Abstract
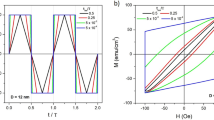
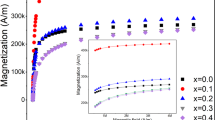
The conversion of electromagnetic energy into heat by nanoparticles has the potential to be a powerful, non-invasive technique for biotechnology applications such as drug release1,2,3, disease treatment4,5,6 and remote control of single cell functions7,8,9, but poor conversion efficiencies have hindered practical applications so far10,11. In this Letter, we demonstrate a significant increase in the efficiency of magnetic thermal induction by nanoparticles. We take advantage of the exchange coupling between a magnetically hard core and magnetically soft shell to tune the magnetic properties of the nanoparticle and maximize the specific loss power, which is a gauge of the conversion efficiency. The optimized core–shell magnetic nanoparticles have specific loss power values that are an order of magnitude larger than conventional iron-oxide nanoparticles. We also perform an antitumour study in mice, and find that the therapeutic efficacy of these nanoparticles is superior to that of a common anticancer drug.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Timko, B. P. et al. Advances in drug delivery. Ann. Rev. Mater. Res. 41, 3.1–3.20 (2011).
Yavuz, M. S. et al. Gold nanocages covered by smart polymers for controlled release with near-infrared light. Nature Mater. 8, 935–939 (2009).
Liu, T. Y., Hub, S. H., Liu, D. M., Chen, S. Y. & Chen, I. W. Biomedical nanoparticle carriers with combined thermal and magnetic responses. Nano Today 4, 52–65 (2009).
Cherukuri, P., Glazer, E. S. & Curley, S. A. Targeted hyperthermia using metal nanoparticles. Adv. Drug. Deliv. Rev. 62, 339–345 (2010).
Kam, N. W. S., O'Connell, M., Wisdom, J. A. & Dai, H. Carbon nanotube as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc. Natl Acad. Sci. USA 102, 11600–11605 (2005).
Thiesen, B. & Jordan, A. Clinical applications of magnetic nanoparticles for hyperthermia. Int. J. Hyperthermia 24, 467–474 (2008).
Huang, H., Delikanli, S., Zeng, H., Ferkey, D. M. & Pralle, A. Remote control of ion channels and neurons through magnetic-field heating of nanoparticles. Nature Nanotech. 5, 602–606 (2010).
Chen, C. C. et al. DNA-gold nanorod conjugates for remote control of localized gene expression by near infrared irradiation. J. Am. Chem. Soc. 128, 3709–3715 (2006).
Jordan, A. et al. Presentation of a new magnetic field therapy system for the treatment of human solid tumors with magnetic fluid hyperthermia. J. Magn. Magn. Mater. 225, 118–126 (2001).
Hergt, R. & Dutz, S. Magnetic particle hyperthermia—biophysical limitations of a visionary tumour therapy. J. Magn. Magn. Mater. 311, 187–191 (2007).
Hergt, R. et al. Physical limits of hyperthermia using magnetite fine particles. IEEE Trans. Magn. 34, 3745–3754 (1998).
Link, S. & El-Sayed, M. A. Shape and size dependence of radiative, non-radiative and photothermal properties of gold nanocrystals. Int. Rev. Phys. Chem. 19, 409–453 (2000).
Lal, S., Clare, S. E. & Halas, N. J. Nanoshell-enabled photothermal cancer therapy: impending clinical impact. Acc. Chem. Res. 41, 1842–1851 (2008).
Norman, R. S., Stone, J. W., Gole, A., Murphy, C. J. & Sabo-Attwood, T. Photothermal destruction of the bacterium Pseudomonas Ariginosa by gold nanorods. Nano Lett. 8, 302–306 (2008).
O'Neal, D. P., Hirsch, L. R., Halas, N. J., Payne, J. D. & West, J. L. Photo-thermal tumor ablation in mice using near infrared-absorbing nanoparticles. Cancer Lett. 209, 171–176 (2004).
Loo, C., Lowery, A., Halas, N., West, J. & Drezek, R. Immunotargeted nanoshells for integrated cancer imaging and therapy. Nano Lett. 5, 709–711 (2005).
Stolik, S., Delgado, J. A., Pérez, A. & Anasagasti, L. Measurement of the penetration depths of red and near infrared light in human ‘ex vivo’ tissues. J. Photochem. Photobiol. B: Biology 57, 90–93 (2000).
Hergt, R., Dutz, S., Müller, R., & Zeisberger, M. Magnetic particle hyperthermia: nanoparticle magnetism and materials development for cancer therapy. J. Phys.: Condens. Matter 18, S2919–S2934 (2006).
Fortin, J-P. et al. Size-sorted anionic iron oxide nanomagnets as colloidal mediators for magnetic hyperthermia. J. Am. Chem. Soc. 129, 2628–2635 (2007).
Rosensweig, R. E. Heating magnetic fluid with alternating magnetic field. J. Magn. Magn. Mater. 252, 370–374 (2002).
Derfus, A. M. et al. Remotely triggered release from magnetic nanoparticles. Adv. Mater. 19, 3932–3936 (2007).
Thomas, C. R. et al. Noninvasive remote-controlled release of drug molecules in vitro using magnetic actuation of mechanized nanoparticles. J. Am. Chem. Soc. 132, 10623–10625 (2010).
Levy, A., Dayan, A., Ben-David, M. & Gannot, I. A new thermography-based approach to early detection of cancer utilizing magnetic nanoparticles theory simulation and in vitro validation. Nanomedicine 6, 786–796 (2010).
Pradhan, P. et al. Comparative evaluation of heating ability and biocompatibility of different ferrite-based magnetic fluids for hyperthermia application. J. Biomed. Mater. Res. B 81, 12–22 (2007).
Habib, A. H., Ondeck, C. L., Chaudhary, P., Bockstaller, M. R. & McHenry, M. E. Evaluation of iron–cobalt/ferrite core shell nanoparticles for cancer thermotherapy. J. Appl. Phys. 103, 07A307 (2008).
Kappiyoor, R., Liangruksa, M., Ganguly, R. & Puri, I. K. The effects of magnetic nanoparticle properties on magnetic fluid hyperthermia. J. Appl. Phys. 108, 094702 (2010).
Kneller, E. F. The exchange-spring magnet: a new material principle for permanent magnets. IEEE Trans. Magn. 27, 3588–3600 (1991).
Zeng, H., Li, J., Liu, J. P., Wang, Z. L. & Sun, S. Exchange-coupled nanocomposite magnets by nanoparticle self-assembly. Nature 420, 395–398 (2002).
Jiang, J. S. et al. A new approach for improving exchange-spring magnets. J. Appl. Phys. 97, 10K311 (2005).
Sun, S. H. et al. Monodisperse MFe2O4 (M=Fe, Co, Mn) nanoparticles. J. Am. Chem. Soc. 126, 273–279 (2004).
Cullity, B. D. Introduction to Magnetic Materials (Addison-Wesley, 1972).
Jang, J-t. et al. Critical enhancements of MRI contrast and hyperthermic effects by dopant-controlled magnetic nanoparticles. Angew. Chem. Int. Ed. 48, 1234–1238 (2009).
Hergt, R. et al. Magnetic properties of bacterial magnetosomes as potential diagnostic and therapeutic tools. J. Magn. Magn. Mater. 293, 80–86 (2005).
Gonzales-Weimuller, G., Zeisberger, M. & Krishnan, K. M. Size-dependent heating rates of iron oxide nanoparticles for magnetic fluid hyperthermia. J. Magn. Magn. Mater. 321, 1947–1950 (2009).
Pankhurst, Q. A., Connolly, J., Jones, S. K. & Dobson, J. Applications of magnetic nanoparticles in biomedicine. J. Phys. D 36, R167–R181 (2003).
Ondeck, C. L. et al. Theory of magnetic fluid heating with an alternating magnetic field with temperature dependent materials properties for self-regulated heating. J. Appl. Phys. 105, 07B324 (2009).
Franco, A. & e Silva, F. C. High temperature magnetic properties of cobalt ferrite nanoparticles, Appl. Phys. Lett. 96, 172505 (2010).
Kulkarni, G. U., Kannan, K. R., Arunarkavalli, T. & Rao, C. N. R. Particle-size effects on the value of Tc of MnFe2O4: evidence for finite-size scaling. Phys. Rev. B 49, 724–727 (1994).
Acknowledgements
This work was supported by Creative Research Initiative (2010-0018286), WCU Program (R32-2009-10217) and BK21 Project. The authors thank H. Nah for preliminary SLP measurements and Y. Jo at KBSI for magnetism measurements. K.I.P. was supported by the Stem Cell Research Center and Korea Healthcare Technology R&D Project (A091159).
Author information
Authors and Affiliations
Contributions
J.C. conceived and designed the experiment. J-H.L, J-t.J., S.H.M., J-G.K. and S-h.N. performed syntheses, characterizations and property measurements of the nanoparticles. J-s.C., I-S.K. and K.I.P. performed in vivo experiments. J-H.L., J-w.K. and J.C. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1601 kb)
Rights and permissions
About this article
Cite this article
Lee, JH., Jang, Jt., Choi, Js. et al. Exchange-coupled magnetic nanoparticles for efficient heat induction. Nature Nanotech 6, 418–422 (2011). https://doi.org/10.1038/nnano.2011.95
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2011.95
This article is cited by
-
Preparation and characterization of β-CD modified magnetic ferrite: the case of doxorubicin
Journal of Materials Science: Materials in Electronics (2024)
-
Scale-up approach for the preparation of magnetic ferrite nanocubes and other shapes with benchmark performance for magnetic hyperthermia applications
Nature Protocols (2023)
-
Folic acid-conjugated dextran-coated Zn0.6Mn0.4Fe2O4 nanoparticles as systemically delivered nano heaters with self-regulating temperature for magnetic hyperthermia therapy of liver tumors
Scientific Reports (2023)
-
Magnetic nanocellulose: influence of structural features on conductivity and magnetic properties
Cellulose (2023)
-
Magnetic, biocompatible CoFe2O4/Fe3O4 core/shell nanoparticles: designing and improved hyperthermia properties
Journal of Nanoparticle Research (2023)