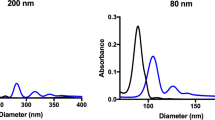
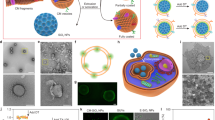
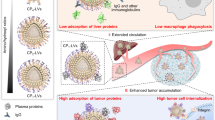
Abstract
The current gold standard to reduce non-specific cellular uptake of drug delivery vehicles is by covalent attachment of poly(ethylene glycol) (PEG). It is thought that PEG can reduce protein adsorption and thereby confer a stealth effect. Here, we show that polystyrene nanocarriers that have been modified with PEG or poly(ethyl ethylene phosphate) (PEEP) and exposed to plasma proteins exhibit a low cellular uptake, whereas those not exposed to plasma proteins show high non-specific uptake. Mass spectrometric analysis revealed that exposed nanocarriers formed a protein corona that contains an abundance of clusterin proteins (also known as apolipoprotein J). When the polymer-modified nanocarriers were incubated with clusterin, non-specific cellular uptake could be reduced. Our results show that in addition to reducing protein adsorption, PEG, and now PEEPs, can affect the composition of the protein corona that forms around nanocarriers, and the presence of distinct proteins is necessary to prevent non-specific cellular uptake.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Tenzer, S. et al. Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology. Nature Nanotech. 8, 772–781 (2013).
Cedervall, T. et al. Understanding the nanoparticle–protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl Acad. Sci. USA 104, 2050–2055 (2007).
Monopoli, M. P., Aberg, C., Salvati, A. & Dawson, K. A. Biomolecular coronas provide the biological identity of nanosized materials. Nature Nanotech. 7, 779–786 (2012).
Aggarwal, P. et al. Nanoparticle interaction with plasma proteins as it relates to particle biodistribution, biocompatibility and therapeutic efficacy. Adv. Drug Deliv. Rev. 61, 428–437 (2009).
Sacchetti, C. et al. Surface polyethylene glycol conformation influences the protein corona of polyethylene glycol-modified single-walled carbon nanotubes: potential implications on biological performance. ACS Nano 7, 1974–1989 (2013).
Veronese, F. M. & Pasut, G. PEGylation, successful approach to drug delivery. Drug Discov. Today 10, 1451–1458 (2005).
Alconcel, S. N. S., Baas, A. S. & Maynard, H. D. FDA-approved poly(ethylene glycol)–protein conjugate drugs. Polym. Chem. 2, 1442–1448 (2011).
Baier, G. et al. Suppressing unspecific cell uptake for targeted delivery using hydroxyethyl starch nanocapsules. Biomacromolecules 13, 2704–2715 (2012).
Landfester, K. & Mailander, V. Nanocapsules with specific targeting and release properties using miniemulsion polymerization. Expert Opin. Drug Deliv. 10, 593–609 (2013).
Worz, A. et al. Protein-resistant polymer surfaces. J. Mater. Chem. 22, 19547–19561 (2012).
Otsuka, H., Nagasaki, Y. & Kataoka, K. PEGylated nanoparticles for biological and pharmaceutical applications. Adv. Drug Deliv. Rev. 64, 246–255 (2012).
Pino, P. d. et al. Protein corona formation around nanoparticles—from the past to the future. Mater. Horiz. 1, 301–313 (2014).
Pelegri-O'Day, E. M., Lin, E. W. & Maynard, H. D. Therapeutic protein–polymer conjugates: advancing beyond PEGylation. J. Am. Chem. Soc. 136, 14323–14332 (2014).
Gref, R. et al. ‘Stealth’ corona–core nanoparticles surface modified by polyethylene glycol (PEG): influences of the corona (PEG chain length and surface density) and of the core composition on phagocytic uptake and plasma protein adsorption. Colloids Surf. B 18, 301–313 (2000).
Kim, H. R. et al. Analysis of plasma protein adsorption onto PEGylated nanoparticles by complementary methods: 2-DE, CE and protein lab-on-chip system. Electrophoresis 28, 2252–2261 (2007).
Ishida, T. et al. Injection of PEGylated liposomes in rats elicits PEG-specific IgM, which is responsible for rapid elimination of a second dose of PEGylated liposomes. J. Control Rel. 112, 15–25 (2006).
Ishida, T. & Kiwada, H. Anti-polyethyleneglycol antibody response to PEGylated substances. Biol. Pharm. Bull. 36, 889–891 (2013).
Shah, S., Prematta, T., Adkinson, N. F. & Ishmael, F. T. Hypersensitivity to polyethylene glycols. J. Clin. Pharmacol. 53, 352–355 (2013).
Bertrand, N. & Leroux, J. C. The journey of a drug-carrier in the body: an anatomo-physiological perspective. J. Control. Rel. 161, 152–163 (2012).
Hamad, I., Hunter, A. C., Szebeni, J. & Moghimi, S. M. Poly(ethylene glycol)s generate complement activation products in human serum through increased alternative pathway turnover and a MASP-2-dependent process. Mol. Immunol. 46, 225–232 (2008).
Amoozgar, Z. & Yeo, Y. Recent advances in stealth coating of nanoparticle drug delivery systems. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 4, 219–233 (2012).
Steinbach, T., Ritz, S. & Wurm, F. R. Water-soluble poly(phosphonate)s via living ring-opening polymerization. ACS Macro Lett. 3, 244–248 (2014).
Marsico, F. et al. Hyperbranched unsaturated polyphosphates as a protective matrix for long-term photon upconversion in air. J. Am. Chem. Soc. 136, 11057–11064 (2014).
Troev, K. D. Polyphosphoesters: Chemistry and Application (Elsevier, 2012).
Steinbach, T. & Wurm, F. R. Poly(phosphoester)s: a new platform for degradable polymers. Angew. Chem. Int. Ed. 54, 6098–6108 (2015).
Zhang, S. Y. et al. Poly(ethylene oxide)-block-polyphosphester-based paclitaxel conjugates as a platform for ultra-high paclitaxel-loaded multifunctional nanoparticles. Chem. Sci. 4, 2122–2126 (2013).
Wang, Y. C. et al. Recent progress in polyphosphoesters: from controlled synthesis to biomedical applications. Macromol. Biosci. 9, 1154–1164 (2009).
Holzapfel, V. et al. Preparation of fluorescent carboxyl and amino functionalized polystyrene particles by miniemulsion polymerization as markers for cells. Macromol. Chem. Phys. 206, 2440–2449 (2005).
Kuga, S. Pore-size distribution analysis of gel substances by size exclusion chromatography. J. Chromatogr. A 206, 449–461 (1981).
Kang, B. et al. Tailoring the stealth properties of biocompatible polysaccharide nanocontainers. Biomaterials 49, 125–134 (2015).
Tenzer, S. et al. Nanoparticle size is a critical physicochemical determinant of the human blood plasma corona: a comprehensive quantitative proteomic analysis. ACS Nano 5, 7155–7167 (2011).
Ritz, S. et al. Protein corona of nanoparticles: distinct proteins regulate the cellular uptake. Biomacromolecules 16, 1311–1321 (2015).
Walkey, C. D. & Chan, W. C. Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem. Soc. Rev. 41, 2780–2799 (2012).
Goppert, T. M. & Muller, R. H. Adsorption kinetics of plasma proteins on solid lipid nanoparticles for drug targeting. Int. J. Pharm. 302, 172–186 (2005).
Blunk, T. et al. Colloidal carriers for intravenous drug targeting: plasma protein adsorption patterns on surface-modified latex particles evaluated by two-dimensional polyacrylamide gel electrophoresis. Electrophoresis 14, 1382–1387 (1993).
Gessner, A. et al. Nanoparticles with decreasing surface hydrophobicities: influence on plasma protein adsorption. Int. J. Pharm. 196, 245–249 (2000).
Vanhooren, V. et al. Protein modification and maintenance systems as biomarkers of ageing. Mech. Ageing Dev. 151, 71–84 (2015).
Koltai, T. Clusterin: a key player in cancer chemoresistance and its inhibition. Oncol. Targets Ther. 7, 447–456 (2014).
Blaschuk, O., Burdzy, K. & Fritz, I. B. Purification and characterization of a cell-aggregating factor (clusterin), the major glycoprotein in ram rete testis fluid. J. Biol. Chem. 258, 7714–7720 (1983).
Nilselid, A. M. et al. Clusterin in cerebrospinal fluid: analysis of carbohydrates and quantification of native and glycosylated forms. Neurochem. Int. 48, 718–728 (2006).
Silajdzic, E., Minthon, L., Bjorkqvist, M. & Hansson, O. No diagnostic value of plasma clusterin in Alzheimer's disease. PLoS ONE 7, e50237 (2012).
Poon, S. et al. Clusterin is an ATP-independent chaperone with very broad substrate specificity that stabilizes stressed proteins in a folding-competent state. Biochemistry 39, 15953–15960 (2000).
Jenne, D. E. & Tschopp, J. Clusterin: the intriguing guises of a widely expressed glycoprotein. Trends Biochem. Sci. 17, 154–159 (1992).
Thiele, L. et al. Competitive adsorption of serum proteins at microparticles affects phagocytosis by dendritic cells. Biomaterials 24, 1409–1418 (2003).
Winzen, S. et al. Complementary analysis of the hard and soft protein corona: sample preparation critically effects corona composition. Nanoscale 7, 2992–3001 (2015).
Rodriguez, P. L. et al. Minimal ‘self’ peptides that inhibit phagocytic clearance and enhance delivery of nanoparticles. Science 339, 971–975 (2013).
Walkey, C. D. et al. Nanoparticle size and surface chemistry determine serum protein adsorption and macrophage uptake. J. Am. Chem. Soc. 134, 2139–2147 (2012).
Barran-Berdon, A. L. et al. Time evolution of nanoparticle–protein corona in human plasma: relevance for targeted drug delivery. Langmuir 29, 6485–6494 (2013).
Zensi, A. et al. Albumin nanoparticles targeted with Apo E enter the CNS by transcytosis and are delivered to neurones. J. Control. Rel. 137, 78–86 (2009).
Kreuter, J. et al. Covalent attachment of apolipoprotein A-I and apolipoprotein B-100 to albumin nanoparticles enables drug transport into the brain. J. Control. Rel. 118, 54–58 (2007).
Acknowledgements
T.S. and F.R.W. acknowledge support from the Max Planck Graduate Center (MPGC). G.B. and T.S. acknowledge support from the Graduate School of Excellence ‘MAINZ’ (Materials Science in Mainz). This work was supported by the DFG/SFB1066 (‘Nanodimensionale polymere Therapeutika für die Tumortherapie’, Q1 and Q2). F.R.W. thanks the DFG WU 750/6-1 for funding.
Author information
Authors and Affiliations
Contributions
F.R.W., V.M. and K.L. supervised the project. F.R.W., V.M., S.S. and G.B. conceived and designed the experiments. G.B. and T.S. synthesized the polymers and nanoparticles. S.S. performed the cell experiments and proteomic analysis. S.W. and K.M. performed the isothermal titration calorimetry measurements. F.R.W. and S.S. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3391 kb)
Rights and permissions
About this article
Cite this article
Schöttler, S., Becker, G., Winzen, S. et al. Protein adsorption is required for stealth effect of poly(ethylene glycol)- and poly(phosphoester)-coated nanocarriers. Nature Nanotech 11, 372–377 (2016). https://doi.org/10.1038/nnano.2015.330
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2015.330
This article is cited by
-
A nanoparticle-based sonodynamic therapy reduces Helicobacter pylori infection in mouse without disrupting gut microbiota
Nature Communications (2024)
-
Cellular uptake and in vivo distribution of mesenchymal-stem-cell-derived extracellular vesicles are protein corona dependent
Nature Nanotechnology (2024)
-
Nano–Bio Interactions: Exploring the Biological Behavior and the Fate of Lipid-Based Gene Delivery Systems
BioDrugs (2024)
-
Transport by circulating myeloid cells drives liposomal accumulation in inflamed synovium
Nature Nanotechnology (2023)
-
Multiomics analysis of naturally efficacious lipid nanoparticle coronas reveals high-density lipoprotein is necessary for their function
Nature Communications (2023)