Abstract
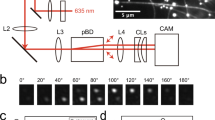
In eukaryotic cells, cargo is transported on self-organized networks of microtubule trackways by kinesin and dynein motor proteins1,2. Synthetic microtubule networks have previously been assembled in vitro3,4,5, and microtubules have been used as shuttles to carry cargoes on lithographically defined tracks consisting of surface-bound kinesin motors6,7. Here, we show that molecular signals can be used to program both the architecture and the operation of a self-organized transport system that is based on kinesin and microtubules and spans three orders of magnitude in length scale. A single motor protein, dimeric kinesin-18, is conjugated to various DNA nanostructures to accomplish different tasks. Instructions encoded into the DNA sequences are used to direct the assembly of a polar array of microtubules and can be used to control the loading, active concentration and unloading of cargo on this track network, or to trigger the disassembly of the network.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Verhey, K. J., Kaul, N. & Soppina, V. Kinesin assembly and movement in cells. Annu. Rev. Biophys. 40, 267–288 (2011).
Goldstein, L. & Yang, Z. Microtubule-based transport systems in neurons: the roles of kinesins and dyneins. Annu. Rev. Neurosci. 23, 39–71 (2000).
Nédélec, F. J., Surrey, T., Maggs, A. C. & Leibler, S. Self-organization of microtubules and motors. Nature 389, 305–308 (1997).
Hess, H. et al. Molecular self-assembly of 'nanowires' and 'nanospools' using active transport. Nano Lett. 5, 629–633 (2005).
Sanchez, T., Welch, D., Nicastro, D. & Dogic, Z. Cilia-like beating of active microtubule bundles. Science 333, 456–459 (2011).
Hiratsuka, Y., Tada, T., Oiwa, K., Kanayama, T. & Uyeda, T. Q. Controlling the direction of kinesin-driven microtubule movements along microlithographic tracks. Biophys. J. 81, 1555–1561 (2001).
Dennis, J. R., Howard, J. & Vogel, V. Molecular shuttles: directed motion of microtubules along nanoscale kinesin tracks. Nanotechnology 10, 232–236 (1999).
Vale, R., Reese, T. & Sheetz, M. Identification of a novel force-generating protein, kinesin, involved in microtubule-based motility. Cell 42, 39–50 (1985).
Goel, A. & Vogel, V. Harnessing biological motors to engineer systems for nanoscale transport and assembly. Nature Nanotech. 3, 465–475 (2008).
Schmidt, C. & Vogel, V. Molecular shuttles powered by motor proteins: loading and unloading stations for nanocargo integrated into one device. Lab Chip. 10, 2195–2198 (2010).
Fischer, T., Agarwal, A. & Hess, H. A smart dust biosensor powered by kinesin motors. Nature Nanotech. 4, 162–166 (2009).
Doot, R. K., Hess, H. & Vogel, V. Engineered networks of oriented microtubule filaments for directed cargo transport. Soft Matter 3, 349–356 (2007).
Idan, O. et al. Nanoscale transport enables active self-assembly of millimeter-scale wires. Nano Lett. 12, 240–245 (2012).
Braun, M., Drummond, D. R., Cross, R. A. & McAinsh, A. D. The kinesin-14 Klp2 organizes microtubules into parallel bundles by an ATP-dependent sorting mechanism. Nature Cell Biol. 11, 724–730 (2009).
Svoboda, K., Schmidt, C. F., Schnapp, B. J. & Block, S. M. Direct observation of kinesin stepping by optical trapping interferometry. Nature 365, 721–727 (1993).
Papworth, M. et al. Inhibition of herpes simplex virus 1 gene expression by designer zinc-finger transcription factors. Proc. Natl Acad. Sci. USA 100, 1621–1626 (2003).
Yajima, J. & Cross, R. A. A torque component in the kinesin-1 power stroke. Nature Cell Biol. 1, 338–341 (2005).
Yurke, B. & Mills, A. P. Using DNA to power nanostructures. Genet. Program. Evolv. Machines 4, 111–122 (2003).
Drew, H. & Wing, R. Structure of a B-DNA dodecamer: conformation and dynamics. Proc. Natl Acad. Sci. USA 78, 2179–2183 (1981).
Surrey, T., Nédélec, F., Leibler, S. & Karsenti, E. Physical properties determining self-organization of motors and microtubules. Science 292, 1167–1171 (2001).
Subramanian, R. & Kapoor, T. M. Building complexity: insights into self-organized assembly of microtubule-based architectures. Dev. Cell 23, 874–885 (2012).
Bouchard, A., Warrender, C. & Osbourn, G. Harnessing microtubule dynamic instability for nanostructure assembly. Phys. Rev. E 74, 1–16 (2006).
Tuma, M. C., Zill, A., Le Bot, N., Vernos, I. & Gelfand, V. Heterotrimeric kinesin II is the microtubule motor protein responsible for pigment dispersion in Xenopus melanophores. J. Cell Biol. 143, 1547–1558 (1998).
Crevel, I. M., Lockhart, A. & Cross, R. A. Weak and strong states of kinesin and ncd. J. Mol. Biol. 257, 66–76 (1996).
Walker, G. M. & Beebe, D. J. A passive pumping method for microfluidic devices. Lab Chip. 2, 131–134 (2002).
Howard, J., Hudspeth, A. & Vale, R. Movement of microtubules by single kinesin molecules. Nature 342, 154–158 (1989).
Kotz, S. et al. Encyclopedia of Statistical Sciences (Wiley, 2006).
Leduc, C. et al. Molecular crowding creates traffic jams of kinesin motors on microtubules. Proc. Natl Acad. Sci. USA 109, 6100–6105 (2012).
Stellwagen, E. & Stellwagen, N. C. Determining the electrophoretic mobility and translational diffusion coefficients of DNA molecules in free solution. Electrophoresis 23, 2794–2803 (2002).
Simmel, F. C. DNA-based assembly lines and nanofactories. Curr. Opin. Biotechnol. 23, 516–520 (2012).
Acknowledgements
The authors thank J. Yajima (Department of Life Sciences, Graduate School of Arts and Sciences, University of Tokyo) for protein design and plasmid production, and M. Alonso, D. R. Drummond and M. Katsuki (Centre for Mechanochemical Cell Biology, Warwick University Medical School) for assistance with protein production and purification. This research was supported by a Engineering and Physical Sciences Research Council grant EP/G037930/1, a Biotechnology and Biological Sciences Research Council grant BB/G019118/1 and a Royal Society–Wolfson Research Merit Award (to A.J.T.).
Author information
Authors and Affiliations
Contributions
Experiments were designed by A.J.M.W. with input from C.S.C., H.M.J.C., R.A.C. and A.J.T. Experiments were performed by A.J.M.W. Data were interpreted and the manuscript written by A.J.M.W. and A.J.T., with assistance from all other authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Information (PDF 2310 kb)
Supplementary movie 1
Supplementary movie 1 (AVI 1759 kb)
Supplementary movie 2
Supplementary movie 2 (AVI 13487 kb)
Supplementary movie 3
Supplementary movie 3 (AVI 4152 kb)
Rights and permissions
About this article
Cite this article
Wollman, A., Sanchez-Cano, C., Carstairs, H. et al. Transport and self-organization across different length scales powered by motor proteins and programmed by DNA. Nature Nanotech 9, 44–47 (2014). https://doi.org/10.1038/nnano.2013.230
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2013.230
This article is cited by
-
Structure and dynamics of an archetypal DNA nanoarchitecture revealed via cryo-EM and molecular dynamics simulations
Nature Communications (2023)
-
A printable active network actuator built from an engineered biomolecular motor
Nature Materials (2021)
-
Programmable Assembly of DNA-protein Hybrid Structures
Chemical Research in Chinese Universities (2020)
-
Molecular dynamics simulation of the nanosecond pulsed electric field effect on kinesin nanomotor
Scientific Reports (2019)
-
Binding to nanopatterned antigens is dominated by the spatial tolerance of antibodies
Nature Nanotechnology (2019)