Abstract
In mammals, many daily cycles are driven by a central circadian clock, which is based on the cell-autonomous rhythmic expression of clock genes. It is not clear, however, how peripheral cells are able to interpret the rhythmic signals disseminated from this central oscillator. Here we show that cycling expression of the clock gene Period1 in rodent pituitary cells depends on the heterologous sensitization of the adenosine A2b receptor, which occurs through the nocturnal activation of melatonin mt1 receptors. Eliminating the impact of the neurohormone melatonin simultaneously suppresses the expression of Period1 and evokes an increase in the release of pituitary prolactin. Our findings expose a mechanism by which two convergent signals interact within a temporal dimension to establish high-amplitude, precise and robust cycles of gene expression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Klein, D. C., Moore, R. Y. & Reppert, S. M. (eds.) Suprachiasmatic Nucleus: The Mind's Clock (Oxford Univ. Press, New York, 1991).
Sun, Z. S. et al. RIGUI, a putative mammalian ortholog of the Drosophila period gene. Cell 90, 1003–1011 (1997).
Shigeyoshi, Y. et al. Light-induced resetting of a mammalian circadian clock is associated with rapid induction of the mPer1 transcript. Cell 91, 1043–1053 (1997).
Shearman, L. P. et al. Interacting molecular loops in the mammalian circadian clock. Science 288, 1013–1019 (2000).
Bae, K. et al. Differential functions of mPer1, mPer2, and mPer3 in the SCN circadian clock. Neuron 30, 525–536 (2001).
Reppert, S. M. & Weaver, D. R. Molecular analysis of mammalian circadian rhythms. Annu. Rev. Physiol. 63, 647–676 (2001).
Balsalobre, A. et al. Resetting of circadian time in peripheral tissues by glucocorticoid signalling. Science 289, 2344–2347 (2000).
Balsalobre, A., Damiola, F. & Schibler, U. A serum shock induces circadian gene expression in mammalian tissue culture cells. Cell 93, 929–937 (1998).
Yamazaki, S. et al. Resetting central and peripheral circadian oscillators in transgentic rats. Science 228, 682–685 (2000).
Rajaratnam, S. M. & Arendt, J. Health in a 24-h society. Lancet 358, 999–1005 (2001).
Stokkan, K. A., Yamazaki, S., Tei, H., Sakaki, Y. & Menaker, M. Entrainment of the circadian clock in the liver by feeding. Science 291, 490–493 (2001).
Takekida, S., Yan, L., Maywood, E. S., Hastings, M. H. & Okamura, H. Differential adrenergic regulation of the circadian expression of the clock genes Period1 and Period2 in the rat pineal gland. Eur. J. Neurosci. 12, 4557–4561 (2000).
von Gall, C. et al. Clock gene protein mPER1 is rhythmically synthesized and under cAMP control in the mouse pineal organ. J. Neuroendocrinol. 13, 313–317 (2001).
Stehle, J. H. et al. Adrenergic signals direct rhythmic expression of transcriptional repressor CREM in the pineal gland. Nature 365, 314–320 (1993).
Maronde, E. et al. Transcription factors in neuroendocrine regulation: rhythmic changes in pCREB and ICER levels frame melatonin synthesis. J. Neurosci. 19, 3326–3336 (1999).
von Gall, C. et al. Transcription factor dynamics and neuroendocrine signalling in the mouse pineal gland: a comparative analysis of melatonin-deficient C57BL mice and melatonin-proficient C3H mice. Eur. J. Neurosci. 12, 964–972 (2000).
Korf, H. W., Schomerus, C. & Stehle, J. H. The pineal organ, its hormone melatonin, and the photoneuroendocrine system. Adv. Anat. Embryol. Cell. Biol. 146, 1–100 (1998).
Hastings, M. H. & Follett, B. K. Toward a molecular biological calendar? J. Biol. Rhythms 16, 424–430 (2001).
Morgan, P. J. The pars tuberalis: the missing link in the photoperiodic regulation of prolactin secretion? J. Neuroendocrinol. 12, 287–295 (2000).
Messager, S., Ross, A. W., Barrett, P. & Morgan, P. J. Decoding photoperiodic time through Per1 and ICER gene amplitude. Proc. Natl. Acad. Sci. USA 96, 9938–9943 (1999).
Messager, S., Garabette, M. L., Hastings, M. L. & Hazlerigg, D. G. Tissue-specific abolition of Per1 expression in the pars tuberalis by pinealectomy in the Syrian hamster. NeuroReport 12, 1–4 (2001).
Carlson, L. L., Weaver, D. R. & Reppert, S. M. Melatonin signal transduction in hamster brain: inhibition of adenylyl cyclase by a pertussis toxin–sensitive G protein. Endocrinology 125, 2670–2676 (1989).
Hazlerigg, D. G., Gonzalez-Brito, A., Lawson, W., Hastings, M. H. & Morgan, P. J. Prolonged exposure to melatonin leads to time-dependent sensitization of adenylate cyclase and down-regulates melatonin receptors in pars tuberalis cells form ovine pituitary. Endocrinology 132, 285–292 (1991).
Stehle, J. H. et al. Molecular cloning and expression of the cDNA for a novel A2-adenosine receptor subtype. Mol. Endocrinol. 6, 384–393 (1992).
Stehle, J. H. Pineal gene expression: dawn in a dark matter. J. Pineal Res. 18, 179–190 (1995).
McNulty, S., Ross, A. W., Shiu, K. Y., Morgan, P. J. & Hastings, M. H. Phosphorylation of CREB in ovine pars tuberalis is regulated both by cyclic AMP-dependent and cyclic AMP-independent mechanisms. J. Neuroendocrinol. 8, 635–644 (1996).
Li, X. M. et al. Relationship of atypical melatonin rhythm with two circadian clock outputs in B6D2F(1) mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 278, 924–930 (2000).
Liu, C. et al. Molecular dissection of two distinct actions of melatonin on the suprachiasmatic circadian clock. Neuron 19, 91–102 (1997).
Porkka-Heiskanen, T. et al. Adenosine: a mediator of the sleep-inducing effects of prolonged wakefulness. Science 276, 1265–1268 (1997).
Stirland, J. A. et al. Photoperiodic regulation of prolactin gene expression in the Syrian hamster by a pars tuberalis–derived factor. J. Neuroendocrinol. 13, 147–157 (2001).
Hazlerigg, D. G. What is the role of melatonin within the anterior pituitary? J. Endocrinol. 170, 493–501 (2001).
Lincoln, G. A. & Clarke, I. J. Role of the pituitary gland in the development of photorefractoriness and generation of long-term changes in prolactin secretion in rams. J. Neuroendocrinol. 7, 637–643 (1994).
von Gall, C. et al. CREB in the mouse SCN: a molecular interface coding the phase adjusting stimuli of light, glutamate, PACAP and melatonin for clockwork access. J. Neurosci. 18, 10389–10397 (1998).
von Gall, C., Weaver, D. R., Kock, M., Korf, H.-W. & Stehle, J. H. Melatonin limits transcriptional impact of phosphoCREB in the mouse SCN via the Mel1a receptor. NeuroReport 11, 1803–1807 (2000).
Pfeffer, M., Kühn, R., Krug, L., Korf, H.-W. & Stehle, J. H. Rhythmic variation in beta1-adrenergic receptor mRNA levels in the rat pineal gland: circadian and developmental regulation. Eur. J. Neurosci. 10, 2896–2904 (1998).
Marquardt, D. L., Walker, L. L. & Heinemann, S. Cloning of two adenosine receptor subtypes from mouse bone marrow–derived mast cells. J. Immunol. 152, 4508–4515 (1994).
Acknowledgements
We thank S. M. Reppert for help with generating the melatonin mt1R−/− mice and for the antibody to mPER1; H. Zimmermann for discussion; M. L. Eifländer, C. Illickovic, D. Kärger, I. Schneider-Hüther and S. Schüßler for technical support; and E. Colnago, M. Holub, M. Kock, S. Leslie, S. Schotten and C. Schultz for help. This work was supported by grants from the Deutsche Forschungsgemeinschaft (to H.W.K. and J.H.S.), the Paul und Ursula Klein-Stiftung and the Heinrich und Fritz Riese-Stiftung (to J.H.S.), the Biotechnology and Biological Sciences Research Council and the Medical Research Council (to M.H.H.) and the NIH (AG09975 to D.R.W.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
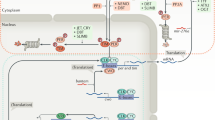
Mechanisms for generating rhythmic gene expression in pituitary cells. At 'night', the convergent impact of adenosine and melatonin leads to acute inhibition of the cAMP signaling pathway in cells of the hypophyseal pars tuberalis, with a simultaneous heterologous sensitization of the adenosine A2b receptor (light green). This intracellular crosstalk is active during the second half of the night, when the secretion of the neurohormone melatonin from the pineal gland is high. With the decline of the inhibitory melatonin signal at 'dawn', adenosine elicits through the sensitized adenosine A2b receptor (dark green) an increased transcription of cAMP-inducible genes through phosphorylation of the activating transcription factor CREB. At 'day', the absence of the melatonin signal results in a desenstization of the adenosine A2b receptor (yellow). As a delayed consequence of the melatonin-sensitized adenosinergic signaling pathway, the translational products of cAMP-sensitive genes, such as PER1, accumulate in pituitary cells. The thickness of arrows indicates the intensity of signaling events. AC, adenylyl cyclase; ado, adenosine; A2b, adenosine 2b receptor; cAMP, cAMP; CRE, camp-responsive element; CREB, CRE-binding protein; G, G protein; P, phosphate group; PKA, protein kinase A; mel, melatonin; mt1R, melatonin mt1 receptor. (PDF 86 kb)
Rights and permissions
About this article
Cite this article
von Gall, C., Garabette, M., Kell, C. et al. Rhythmic gene expression in pituitary depends on heterologous sensitization by the neurohormone melatonin. Nat Neurosci 5, 234–238 (2002). https://doi.org/10.1038/nn806
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn806
This article is cited by
-
Identification of an endocannabinoid system in the rat pars tuberalis—a possible interface in the hypothalamic-pituitary-adrenal system?
Cell and Tissue Research (2017)
-
Melatonin receptor deficiency decreases and temporally shifts ecto-5′-nucleotidase mRNA levels in mouse prosencephalon
Cell and Tissue Research (2016)
-
Influence of the modern light environment on mood
Molecular Psychiatry (2013)
-
Localization of an endocannabinoid system in the hypophysial pars tuberalis and pars distalis of man
Cell and Tissue Research (2010)
-
Cell organization of the rat pars tuberalis. Evidence for open communication between pars tuberalis cells, cerebrospinal fluid and tanycytes
Cell and Tissue Research (2010)