Abstract
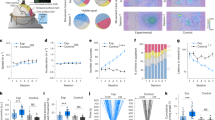
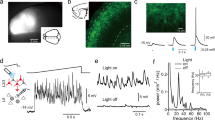
Cortical layering is a hallmark of the mammalian neocortex and a major determinant of local synaptic circuit organization in sensory systems. In motor cortex, the laminar organization of cortical circuits has not been resolved, although their input-output operations are crucial for motor control. Here, we developed a general approach for estimating layer-specific connectivity in cortical circuits and applied it to mouse motor cortex. From these data we computed a laminar presynaptic → postsynaptic connectivity matrix, Wpost,pre, revealing a complement of stereotypic pathways dominated by layer 2 outflow to deeper layers. Network modeling predicted, and experiments with disinhibited slices confirmed, that stimuli targeting upper, but not lower, cortical layers effectively evoked network-wide events. Thus, in motor cortex, descending excitation from a preamplifier-like network of upper-layer neurons drives output neurons in lower layers. Our analysis provides a quantitative wiring-diagram framework for further investigation of the excitatory networks mediating cortical mechanisms of motor control.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Wise, S.P. & Donoghue, J.P. Motor cortex of rodents. in Cerebral Cortex: Sensory-Motor Areas and Aspects of Cortical Connectivity (eds. Jones, E. G. & Peters, A.) 243–270 (Plenum, New York, 1986).
Keller, A. Intrinsic synaptic organization of the motor cortex. Cereb. Cortex 3, 430–441 (1993).
Schieber, M.H. Constraints on somatotopic organization in the primary motor cortex. J. Neurophysiol. 86, 2125–2143 (2001).
Callaway, E.M. Local circuits in primary visual cortex of the macaque monkey. Annu. Rev. Neurosci. 21, 47–74 (1998).
Douglas, R.J. & Martin, K.A. Neuronal circuits of the neocortex. Annu. Rev. Neurosci. 27, 419–451 (2004).
Silberberg, G., Grillner, S., LeBeau, F.E., Maex, R. & Markram, H. Synaptic pathways in neural microcircuits. Trends Neurosci. 28, 541–551 (2005).
Alloway, K.D. Information processing streams in rodent barrel cortex: the differential functions of barrel and septal circuits. Cereb. Cortex published online, doi:10.1093/cercor/bhm138 (16 August 2007).
Lübke, J. & Feldmeyer, D. Excitatory signal flow and connectivity in a cortical column: focus on barrel cortex. Brain Struct. Funct. 212, 3–17 (2007).
Schubert, D., Kötter, R. & Staiger, J.F. Mapping functional connectivity in barrel-related columns reveals layer- and cell type–specific microcircuits. Brain Struct. Funct. 212, 107–119 (2007).
Thomson, A.M. & Lamy, C. Functional maps of neocortical local circuitry. Front. Neurosci. 1, 19–42 (2007).
Petersen, C.C. The functional organization of the barrel cortex. Neuron 56, 339–355 (2007).
Shepherd, G.M.G. & Svoboda, K. Laminar and columnar organization of ascending excitatory projections to layer 2/3 pyramidal neurons in rat barrel cortex. J. Neurosci. 25, 5670–5679 (2005).
Bureau, I., von Saint Paul, F. & Svoboda, K. Interdigitated paralemniscal and lemniscal pathways in the mouse barrel cortex. PLoS Biol. 4, e382 (2006).
Kaneko, T., Caria, M.A. & Asanuma, H. Information processing within the motor cortex. II. Intracortical connections between neurons receiving somatosensory cortical input and motor output neurons of the cortex. J. Comp. Neurol. 345, 172–184 (1994).
Kaneko, T., Cho, R., Li, Y., Nomura, S. & Mizuno, N. Predominant information transfer from layer III pyramidal neurons to corticospinal neurons. J. Comp. Neurol. 423, 52–65 (2000).
Caviness, V.S., Jr. Architectonic map of neocortex of the normal mouse. J. Comp. Neurol. 164, 247–263 (1975).
Brecht, M. et al. Organization of rat vibrissa motor cortex and adjacent areas according to cytoarchitectonics, microstimulation, and intracellular stimulation of identified cells. J. Comp. Neurol. 479, 360–373 (2004).
Song, S., Sjostrom, P.J., Reigl, M., Nelson, S. & Chklovskii, D.B. Highly nonrandom features of synaptic connectivity in local cortical circuits. PLoS Biol. 3, e1 (2005).
Dantzker, J.L. & Callaway, E.M. Laminar sources of synaptic input to cortical inhibitory interneurons and pyramidal neurons. Nat. Neurosci. 3, 701–707 (2000).
Shepherd, G.M.G., Stepanyants, A., Bureau, I., Chklovskii, D.B. & Svoboda, K. Geometric and functional organization of cortical circuits. Nat. Neurosci. 8, 782–790 (2005).
Lehky, S.R. & Sejnowski, T.J. Network model of shape-from-shading: neural function arises from both receptive and projective fields. Nature 333, 452–454 (1988).
Callaway, E.M. Feedforward, feedback and inhibitory connections in primate visual cortex. Neural Netw. 17, 625–632 (2004).
Connors, B.W. Initiation of synchronized neuronal bursting in neocortex. Nature 310, 685–687 (1984).
Chagnac-Amitai, Y. & Connors, B.W. Synchronized excitation and inhibition driven by intrinsically bursting neurons in neocortex. J. Neurophysiol. 62, 1149–1162 (1989).
Crick, F. & Koch, C. Constraints on cortical and thalamic projections: the no-strong-loops hypothesis. Nature 391, 245–250 (1998).
Strick, P.L. & Sterling, P. Synaptic termination of afferents from the ventrolateral nucleus of the thalamus in the cat motor cortex. A light and electron microscopy study. J. Comp. Neurol. 153, 77–106 (1974).
Jones, E.G. Lamination and differential distribution of thalamic afferents within the sensory-motor cortex of the squirrel monkey. J. Comp. Neurol. 160, 167–203 (1975).
Nelson, S. Cortical microcircuits: diverse or canonical? Neuron 36, 19–27 (2002).
Fregnac, Y. et al. The interface between neurons and global brain function. in Microcircuits (eds. Grillner, S. & Graybiel, A. M.) 393–433 (MIT Press, Cambridge, Massachusetts, 2006).
Ohki, K. & Reid, R.C. Specificity and randomness in the visual cortex. Curr. Opin. Neurobiol. 17, 401–407 (2007).
Binzegger, T., Douglas, R.J. & Martin, K.A. A quantitative map of the circuit of cat primary visual cortex. J. Neurosci. 24, 8441–8453 (2004).
Stepanyants, A. et al. Local potential connectivity in cat primary visual cortex. Cereb. Cortex 18, 13–28 (2008).
Lübke, J., Roth, A., Feldmeyer, D. & Sakmann, B. Morphometric analysis of the columnar innervation domain of neurons connecting layer 4 and layer 2/3 of juvenile rat barrel cortex. Cereb. Cortex 13, 1051–1063 (2003).
Helmstaedter, M., de Kock, C.P., Feldmeyer, D., Bruno, R.M. & Sakmann, B. Reconstruction of an average cortical column in silico. Brain Res. Rev. 55, 193–203 (2007).
Braitenberg, V.B. & Schüz, A. Cortex: Statistics and Geometry of Neuronal Connectivity (Springer, Berlin, 1998).
Stepanyants, A. & Chklovskii, D.B. Neurogeometry and potential synaptic connectivity. Trends Neurosci. 28, 387–394 (2005).
Armstrong-James, M., Fox, K. & Das-Gupta, A. Flow of excitation within rat barrel cortex on striking a single vibrissa. J. Neurophysiol. 68, 1345–1358 (1992).
Schubert, D., Kötter, R., Zilles, K., Luhmann, H.J. & Staiger, J.F. Cell type–specific circuits of cortical layer IV spiny neurons. J. Neurosci. 23, 2961–2970 (2003).
Kaneko, T., Kang, Y. & Mizuno, N. Glutaminase-positive and glutaminase-negative pyramidal cells in layer VI of the primary motor and somatosensory cortices: a combined analysis by intracellular staining and immunocytochemistry in the rat. J. Neurosci. 15, 8362–8377 (1995).
Hirsch, J.A. Synaptic integration in layer 4 of the ferret striate cortex. J. Physiol. (Lond.) 483, 183–199 (1995).
Zhang, Z.W. & Deschenes, M. Intracortical axonal projections of lamina VI cells of the primary somatosensory cortex in the rat: a single-cell labeling study. J. Neurosci. 17, 6365–6379 (1997).
Tarczy-Hornoch, K., Martin, K.A., Stratford, K.J. & Jack, J.J. Intracortical excitation of spiny neurons in layer 4 of cat striate cortex in vitro. Cereb. Cortex 9, 833–843 (1999).
Briggs, F. & Callaway, E.M. Laminar patterns of local excitatory input to layer 5 neurons in macaque primary visual cortex. Cereb. Cortex 15, 479–488 (2005).
Petreanu, L., Huber, D., Sobczyk, A. & Svoboda, K. Channelrhodopsin 2–assisted circuit mapping of long-range callosal projections. Nat. Neurosci. 10, 663–668 (2007).
Schubert, D. et al. Layer-specific intracolumnar and transcolumnar functional connectivity of layer V pyramidal cells in rat barrel cortex. J. Neurosci. 21, 3580–3592 (2001).
Shipp, S. The importance of being agranular: a comparative account of visual and motor cortex. Phil. Trans. R. Soc. Lond. B 360, 797–814 (2005).
Llinás, R. I of the Vortex: From Neurons to Self (M.I.T. Press, Cambridge, Massachusetts, 2002).
Huber, D. et al. Sparse optical microstimulation in barrel cortex drives learned behaviour in freely moving mice. Nature 451, 61–64 (2007).
Castro-Alamancos, M.A. Origin of synchronized oscillations induced by neocortical disinhibition in vivo. J. Neurosci. 20, 9195–9206 (2000).
Pouille, F. & Scanziani, M. Routing of spike series by dynamic circuits in the hippocampus. Nature 429, 717–723 (2004).
Acknowledgements
We thank W. Kath, A. Stepanyants, K. Svoboda, M. Tresch, and J. Waters for comments and suggestions. We appreciate funding support provided through grants from the Whitehall Foundation, Simons Foundation and Rett Syndrome Research Foundation (G.M.G.S.).
Author information
Authors and Affiliations
Contributions
Experiments were conducted by N.W., L.W., J.Y. and G.M.G.S., and simulations by S.A.S. and G.M.G.S. All authors participated in data analysis and interpretation. G.M.G.S. supervised the project and drafted the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Methods (PDF 291 kb)
Rights and permissions
About this article
Cite this article
Weiler, N., Wood, L., Yu, J. et al. Top-down laminar organization of the excitatory network in motor cortex. Nat Neurosci 11, 360–366 (2008). https://doi.org/10.1038/nn2049
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn2049
This article is cited by
-
Coenzyme Q10 supplementation improves the motor function of middle-aged mice by restoring the neuronal activity of the motor cortex
Scientific Reports (2023)
-
MeCP2 deficiency impairs motor cortical circuit flexibility associated with motor learning
Molecular Brain (2022)
-
Modelling the laminar connectome of the human brain
Brain Structure and Function (2022)
-
Untangling the cortico-thalamo-cortical loop: cellular pieces of a knotty circuit puzzle
Nature Reviews Neuroscience (2021)
-
The human motor cortex microcircuit: insights for neurodegenerative disease
Nature Reviews Neuroscience (2020)