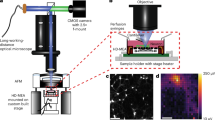
Abstract
Electrical measurements from large populations of animals would help reveal fundamental properties of the nervous system and neurological diseases. Small invertebrates are ideal for these large-scale studies; however, patch-clamp electrophysiology in microscopic animals typically requires invasive dissections and is low-throughput. To overcome these limitations, we present nano-SPEARs: suspended electrodes integrated into a scalable microfluidic device. Using this technology, we have made the first extracellular recordings of body-wall muscle electrophysiology inside an intact roundworm, Caenorhabditis elegans. We can also use nano-SPEARs to record from multiple animals in parallel and even from other species, such as Hydra littoralis. Furthermore, we use nano-SPEARs to establish the first electrophysiological phenotypes for C. elegans models for amyotrophic lateral sclerosis and Parkinson's disease, and show a partial rescue of the Parkinson's phenotype through drug treatment. These results demonstrate that nano-SPEARs provide the core technology for microchips that enable scalable, in vivo studies of neurobiology and neurological diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Davis, R. H. The age of model organisms. Nat. Rev. Genet. 5, 69–76 (2004).
Goodman, M. B., Hall, D. H., Avery, L. & Lockery, S. R. Active current regulate sensitivity and dynamic range in C. elegans neurons. Neuron 20, 763–772 (1998).
Broadie, S. K. & Bate, M. Development of the embryonic neuromuscular synapse of Drosophila melanogaster. J. Neurosci. 13, 144–166 (1993).
Drapeau, P., Ali, D. W., Buss, R. R. & Saint-Amant, L. In vivo recording from identifiable neurons of the locomotor network in the developing zebrafish. J. Neurosci. Methods 88, 1–13 (1999).
Richmond, J. E. & Jorgensen, E. M. One GABA and two acetylcholine receptors function at the C. elegans neuromuscular junction. Nat. Neurosci. 2, 791–797 (1999).
Goodman, M. B., Lindsay, T. H., Lockery, S. R. & Richmond, J. E. Electrophysiological methods for Caenorhabditis elegans neurobiology. Methods Cell Biol. 107, 409–436 (2012).
Liu, P. et al. Genetic dissection of ion currents underlying all-or-none action potentials in C. elegans body-wall muscle cells. J. Physiol. 589, 101–117 (2011).
Liu, P., Chen, B. & Wang, Z.-W. Postsynaptic current bursts instruct action potential firing at a graded synapse. Nat. Commun. 4, 1911 (2013).
Butler, V. J. et al. A consistent muscle activation strategy underlies crawling and swimming in Caenorhabditis elegans. J. R. Soc. Interface 12, 20140963 (2015).
Kerr, R. et al. Optical imaging of calcium transients in neurons and pharyngeal muscle of C. elegans. Neuron 26, 583–594 (2000).
Flytzanis, N. C. et al. Archaerhodopsin variants with enhanced voltage-sensitive fluorescence in mammalian and Caenorhabditis elegans neurons. Nat. Commun. 5, 4894 (2014).
Prevedel, R. et al. Simultaneous whole-animal 3D imaging of neuronal activity using light-field microscopy. Nat. Methods 11, 727–730 (2014).
Larsch, J., Ventimiglia, D., Bargmann, C. I. & Albrecht, D. R. High-throughput imaging of neuronal activity in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 110, E4266–E4273 (2013).
Saul Kato, A. et al. Global brain dynamics embed the motor command sequence of Caenorhabditis elegans. Cell 163, 1–14 (2015).
Larsch, J. et al. A circuit for gradient climbing in C. elegans chemotaxis. Cell Rep. 12, 1748–1760 (2015).
Piggott, B. J., Liu, J., Feng, Z., Wescott, S. A. & Xu, X. Z. S. The neural circuits and synaptic mechanisms underlying motor initiation in C. elegans. Cell 147, 922–933 (2011).
Venkatachalam, V. et al. Pan-neuronal imaging in roaming Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 113, E1082–E1088 (2015).
Nguyen, J. P. et al. Whole-brain calcium imaging with cellular resolution in freely behaving Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 113, E1074–E1081 (2015).
Broussard, G. J., Liang, R. & Tian, L. Monitoring activity in neural circuits with genetically encoded indicators. Front. Mol. Neurosci. 7, 910 (2014).
Raizen, D. M. & Avery, L. Electrical activity and behavior in the pharynx of Caenorhabditis elegans. Neuron 12, 483–495 (1994).
Lockery, S. R. et al. A microfluidic device for whole-animal drug screening using electrophysiological measures in the nematode C. elegans. Lab Chip. 12, 2211–2220 (2012).
Rohde, C. B., Zeng, F., Gonzalez-Rubio, R., Angel, M. & Yanik, M. F. Microfluidic system for on-chip high-throughput whole-animal sorting and screening at subcellular resolution. Proc. Natl Acad. Sci USA 104, 13891–13895 (2007).
Chung, K., Crane, M. M. & Lu, H. Automated on-chip rapid microscopy, phenotyping and sorting of C. elegans. Nat. Methods 5, 637–643 (2008).
Swierczek, N. A., Giles, A. C., Rankin, C. H. & Kerr, R. A. High-throughput behavioral analysis in C. elegans. Nat. Methods 8, 592–598 (2011).
Hai, A., Shappir, J. & Spira, M. E. In-cell recordings by extracellular microelectrodes. Nat. Methods 7, 200–202 (2010).
Tian, B. et al. Three-dimensional, flexible nanoscale field-effect transistors as localized bioprobes. Science 329, 830–834 (2010).
Duan, X. et al. Intracellular recordings of action potentials by an extracellular nanoscale field-effect transistor. Nat. Nanotech. 7, 174–179 (2011).
Robinson, J. T. et al. Vertical nanowire electrode arrays as a scalable platform for intracellular interfacing to neuronal circuits. Nat. Nanotech. 7, 180–184 (2012).
Xie, C., Lin, Z., Hanson, L., Cui, Y. & Cui, B. Intracellular recording of action potentials by nanopillar electroporation. Nat. Nanotech. 7, 185–190 (2012).
Angle, M. R., Cui, B. & Melosh, N. A. Nanotechnology and neurophysiology. Curr. Opin. Neurobiol. 32, 132–140 (2015).
Han, X. & Boyden, E. S. Multiple-color optical activation, silencing, and desynchronization of neural activity, with single-spike temporal resolution. PLoS ONE 2, e299 (2007).
Gao, S. & Zhen, M. Action potentials drive body wall muscle contractions in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 108, 2557–2562 (2011).
Jospin, M., Jacquemond, V., Mariol, M.-C., Ségalat, L. & Allard, B. The L-type voltage-dependent Ca2+ channel EGL-19 controls body wall muscle function in Caenorhabditis elegans. J. Cell Biol. 159, 337–348 (2002).
Wen, Q. et al. Proprioceptive coupling within motor neurons drives C. elegans forward locomotion. Neuron 76, 750–761 (2012).
Westfall, J. A., Yamataka, S. & Enos, P. D. Ultrastructural evidence of polarized synapses in the nerve net of Hydra. J. Cell Biol. 51, 318–323 (1971).
Jorgensen, E. M. & Nonet, M. L. Neuromuscular junctions in the nematode C. elegans. Semin. Dev. Biol. 6, 207–220 (1995).
Liu, Q., Hollopeter, G. & Jorgensen, E. M. Graded synaptic transmission at the Caenorhabditis elegans neuromuscular junction. Proc. Natl Acad. Sci. USA 106, 10823–10828 (2009).
Kaletta, T. & Hengartner, M. O. Finding function in novel targets: C. elegans as a model organism. Nat. Rev. Drug Discov. 5, 387–398 (2006).
Jones, A. K., Buckingham, S. D. & Sattelle, D. B. Chemistry-to-gene screens in Caenorhabditis elegans. Nat. Rev. Drug Discov. 4, 321–330 (2005).
Bargmann, C. I. Neurobiology of the Caenorhabditis elegans genome. Science 282, 2028–2033 (1998).
Lublin, A. L. & Link, C. D. Alzheimer's disease drug discovery: in vivo screening using Caenorhabditis elegans as a model for beta-amyloid peptide-induced toxicity. Drug Discov. Today Technol. 10, e115–e119 (2013).
Sin, O., Michels, H. & Nollen, E. A. A. Genetic screens in Caenorhabditis elegans models for neurodegenerative diseases. Biochim. Biophys. Acta Mol. Basis Dis. 1842, 1951–1959 (2014).
van Ham, T. J. et al. C. elegans model identifies genetic modifiers of α-synuclein inclusion formation during aging. PLoS Genet. 4, e1000027 (2008).
Wang, J. et al. An ALS-linked mutant SOD1 produces a locomotor defect associated with aggregation and synaptic dysfunction when expressed in neurons of Caenorhabditis elegans. PLoS Genet. 5, e1000350 (2009).
Tardiff, D. F., Tucci, M. L., Caldwell, K. A., Caldwell, G. A. & Lindquist, S. Different 8-hydroxyquinolines protect models of TDP-43 protein, alpha-synuclein, and polyglutamine proteotoxicity through distinct mechanisms. J. Biol. Chem. 287, 4107–4120 (2012).
Frankowski, H. et al. Dimethyl sulfoxide and dimethyl formamide increase lifespan of C. elegans in liquid. Mech. Ageing Dev. 134, 69–78 (2013).
Yanik, M. F., Rohde, C. B. & Pardo-Martin, C. Technologies for micromanipulating, imaging, and phenotyping small invertebrates and vertebrates. Annu. Rev. Biomed. Eng. 13, 185–217 (2011).
Cronin, C. J. et al. An automated system for measuring parameters of nematode sinusoidal movement. BMC Genet. 6, 5 (2005).
Madou, M. J. Manufacturing Techniques for Microfabrication and Nanotechnology (CRC Press, 2011).
Lockery, S. R. et al. Artificial dirt: microfluidic substrates for nematode neurobiology and behavior. J. Neurophysiol. 99, 3136–3143 (2008).
Yu, H. et al. Systematic profiling of Caenorhabditis elegans locomotive behaviors reveals additional components in G-protein Gαq signaling. Proc. Natl Acad. Sci. USA 110, 11940–11945 (2013).
Edwards, S. L. et al. A novel molecular solution for ultraviolet light detection in Caenorhabditis elegans. PLoS Biol. 6, 1715–1729 (2008).
Passano, L. M. & McCullough, C. B. The light response and the rhythmic potentials of hydra. Proc. Natl Acad. Sci. USA 48, 1376–1382 (1962).
Acknowledgements
We thank C. Kemere for discussions on extracellular recordings and spectral analysis. Several strains were provided by the Caenorhabditis Genetics Center, which is funded by National Institutes of Health (NIH) Office of Research Infrastructure Programs (P40 OD010440). J. Wang provided the ALS worm strains and A. Fouad from the Fang-Yen Lab provided the YX9 animals. This work is funded by the Defense Advanced Research Projects Agency Young Faculty Award D14AP00049 (J.T.R.), NIH grant DA018341 (W.Z.) and the Hamill Foundation (J.T.R. and W.Z.). D.L.G. is funded by the National Science Foundation (NSF) Graduate Research Fellowship Program 0940902. D.L.G. and K.N.B. are funded by training fellowships from the Keck Center of the Gulf Coast Consortia on the NSF Integrative Graduate Education and Research Traineeship (IGERT): Neuroengineering from Cells to Systems 1250104. We also thank the Rice Shared Equipment Authority and the University of Houston Nanofabrication Facility where devices were fabricated.
Author information
Authors and Affiliations
Contributions
D.L.G. performed and analysed C. elegans experiments. K.N.B. performed and analysed fluorescence microscopy measurements and Hydra recordings. D.L.G., K.N.B. and D.G.V. developed the fabrication process. B.W.A. provided hardware and software support. Z.L. outcrossed shk-1(lf). W.Z. provided support in outcrossing and locomotive phenotyping. J.T.R. directed the research. D.L.G. and J.T.R. co-wrote the paper. All authors read and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1099 kb)
Supplementary Movie 1
Supplementary Movie 1 (MP4 3666 kb)
Supplementary Movie 2
Supplementary Movie 2 (MP4 4361 kb)
Supplementary Movie 3
Supplementary Movie 3 (MP4 4152 kb)
Rights and permissions
About this article
Cite this article
Gonzales, D., Badhiwala, K., Vercosa, D. et al. Scalable electrophysiology in intact small animals with nanoscale suspended electrode arrays. Nature Nanotech 12, 684–691 (2017). https://doi.org/10.1038/nnano.2017.55
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2017.55
This article is cited by
-
Nanotechnology: new opportunities for the development of patch‐clamps
Journal of Nanobiotechnology (2021)
-
An atlas of nano-enabled neural interfaces
Nature Nanotechnology (2019)
-
A microfluidic-induced C. elegans sleep state
Nature Communications (2019)