et al. Subthalamic deep brain stimulation can improve gastric emptying in Parkinson’s disease. Brain 135, 1478–1485 (2012).
Zibetti, M. et al. Motor and nonmotor symptom follow-up in parkinsonian patients after deep brain stimulation of the subthalamic nucleus. Eur. Neurol. 58, 218–223 (2007).
Troche, M. S., Brandimore, A. E., Foote, K. D. & Okun, M. S. Swallowing and deep brain stimulation in Parkinson’s disease: a systematic review. Parkinsonism Relat. Disord. 19, 783–788 (2013).
Derrey, S. et al. Impact of deep brain stimulation on pharyngo-esophageal motility: a randomized cross-over study. Neurogastroenterol. Motil. 27, 1214–1222 (2015).
Suttrup, I. & Warnecke, T. Dysphagia in Parkinson’s disease. Dysphagia 31, 24–32 (2016).
Stemper, B. et al. Deep brain stimulation improves orthostatic regulation of patients with Parkinson disease. Neurology 67, 1781–1785 (2006).
Trachani, E. et al. Heart rate variability in Parkinson’s disease unaffected by deep brain stimulation. Acta Neurol. Scand. 126, 56–61 (2012).
Sumi, K. et al. Effect of subthalamic nucleus deep brain stimulation on the autonomic nervous system in Parkinson’s disease patients assessed by spectral analyses of R-R interval variability and blood pressure variability. Stereotact. Funct. Neurosurg. 90, 248–254 (2012).
Hummel, T., Jahnke, U., Sommer, U., Reichmann, H. & Müller, A. Olfactory function in patients with idiopathic Parkinson’s disease: effects of deep brain stimulation in the subthalamic nucleus. J. Neural Transm. 112, 669–676 (2005).
Fabbri, M. et al. Subthalamic deep brain stimulation effects on odor identification in Parkinson’s disease. Eur. J. Neurol. 22, 207–210 (2015).
Guo, X. et al. Effects of bilateral deep brain stimulation of the subthalamic nucleus on olfactory function in Parkinson’s disease patients. Stereotact. Funct. Neurosurg. 86, 237–244 (2008).
Chen, H., Zhang, S. M., Hernán, M. A., Willett, W. C. & Ascherio, A. Weight loss in Parkinson’s disease. Ann. Neurol. 53, 676–679 (2003).
Rieu, I. et al. Body weight gain and deep brain stimulation. J. Neurol. Sci. 310, 267–270 (2011).
Strowd, R. E. et al. Weight change following deep brain stimulation for movement disorders. J. Neurol. 257, 1293–1297 (2010).
Strowd, R. E. et al. Association between subthalamic nucleus deep brain stimulation and weight gain: results of a case-control study. Clin. Neurol. Neurosurg. 140, 38–42 (2016).
Gironell, A., Pascual-Sedano, B., Otermin, P. & Kulisevsky, J. Weight gain after functional surgery for Parkinsons disease. Neurologia 17, 310–316 (2002).
Foubert-Samier, A. et al. A long-term follow-up of weight changes in subthalamic nucleus stimulated Parkinson’s disease patients. Rev. Neurol. 168, 173–176 (2012).
Montaurier, C. et al. Mechanisms of body weight gain in patients with Parkinson’s disease after subthalamic stimulation. Brain 130, 1808–1818 (2007).
Walker, H. C. et al. Weight changes associated with unilateral STN DBS and advanced PD. Parkinsonism Relat. Disord. 15, 709–711 (2009).
Nirenberg, M. J. & Waters, C. Compulsive eating and weight gain related to dopamine agonist use. Mov. Disord. 21, 524–529 (2006).
Macia, F. et al. Parkinson’s disease patients with bilateral subthalamic deep brain stimulation gain weight. Mov. Disord. 19, 206–212 (2004).
Weintraub, D. et al. Validation of the questionnaire for impulsive-compulsive disorders in Parkinson’s disease. Mov. Disord. 24, 1461–1467 (2009).
Sixel-Döring, F., Schweitzer, M., Mollenhauer, B. & Trenkwalder, C. Intraindividual variability of REM sleep behavior disorder in Parkinson’s disease: a comparative assessment using a new REM sleep behavior disorder severity scale (RBDSS) for clinical routine. J. Clin. Sleep Med. 7, 75–80 (2011).
Okun, M. S. et al. Cognition and mood in Parkinson’s disease in subthalamic nucleus versus globus pallidus interna deep brain stimulation: the COMPARE trial. Ann. Neurol. 65, 586–595 (2009).
Anderson, V. C., Burchiel, K. J., Hogarth, P., Favre, J. & Hammerstad, J. P. Pallidal vs subthalamic nucleus deep brain stimulation in Parkinson disease. Arch. Neurol. 62, 554–560 (2005).
Reker, P., Dembek, T. A., Becker, J., Visser-Vandewalle, V. & Timmermann, L. Directional deep brain stimulation: a case of avoiding dysarthria with bipolar directional current steering. Parkinsonism Relat. Disord. 31, 156–158 (2016).
Steigerwald, F., Müller, L., Johannes, S., Matthies, C. & Volkmann, J. Directional deep brain stimulation of the subthalamic nucleus: a pilot study using a novel neurostimulation device. Mov. Disord. 31, 1240–1243 (2016).
Marras, C. & Chaudhuri, K. R. Nonmotor features of Parkinson’s disease subtypes. Mov. Disord. 31, 1095–1102 (2016).
Author information
Authors and Affiliations
Contributions
M.M.K. performed the bibliographical search and wrote the final draft of the manuscript and tables. T.R. wrote the first draft of the sleep, pain, and dysautonomia sections and tables and contributed to the bibliographic search. L.F.D. wrote the first draft of the cognitive, neuropsychiatric, and weight sections and tables. H.S.D. reviewed the final draft and contributed to the bibliographic search.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kurtis, M., Rajah, T., Delgado, L. et al. The effect of deep brain stimulation on the non-motor symptoms of Parkinson’s disease: a critical review of the current evidence. npj Parkinson's Disease 3, 16024 (2017). https://doi.org/10.1038/npjparkd.2016.24
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/npjparkd.2016.24
This article is cited by
-
Architecture of the subthalamic nucleus
Communications Biology (2024)
-
Advanced therapies in Parkinson’s disease: an individualized approach to their indication
Journal of Neural Transmission (2024)
-
Deep Brain Stimulation in Parkinson Disease: A Meta-analysis of the Long-term Neuropsychological Outcomes
Neuropsychology Review (2023)
-
Exploring the network effects of deep brain stimulation for rapid eye movement sleep behavior disorder in Parkinson’s disease
Acta Neurochirurgica (2023)
-
Efficacy of safinamide as add-on therapy after subthalamic nucleus deep brain stimulation in Parkinson disease
Neurological Sciences (2022)