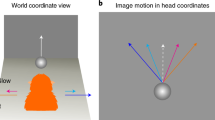
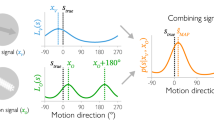
Abstract
In the middle temporal (MT) area of primates, many motion-sensitive neurons with a wide range of preferred directions respond to a stimulus moving in a single direction. These neurons are involved in direction perception, but it is not clear how perceptual decisions are related to the population response. We recorded the activities of MT neurons in rhesus monkeys while they discriminated closely related directions, and examined the relationship between the activities of neurons tuned to different directions and the monkeys' choices. Perceptual decisions were significantly correlated with the activities of the highest-precision neurons but not with those of the lowest-precision neurons. The combined performance of the high-precision neurons matched the monkeys' behavior, whereas the ability to predict behavior based on the entire active population was poor. These results suggest that fine discrimination decisions are crucially dependent on the activities of the most informative neurons.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Barlow, H.B. Single units and sensation: a neuron doctrine for perceptual psychology? Perception 1, 371–394 (1972).
Albright, T.D., Jessell, T.M., Kandel, E.R. & Posner, M.I. Neural science: a century of progress and the mysteries that remain. Neuron 25, S1–S55 (2000).
deCharms, R.C. & Zador, A. Neural representation and the cortical code. Annu. Rev. Neurosci. 23, 613–647 (2000).
Erickson, R.P. The evolution and implications of population and modular neural coding ideas. (Chapter 2) in Progress in Brain Research Vol. 130 (ed. Nicolelis, M.A.L.) 9–29 (Elsevier, Amsterdam, 2001).
Young, T. The Bakerian Lecture: on the theory of light and colours. Phil. Trans. R. Soc. Lond. 92, 12–48 (1802).
Barlow, H.B. The neuron doctrine in perception. in The Cognitive Neurosciences (ed. Gazzaniga, M.S.) 415–435 (Cambridge, Massachusetts, 1995).
De Valois, R.L., Abramov, I. & Mead, W.R. Single cell analysis of wavelength discrimination at the lateral geniculate nucleus in the macaque. J. Neurophysiol. 30, 415–433 (1967).
Bradley, A., Skottun, B.C., Ohzawa, I., Sclar, G. & Freeman, R.D. Visual orientation and spatial frequency discrimination: a comparison of single neurons and behavior. J. Neurophysiol. 57, 755–772 (1987).
Ohzawa, I., DeAngelis, G.C. & Freeman, R.D. Stereoscopic depth discrimination in the visual cortex: neurons ideally suited as disparity detectors. Science 249, 1037–1041 (1990).
Parker, A.J. & Newsome, W.T. Sense and the single neuron: probing the physiology of perception. Annu. Rev. Neurosci. 21, 227–277 (1998).
Dubner, R. & Zeki, S. Response properties and receptive fields of cells in an anatomically defined region of the superior temporal sulcus in the monkey. Brain Res. 35, 528–532 (1971).
Allman, J.M. & Kaas, K.H. A representation of the visual field in the caudal third of the middle temporal gyrus of the owl monkey (Aotus trivirgatus). Brain Res. 31, 85–105 (1971).
Zeki, S. J. Physiol. (Lond.) Functional organization of a visual area in the posterior bank of the superior temporal sulcus of the rhesus monkey. 236, 549–573 (1974).
Maunsell, J.H.R. & Van Essen, D.C. Functional properties of neurons in middle temporal visual area of the macaque monkey. I. Selectivity for stimulus direction, speed, and orientation. J. Neurophysiol. 49, 1127–1147 (1983).
Albright, T.D. Direction and orientation selectivity of neurons in visual area MT of the macaque. J. Neurophysiol. 52, 1106–1130 (1984).
Snowden, R.J., Treue, S. & Andersen, R.A. The response of neurons in areas V1 and MT of the alert rhesus monkey to moving random dot patterns. Exp. Brain Res. 88, 389–400 (1992).
Newsome, W.T., Britten, K.H. & Movshon, J.A. Neuronal correlates of a perceptual decision. Nature 341, 52–54 (1989).
Britten, K.H., Shadlen, M.N., Newsome, W.T. & Movshon, J.A. The analysis of visual motion: a comparison of neuronal and psychophysical performance. J. Neurosci. 12, 4745–4765 (1992).
Britten, K.H., Newsome, W.T., Shadlen, M.N., Celebrini, S. & Movshon, J.A. A relationship between behavioral choice and the visual responses of neurons in macaque MT. Vis. Neurosci. 13, 87–100 (1996).
Shadlen, M.N., Britten, K.H., Newsome, W.T. & Movshon, J.A. A computational analysis of the relationship between neuronal and behavioral responses to visual motion. J. Neurosci. 16, 1486–1510 (1996).
Treue, S., Hol, K. & Rauber, H.J. Seeing multiple directions of motion-physiology and psychophysics. Nat. Neurosci. 3, 270–276 (2000).
Sekuler, R., Watamaniuk, S.N.J. & Blake, R. in Steven's Handbook of Experimental Psychology Vol. 1 (eds. Yantis, S. & Pashler, H.) 121–176 (Wiley, New York, 2002).
Watamaniuk, S.N.J., Sekuler, R. & Williams, D.W. Direction perception in complex dynamic displays: the integration of direction information. Vision Res. 29, 47–59 (1989).
Watamaniuk, S.N.J. & Sekuler, R. Temporal and spatial integration in dynamic random-dot stimuli. Vision Res. 32, 2341–2347 (1992).
Green, D.M. & Swets, J.A. Signal Detection Theory and Psychophysics (Wiley, New York, 1966).
Regan, D. & Beverly, K.I. Spatial frequency discrimination and detection: comparision of postadaptation thresholds. J. Opt. Soc. Am. 73, 1684–1690 (1983).
Regan, D. & Beverly, K.I. Postadaptation orientation discrimination. J. Opt. Soc. Am. A 2, 147–155 (1985).
Vogels, R. & Orban, G.A. How well do response changes of striate neurons signal differences in orientation: a study in the discriminating monkey. J. Neurosci. 10, 3543–3558 (1990).
Wilson, H.R. & Regan, D. Spatial frequency adaptation and grating discrimination: predictions of a line element model. J. Opt. Soc. Am. A 1, 1091–1096 (1984).
Hol, K. & Treue, S. Different populations of neurons contribute to the detection and discrimination of visual motion. Vision Res. 41, 685–689 (2001).
Ash, R. Information Theory (Wiley, New York, 1965).
Reza, F. An introduction to information theory (Dover, New York, 1994).
Quick, R.F. A vector-magnitude model of contrast detection. Kybernetik 16, 65–67 (1974).
Seung, H.S. & Sompolinsky, H. Simple models for reading neuronal population codes. Proc. Natl Acad. Sci. USA 90, 10749–10753 (1993).
Osborne, L.C., Bialek, W. & Lisberger, S.G. Time course of information about motion direction in visual area MT of macaque monkeys. J. Neurosci. 24, 3210–3222 (2004).
Mazurek, M.E., Roitman, J.D., Ditterich, J. & Shadlen, M.N. A role for neural integrators in perceptual decision making. Cereb. Cortex 13, 1257–1269 (2003).
Cook, E.P. & Maunsell, J.H.R. Dynamics of neuronal responses in macaque MT and VIP during motion detection. Nat. Neurosci. 5, 985–994 (2002).
Gilbert, C.D., Sigman, M. & Crist, R.E. The neural basis of perceptual learning. Neuron 31, 681–697 (2001).
Poggio, T. & Shelton, C.R. Learning in brains and machines. Spat. Vis. 13, 287–296 (2000).
Schultz, W. Predictive reward signal of dopamine neurons. J. Neurophysiol. 80, 1–27 (1998).
Schoups, A., Vogels, R., Qian, N. & Orban, G. Practising orientation identification improves orientation coding in V1 neurons. Nature 412, 549–553 (2001).
Judge, S.J., Richmond, B.J. & Chu, F.C. Implantation of magnetic search coils for measurement of eye position: an improved method. Vision Res. 20, 535–538 (1980).
Liu, J. & Newsome, W.T. Functional organization of speed tuned neurons in visual area MT. J. Neurophysiol. 89, 246–256 (2003).
Pelli, D.G. Uncertainty explains many aspects of visual contrast detection and discrimination. J. Opt. Soc. Am. A 2, 1508–1532 (1985).
Zohary, E., Shadlen, M.N. & Newsome, W.T. Correlated neuronal discharge rate and its implications for psychophysical performance. Nature 370, 140–143 (1994).
Acknowledgements
Supported by US National Institutes of Health grant R01 EY013138. We thank H. Bedell, R. Born, W. Bosking, K. Britten, G. DeAngelis, M. Fukushima, W. Newsome, H. Ogmen, S. Patel, B. Scott, M. Shadlen, P. Wallisch and S. Watamaniuk for suggestions and comments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Neural precision is plotted as a function of the slope of the direction-tuning curve near the stimuli directions. (GIF 9 kb)
Supplementary Fig. 2
Psychometric function for reference stimulus directions of 85° and 95°, pooled from data over several recording sessions. If the monkey had ignored the reference, then the responses would have been at the 100% for all direction differences about 85° reference and at 0% for all direction differences about the 95° reference, resulting in a flat line near the 50% level. (GIF 16 kb)
Supplementary Fig. 3
Threshold ratio of neuron to behaviour calculated using the firing rates from the test interval (for the trials in which the test direction = reference direction) as the reference histogram. These ratios are not statistically different from those in Fig. 3a (t-test, p>0.8). (GIF 15 kb)
Supplementary Fig. 4
Thresholds are shown as a function of integration time. The top figure shows the psychophysical data and the bottom, the neural data. (GIF 9 kb)
Supplementary Fig. 5
Psychophysical and neurometric functions for three neurons are shown comparatively for two ranges of direction-differences. The left row shows psychometric and neurometric functions for the smaller range of direction differences and the right row for the larger range. Each row of data is for one neuron. “N Thresh” is the neurometric threshold and “P Thresh” is the psychophysical threshold. (GIF 20 kb)
Supplementary Fig. 6
Psychophysical thresholds are compared for the two ranges of direction-differences. (GIF 9 kb)
Supplementary Fig. 7
Ratio of the neurometric threshold estimated with the wider range of direction differences (−16°,+16°) to that estimated with the shorter range (−3°,+3°) is shown for 132 neurons. (GIF 13 kb)
Rights and permissions
About this article
Cite this article
Purushothaman, G., Bradley, D. Neural population code for fine perceptual decisions in area MT. Nat Neurosci 8, 99–106 (2005). https://doi.org/10.1038/nn1373
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1373
This article is cited by
-
Neural signature of the perceptual decision in the neural population responses of the inferior temporal cortex
Scientific Reports (2022)
-
A neural correlate of perceptual segmentation in macaque middle temporal cortical area
Nature Communications (2022)
-
Revealing nonlinear neural decoding by analyzing choices
Nature Communications (2021)
-
Population codes of prior knowledge learned through environmental regularities
Scientific Reports (2021)
-
Optimality and heuristics in perceptual neuroscience
Nature Neuroscience (2019)