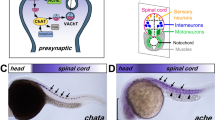
Abstract
The neurotransmitter acetylcholine (ACh) has a crucial role in central and neuromuscular synapses of the cholinergic system. After release into the synaptic cleft, ACh is rapidly degraded by acetylcholinesterase (AChE). We have identified a mutation in the ache gene of the zebrafish, which abolishes ACh hydrolysis in homozygous animals completely. Embryos are initially motile but subsequently develop paralysis. Mutant embryos show defects in muscle fiber formation and innervation, and primary sensory neurons die prematurely. The neuromuscular phenotype in ache mutants is suppressed by a homozygous loss-of-function allele of the α-subunit of the nicotinic acetylcholine receptor (nAChR), indicating that the impairment of neuromuscular development is mediated by activation of nAChR in the mutant. Here we provide genetic evidence for non-classical functions of AChE in vertebrate development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Soreq, H. & Seidman, S. Acetylcholinesterase—new roles for an old actor. Nat. Rev. Neurosci. 2, 294–302 (2001).
Massoulie, J., Pezzementi, L., Bon, S., Krejci, E. & Vallette, F. M. Molecular and cellular biology of cholinesterases. Prog. Neurobiol. 41, 31–91 (1993).
Millard, C. B. & Broomfield, C. A. Anticholinesterases: medical applications of neurochemical principles. J. Neurochem. 64, 1909–1918 (1995).
Parnetti, L. Clinical pharmacokinetics of drugs for Alzheimer's disease. Clin. Pharmacokinet. 29, 110–129 (1995).
Linton, D. M. & Philcox, D. Myasthenia gravis. Dis. Mon. 36, 593–637 (1990).
Bertrand, C. et al. Zebrafish acetylcholinesterase is encoded by a single gene localized on linkage group 7. Gene structure and polymorphism; molecular forms and expression pattern during development. J. Biol. Chem. 276, 464–474 (2001).
Hanneman, E., Trevarrow, B., Metcalfe, W. K., Kimmel, C. B. & Westerfield, M. Segmental pattern of development of the hindbrain and spinal cord of the zebrafish embryo. Development 103, 49–58 (1988).
Hanneman, E. & Westerfield, M. Early expression of acetylcholinesterase activity in functionally distinct neurons of the zebrafish. J. Comp. Neurol. 284, 350–361 (1989).
Layer, P. G. Cholinesterases preceding major tracts in vertebrate neurogenesis. Bioessays 12, 415–420 (1990).
Layer, P. G. & Willbold, E. Novel functions of cholinesterases in development, physiology and disease. Prog. Histochem. Cytochem. 29, 1–94 (1995).
Xie, W. et al. Postnatal developmental delay and supersensitivity to organophosphate in gene-targeted mice lacking acetylcholinesterase. J. Pharmacol. Exp. Ther. 293, 896–902 (2000).
Radic, Z., Pickering, N. A., Vellom, D. C., Camp, S. & Taylor, P. Three distinct domains in the cholinesterase molecule confer selectivity for acetyl- and butyrylcholinesterase inhibitors. Biochemistry 32, 12074–12084 (1993).
Karnovsky, M. J. & Roots, L. A direct colouring thiocholine method for cholinesterases. J. Histochem. Cytochem. 12, 219–221 (1964).
Cousin, X., Hotelier, T., Giles, K., Toutant, J. P. & Chatonnet, A. aCHEdb: the database system for ESTHER, the α/β fold family of proteins and the Cholinesterase gene server. Nucleic Acids Res. 26, 226–228 (1998).
Sussman, J. L. et al. Atomic structure of acetylcholinesterase from Torpedo californica: a prototypic acetylcholine-binding protein. Science 253, 872–879 (1991).
Simon, S., Le Goff, A., Frobert, Y., Grassi, J. & Massoulie, J. The binding sites of inhibitory monoclonal antibodies on acetylcholinesterase. Identification of a novel regulatory site at the putative “back door.” J. Biol. Chem. 274, 27740–27746 (1999).
Westerfield, M., Wegener, J., Jegalian, B. G., DeRobertis, E. M. & Püschel, A. W. Specific activation of mammalian Hox promoters in mosaic transgenic zebrafish. Genes Dev. 6, 591–598 (1992).
Ticho, B. S., Stainier, D. Y., Fishman, M. C. & Breitbart, R. E. Three zebrafish MEF2 genes delineate somitic and cardiac muscle development in wild-type and mutant embryos. Mech. Dev. 59, 205–218 (1996).
Weinberg, E. S. et al. Developmental regulation of zebrafish MyoD in wild-type, no tail and spadetail embryos. Development 122, 271–280 (1996).
Devoto, S. H., Melancon, E., Eisen, J. S. & Westerfield, M. Identification of separate slow and fast muscle precursor cells in vivo, prior to somite formation. Development 122, 3371–3380 (1996).
Miller, J. B., Teal, S. B. & Stockdale, F. E. Evolutionarily conserved sequences of striated muscle myosin heavy chain isoforms. Epitope mapping by cDNA expression. J. Biol. Chem. 264, 13122–13130 (1989).
Sternfeld, M. et al. Acetylcholinesterase enhances neurite growth and synapse development through alternative contributions of its hydrolytic capacity, core protein, and variable C termini. J. Neurosci. 18, 1240–1249 (1998).
Koenigsberger, C., Chiappa, S. & Brimijoin, S. Neurite differentiation is modulated in neuroblastoma cells engineered for altered acetylcholinesterase expression. J. Neurochem. 69, 1389–1397 (1997).
Coronas, V., Durand, M., Chabot, J. G., Jourdan, F. & Quirion, R. Acetylcholine induces neuritic outgrowth in rat primary olfactory bulb cultures. Neuroscience 98, 213–219 (2000).
Trevarrow, B., Marks, D. L. & Kimmel, C. B. Organization of hindbrain segments in the zebrafish embryo. Neuron 4, 669–679 (1990).
Myers, P. Z., Eisen, J. S. & Westerfield, M. Development and axonal outgrowth of identified motoneurons in the zebrafish. J. Neurosci. 6, 2278–2289 (1986).
Eisen, J. S., Myers, P. Z. & Westerfield, M. Pathway selection by growth cones of identified motoneurones in live zebra fish embryos. Nature 320, 269–271 (1986).
Westerfield, M., Liu, D. W., Kimmel, C. B. & Walker, C. Pathfinding and synapse formation in a zebrafish mutant lacking functional acetylcholine receptor. Neuron 4, 867–874 (1990).
Hensey, C. & Gautier, J. A developmental timer that regulates apoptosis at the onset of gastrulation. Mech. Dev. 69, 183–195 (1997).
Williams, J. A. et al. Programmed cell death in zebrafish rohon beard neurons is influenced by TrkC1/NT-3 signaling. Dev. Biol. 226, 220–230 (2000).
Sepich, D. S., Wegner, J., O' Shea, S. & Westerfield, M. An altered intron inhibits synthesis of the acetylcholine receptor α-subunit in the paralyzed zebrafish mutant nic1. Genetics 148, 361–372 (1998).
Nasevicius, A. & Ekker, S. C. Effective targeted gene 'knockdown' in zebrafish. Nat. Genet. 26, 216–220 (2000).
Silver, A. The Biology of Cholinesterases (North Holland, Amsterdam, 1974).
Tessier-Lavigne, M. Axon guidance by diffusible repellants and attractants. Curr. Opin. Genet. Dev. 4, 596–601 (1994).
Liu, D. W. & Westerfield, M. Clustering of muscle acetylcholine receptors requires motoneurons in live embryos, but not in cell culture. J. Neurosci. 12, 1859–1866 (1992).
Lin, W. et al. Distinct roles of nerve and muscle in postsynaptic differentiation of the neuromuscular synapse. Nature 410, 1057–1064 (2001).
Laskowski, M. B., Olson, W. H. & Dettbarn, W. D. Initial ultrastructural abnormalities at the motor end plate produced by a cholinesterase inhibitor. Exp. Neurol. 57, 13–33 (1977).
Leonard, J. P. & Salpeter, M. M. Agonist-induced myopathy at the neuromuscular junction is mediated by calcium. J. Cell Biol. 82, 811–819 (1979).
Wecker, L., Mrak, R. E. & Dettbarn, W. D. Evidence of necrosis in human intercostal muscle following inhalation of an organophosphate insecticide. Fundam. Appl. Toxicol. 6, 172–174 (1986).
Feng, G. et al. Genetic analysis of collagen Q: roles in acetylcholinesterase and butyrylcholinesterase assembly and in synaptic structure and function. J. Cell Biol. 144, 1349–1360 (1999).
Buonanno, A. & Fields, R. D. Gene regulation by patterned electrical activity during neural and skeletal muscle development. Curr. Opin. Neurobiol. 9, 110–120 (1999).
Westerfield, M. The Zebra Fish Book (University of Oregon Press, Eugene, 1993).
Solnica-Krezel, L., Schier, A. & Driever, W. Efficient recovery of ENU-induced mutations from the zebrafish germline. Genetics 136, 1401–1420 (1994).
Mullins, M. C., Hammerschmidt, M., Haffter, P. & Nüsslein-Volhard, C. Large-scale mutagenesis in the zebrafish: in search of genes controlling development in a vertebrate. Curr. Biol. 4, 189–202 (1994).
Sambrock, J. & Russel, D. W. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Press, New York, 2001).
Müller, F. et al. Intronic enhancers control expression of zebrafish sonic hedgehog in floor plate and notochord. Development 126, 2103–2116 (1999).
Oxtoby, E. & Jowett, T. Cloning of the zebrafish krox-20 gene (krx-20) and its expression during hindbrain development. Nucl. Acids Res. 21, 1087–1095 (1993).
Crow, M. T. & Stockdale, F. E. Myosin expression and specialization among the earliest muscle fibers of the developing avian limb. Dev. Biol. 113, 238–254 (1986).
Piperno, G. & Fuller, M. T. Monoclonal antibodies specific for an acetylated form of α-tubulin recognize the antigen in cilia and flagella from a variety of organisms. J. Cell Biol. 101, 2085–2094 (1985).
Ellman, G. L., Courtney, K. D., Andres, V. & Featherstone, R. M. A new and rapid colorimetric dtermination of acetylcholinesterase activity. Biochem. Pharmacol. 7, 88–95 (1961).
Acknowledgements
We thank V. Korzh, H.G. Frohnhöfer, B. Ticho, J. Grassi, E. Krejci, H. Blau, B. Trevarrow, M. Westerfield and the Developmental Studies Hybridoma Bank (University of Iowa) for mutants and materials. We thank A. Gansmüller and M. Digelmann for help with electron microscopy, and P. Blader, F. Müller and M. König for critically reading the manuscript. We also thank A. Karmin and O. Nkundwa, for care of the fish and to D. Hentsch, M. Bogelin for help with the confocal microscope. We thank N. Fischer for technical assistance and INRA, INSERM, CNRS, HUS, AFM, ARC and ACI for support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Animations.
3D reconstructions were calculated from confocal optical sections of a wild-type (wt), mutant ache -/- (ache) and ache morpholino injected (Mo-ache) embryo. The sections cover half of the width of the embryos, at the level of the hindgut extension. Bound znp1 antibody was revealed with a secondary coupled to cy3, staining the motor neurons and the neural tube (red). The a-bungarotoxin coupled to alexa green, visualizes the AChRs in somites (green). The overlay of the two shows the distribution of the neuromuscular junctions. Animation 4 is a explanatory scheme, showing the projections of the three primary motor neurons (RoP, MiP and CaP) in wild-type (red) compared to ache-/- and MO-ache injected embryos (blue). The projections of RoP projecting along the horizontal myoseptum are not affected by the mutation at 27hpf. The MiP does not project as far caudally in the mutant as in the wild-type somite. The CaP projecting ventrally, runs more rostrally in the mutant when compared to the wild type. The arc of the motor innervation by MiP, and CaP covering the entire somite in the wild type is significantly shallower in the mutant and in the Mo-ache injected embryos. (MPG 331 kb)
Supplementary Figure 1.
Computer model of the active site in the wild-type (a) and mutant AChE (b). A potential hydrogen bond between Ser226 and Glu327, which is one of the three amino acids directly involved in hydrolysis of ACh, may be disrupted by the Asn in the mutant. (JPG 133 kb)
Supplementary Figure 2.
Electron micrographs of wild-type and ache mutant muscle fibers at 27 hpf. The ultrastructure of differentiating muscle fibers is the same in mutant and wild-type embryos. In contrast to older stages, necrosis cannot be detected in the mutant muscle at this stage. Sections are close to the medial midline and thus represent fast muscle fibers. (JPG 110 kb)
Supplementary Figure 3.
Slow muscle cells express less slow muscle myosin, form striated muscle fibrils and are positioned correctly at the surface of the somite in ache mutants. Wild-type (a, c, e) and ache mutant somites (b, d, f) stained with F59 antibody. F59 staining of slow muscle cells is reduced but not completely abolished in the mutant. Furthermore, as seen in cross sections (right panels in a, c), the slow muscle cells reside at the periphery of the somite (arrowhead). (d, e) DAPI staining of sections to outline the somite and the position of the F59 staining in (c) and (d). Thus, migration of slow muscle cells does not appear to be affected by the mutation. Furthermore, high-power magnification reveals striated muscle fibrils in this superficial muscle layer (e, f). The fibrils in the mutant are as abundant as in wild type. Note the detection system of the confocal microscope was twice and 4 times more sensitive in (b) and (d), respectively. Lateral views and cross section in (a) and (b) are projections of stacks of confocal sections collected through half the embryo. Scale bars, 30 mm (a-d), 2 mm (e, f). (JPG 82 kb)
Supplementary Figure 4.
Ubiquitous alexa-bungarotoxin staining in ache mutant somites is specific. Wild-type (wt) and ache mutant embryos were stained with alexa conjugated bungarotoxin alone (1/0) or with increasing concentrations of unlabeled bungarotoxin (1/0.2), (1/0.4), (1/0.6). Equal molar ratios (1/1) abolished both staining of synapses and aberrant staining throughout the mutant somite. (JPG 74 kb)
Supplementary Figure 5.
Cap and MiP motor axons of achesb55 mutants and MO-ache injected embryos cover a smaller area of the somite. The width of area covered by the CaP and MiP axons on the somite (hindgut area of 27 h old embryos) was measured in wild-type (wt, 30 somites, 10 embryos), achesb55 mutants (ache, 30 somites, 10 embryos), control morpholino-injected embryos (co, 24 somites, 8 embryos) and embryos injected with MO-ache (27 somites, 9 embryos). Bars, averages in mm; standard deviations are indicated. (JPG 40 kb)
Supplementary Figure 6.
Quantification of length of RB dendrites in wild-type (wt) and ache mutant embryos and embryos that were exposed to 10 mM tetrodotoxin (TTX). Dendritic extensions were grouped into three categories longer than 120 mm, between 120 and 60 mm and below 60 mm. Length of dendrites was measured in the posterior trunk over the hindgut extension and in the tail of 27 hpf embryos. Embryos were bathed in 10 mM TTX from the 16 somite stage onwards. This treatment abolished motility completely but was not toxic, as wild-type embryos recovered motility after removal of TTX even if they had been treated until 48 hpf. Number of long dendrites (longer than 120 mm) is reduced by approximately twofold in the ache mutant. The frequency of intermediate length dendrites (between 120 and 60 mm) were reduced only slightly. Short dendrites (below 60 mm) are not present in 27 hpf wild-type embryos at all but are very abundant in the ache mutant. TTX did not suppress the ache phenotype, and dendrite extensions in wild-type embryos were not affected by the mutation. Bars, average from 4 wild-type, 4 ache mutant 15 TTX-treated wild-type and 5 TTX-treated ache mutants; standard deviations of measurements are indicated. (JPG 54 kb)
Supplementary Figure 7.
Quantification of apoptosis in Rohon Beard (RB) cells in wild-type, ache mutant and MO-ache injected embryos. Each bar represents the average number of TUNEL-positive RB cells per somite. The average is calculated from at least 19 somites per embryo and n = 10 for wild-type and ache mutant, respectively, and n = 4 for MO-ache injected and control, respectively. The standard deviation is indicated. (JPG 44 kb)
Supplementary Figure 8.
Double staining to confirm identity of TUNEL-positive cells at the dorsal neural tube. Dorsal view of ache mutant embryo stained with the TUNEL procedure (blue black nuclei) to reveal dying RB cells. Embryos were immunohistochemically (brown) re-stained with the antibody Zn12, which recognizes the HNK1/L2 epitope on RB cells. Arrowheads indicate TUNEL-positive/Zn12-positive nuclei, arrows point at TUNEL-negative, Zn12-positive RB cells and asterisks indicate TUNEL-positive and Zn12-negative cells. RB cells are recognized by their large cell bodies (light brown staining). (JPG 120 kb)
Rights and permissions
About this article
Cite this article
Behra, M., Cousin, X., Bertrand, C. et al. Acetylcholinesterase is required for neuronal and muscular development in the zebrafish embryo. Nat Neurosci 5, 111–118 (2002). https://doi.org/10.1038/nn788
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn788
This article is cited by
-
Elucidating the toxicity mechanisms of organophosphate esters by adverse outcome pathway network
Archives of Toxicology (2024)
-
Enzymatic reaction modulated DNA assembly on graphitic carbon nitride nanosheets for sensitive fluorescence detection of acetylcholinesterase activity and inhibition
Microchimica Acta (2023)
-
The sensitivity of the zebrafish embryo coiling assay for the detection of neurotoxicity by compounds with diverse modes of action
Environmental Science and Pollution Research (2023)
-
Benzophenone-3 does not Cause Oxidative Stress or B-esterase Inhibition During Embryo Development of Octopus maya (Voss and Solís Ramírez, 1966)
Bulletin of Environmental Contamination and Toxicology (2023)
-
Prolonged electrolysis injures the neural development of zebrafish (Danio rerio)
Environmental Science and Pollution Research (2022)