Abstract
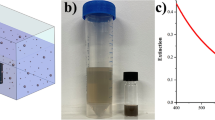
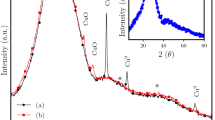
Nanoparticles play an important role in chemical and biological sciences due to their ability to bind and concentrate many molecules on their surface1. Polymers1,2 and silica3,4,5,6,7 are widely used to make nanoparticles, but efforts to make nanoparticles from borosilicate glass—which exhibits high tolerance to chemicals and solvents, combined with excellent mechanical and thermal stability8,9,10—have proved unsuccessful. Here we show that borosilicate nanoparticles (100–500 nm in size) can be synthesized by simply mixing a silicon–boron binary oxide solution, prepared using non-aqueous organic solvents, with water. This induces a vigorous exothermic phase separation in which borosilicate nanoparticles burst out of a silica phase. In addition to potential applications in the life sciences, monodisperse borosilicate particles could also have applications in the production of photonic bandgap devices with high optical contrast, contrast agents for ultrasonic microscopy or chemical filtration membranes11,12,13.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
23 September 2008
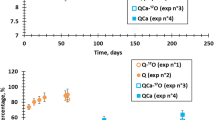
The original version of this letter published online has now been corrected at the end of the abstract, in Fig. 1b, Fig. 5 caption, and in the 'NMR Spectroscopy' section of the Methods. These corrections have been made for all versions of the letter.
References
Kawaguchi, H. Functional polymer microspheres. Prog. Polym. Sci. 25, 1171–1210 (2000).
McCarthy, J. R., Perez, J. M., Bruckner, C. & Weissleder, R. Polymeric nanoparticle preparation that eradicates tumours. Nano Lett. 5, 2552–2556 (2005).
Stöber, W., Fink, A. & Bohn, E. Controlled growth of monodisperse silica spheres in micron size range. J. Colloid Interface Sci. 26, 62–69 (1968).
Barbe, C. et al. Silica particles: A novel drug-delivery system. Adv. Mater. 16, 1959–1966 (2004).
Beck, C., Hartl, W. & Hempelmann, R. Covalent surface functionalization and self-organization of silica nanoparticles. Angew. Chem. Int. Ed. 38, 1297–1300 (1999).
Lu, C. W. et al. Bifunctional magnetic silica nanoparticles for highly efficient human stem cell labelling. Nano Lett. 7, 149–154 (2007).
Iskandar, F., Mikrajuddin & Okuyama, K. In situ production of spherical silica particles containing self-organized mesopores. Nano Lett. 1, 231–234 (2001).
Plodinec, M. J. Borosilicate glasses for nuclear waste immobilization. Glass Technol. 41, 186–192 (2000).
Ehrt, D. Structure, properties and applications of borate glasses. Glass Technol. 41, 182–185 (2000).
Newton, R. G. & Paul, A. A new approach to predicting the durability of glasses from their chemical compositions. Glass Technol. 21, 307–309 (1980).
Sharrna, P., Brown, S., Walter, G., Santra, S. & Moudgil, B. Nanoparticles for bioimaging. Adv. Colloid Interface Sci. 123, 471–485 (2006).
Liu, J. et al. Nanoparticles as image enhancing agents for ultrasonography. Phys. Med. Biol. 51, 2179–2189 (2006).
Muller, M., Zentel, R., Maka, T., Romanov, S. G. & Torres, C. M. S. Dye-containing polymer beads as photonic crystals. Chem. Mater. 12, 2508–2512 (2000).
Benbahouche, S., Roumili, F., Seghir, A. & Zegadi, R. Effect of water on the transmittance of glass plates with eroded surfaces. J. Eur. Ceram. Soc. 26, 1673–1678 (2006).
Zha, C., Atkins, G. R. & Masters, A. F. Preparation and spectroscopy of anhydrous borosilicate sols and their application to thin films. J. Non-Cryst. Solids 242, 63–67 (1998).
Beckett, M. A., Rugen-Hankey, M. P. & Varma, K. S. Formation of borosilicate glasses from silicon alkoxides and metaborate esters in dry non-aqueous solvents. J. Sol-Gel Sci. Technol. 39, 95–101 (2006).
Beckett, M. A., Rugen-Hankey, M. P. & Varma, K. S. Trimethoxyboroxine as an oxygen-transfer reagent: a non-aqueous sol-gel route to alkali-free borosilicate glass. Chem. Commun. 1499–1500 (2000).
Beckett, M. A. et al. [π]-Bonding in B–O ring species: Lewis acidity of Me3B3O3, synthesis of amine Me3B3O3 adducts, and the crystal and molecular structure of Me3B3O3.NH2iBu·MeB(OH)2 . J. Organometall. Chem. 585, 7–11 (1999).
Sharp, K. G. & Scherer, G. W. Interaction of formic acid with the silica gel network. J. Sol-Gel Sci. Technol. 8, 165–171 (1997).
Sauer, H., Brydson, R., Rowley, P. N., Engel, W. & Thomas, J. M. Determination of coordinations and coordination-specific site occupancies by electron energy-loss spectroscopy—an investigation of boron oxygen compounds. Ultramicroscopy 49, 198–209 (1993).
Yang, G., Mobus, G. & Hand, R. J. EELS study of boron coordination in alkali borosilicate glasses under extensive electron irradiation. Phys. Chem. Glasses—Europ. J. Glass Sci. Technol. Part B 47, 507–512 (2006).
Villegas, M. A. & Navarro, J. M. F. Characterization of B2O3–SiO2 glasses prepared via sol-gel. J. Mater. Sci. 23, 2464–2478 (1988).
Wood, D. L. & Rabinovich, E. M. Study of alkoxide silica-gels by infrared-spectroscopy. Appl. Spectrosc. 43, 263–267 (1989).
Tsvetkova, I. N. et al. Sol-gel synthesis and investigation of hybrid organic–inorganic borosilicate nanocomposites. Glass Phys. Chem. 32, 218–227 (2006).
Irwin, A. D., Holmgren, J. S., Zerda, T. W. & Jonas, J. Spectroscopic investigations of borosiloxane bond formation in the sol-gel process. J. Non-Cryst. Solids 89, 191–205 (1987).
Kidd, R. G. in NMR of Newly Accessible Nuclei Vol. 2 (ed. Laszlo, P.) 49–77 (Academic Press, New York, 1983).
Kroeker, S. & Stebbins, J. F. Three-coordinated boron-11 chemical shifts in borates. Inorg. Chem. 40, 6239–6246 (2001).
van Wüllen, L., Müller-Warmuth, W., Papageorgiou, D. & Pentinghaus, H. J. Characterization and structural developments of gel-derived borosilicate glasses: a multinuclear MAS-NMR study. J. Non-Cryst. Solids 171, 53–67 (1994).
Martens, R. & Müller-Warmuth, W. Structural groups and their mixing in borosilicate glasses of various compositions—an NMR study. J. Non-Cryst. Solids 265, 167–175 (2000).
Prasad, S. et al. Solid-state multinuclear magnetic resonance investigation of Pyrex((R)). J. Non-Cryst. Solids 352, 2834–2840 (2006).
Du, L. S. & Stebbins, J. F. Network connectivity in aluminoborosilicate glasses: A high-resolution B-11, Al-27 and O-17 NMR study. J. Non-Cryst. Solids 351, 3508–3520 (2004).
Massiot, D. et al. Modelling one- and two-dimensional solid-state NMR spectra. Magn. Reson. Chem. 40, 70–76 (2002).
Assink, R. A. & Kay, B. D. Study of sol-gel chemical-reaction kinetics by NMR. Annu. Rev. Mater. Sci. 21, 491–513 (1991).
Nanba, T., Nishimura, M. & Miura, Y. A theoretical interpretation of the chemical shift of Si-29 NMR peaks in alkali borosilicate glasses. Geochim. Cosmochim. Acta 68, 5103–5111 (2004).
Parkinson, B. G. et al. Quantitative measurement of Q3 species in silicate and borosilicate glasses using Raman spectroscopy. J. Non-Cryst. Solids 354, 1936–1942 (2008).
Hall, D. G. in Boronic Acids (ed. Hall, D. G.) 1–99 (Wiley-VCH, Weinheim, 2006).
Acknowledgements
The authors acknowledge aid from the Swiss National Science Foundation (grant no. 200020-101466) and thank A.-S. Chauvin, P. Pechy and especially A. Abraham for help with the NMR experiments and Y. Moser for help with the temperature measurements.
Author information
Authors and Affiliations
Contributions
V.K.P., J.-B.O. and A.S. conceived, designed and performed the experiments. V.K.P., J.-B.O., M.C. and M.A.M.G. analysed the data. V.K.P. and M.A.M.G. co-wrote the paper. M.C. was responsible for the EELS analysis.
Corresponding author
Rights and permissions
About this article
Cite this article
Parashar, V., Orhan, JB., Sayah, A. et al. Borosilicate nanoparticles prepared by exothermic phase separation. Nature Nanotech 3, 589–594 (2008). https://doi.org/10.1038/nnano.2008.262
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2008.262
This article is cited by
-
Mesoscale modelling of miscible and immiscible multicomponent fluids
Scientific Reports (2019)
-
Liquid-liquid phase separation of freely falling undercooled ternary Fe-Cu-Sn alloy
Scientific Reports (2015)
-
Synthesis and characterisation of functionalized borosilicate nanoparticles for boron neutron capture therapy applications
Journal of Sol-Gel Science and Technology (2012)