Abstract
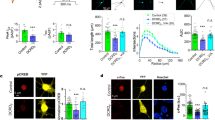
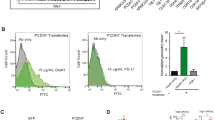
Growth cone motility is regulated by both fast voltage-dependent Ca2+ channels and by unknown receptor-operated Ca2+ entry mechanisms. Transient receptor potential (TRP) homomeric TRPC5 ion channels are receptor-operated, Ca2+-permeable channels predominantly expressed in the brain. Here we show that TRPC5 is expressed in growth cones of young rat hippocampal neurons. Our results indicate that TRPC5 channel subunits interact with the growth cone–enriched protein stathmin 2, are packaged into vesicles and are carried to newly forming growth cones and synapses. Once in the growth cone, TRPC5 channels regulate neurite extension and growth-cone morphology. Dominant-negative TRPC5 expression allowed significantly longer neurites and filopodia to form. We conclude that TRPC5 channels are important components of the mechanism controlling neurite extension and growth cone morphology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Berridge, M.J. Neuronal calcium signaling. Neuron 21, 13–26 (1998).
Berridge, M.J., Lipp, P. & Bootman, M.D. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell. Biol. 1, 11–21 (2000).
Crabtree, G.R. Calcium, calcineurin, and the control of transcription. J. Biol. Chem. 276, 2313–2316 (2001).
Gomez, T.M., Robles, E., Poo, M. & Spitzer, N.C. Filopodial calcium transients promote substrate-dependent growth cone turning. Science 291, 1983–1987 (2001).
Gomez, T.M. & Spitzer, N.C. In vivo regulation of axon extension and pathfinding by growth-cone calcium transients. Nature 397, 350–355 (1999).
Spitzer, N.C., Lautermilch, N.J., Smith, R.D. & Gomez, T.M. Coding of neuronal differentiation by calcium transients. Bioessays 22, 811–817 (2000).
Clapham, D.E., Runnels, L.W. & Strübing, C. The TRP ion channel family. Nat. Rev. Neurosci. 2, 387–396 (2001).
Harteneck, C., Plant, T.D. & Schultz, G. From worm to man: three subfamilies of TRP channels. Trends Neurosci. 23, 159–166 (2000).
Montell, C., Birnbaumer, L. & Flockerzi, V. The TRP channels, a remarkably functional family. Cell 108, 595–598 (2002).
Strübing, C., Krapivinsky, G., Krapivinsky, L. & Clapham, D.E. TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron 29, 645–655 (2001).
Schaefer, M. et al. Receptor-mediated regulation of the nonselective cation channels TRPC4 and TRPC5. J. Biol. Chem. 275, 17517–17526 (2000).
Hofmann, T., Schaefer, M., Schultz, G. & Gudermann, T. Subunit composition of mammalian transient receptor potential channels in living cells. Proc. Natl. Acad. Sci. USA 99, 7461–7466 (2002).
Okada, T. et al. Molecular cloning and functional characterization of a novel receptor- activated TRP Ca2+ channel from mouse brain. J. Biol. Chem. 273, 10279–10287 (1998).
Okada, T. et al. Molecular and functional characterization of a novel mouse transient receptor potential protein homologue TRP7. Ca2+-permeable cation channel that is constitutively activated and enhanced by stimulation of G protein-coupled receptor. J. Biol. Chem. 274, 27359–27370 (1999).
Bartlett, W.P. & Banker, G.A. An electron microscopic study of the development of axons and dendrites by hippocampal neurons in culture. I. Cells which develop without intercellular contacts. J. Neurosci. 4, 1944–1953 (1984).
Weed, S.A. & Parsons, J.T. Cortactin: coupling membrane dynamics to cortical actin assembly. Oncogene 20, 6418–6434 (2001).
Weaver, A.M. et al. Cortactin promotes and stabilizes Arp2/3-induced actin filament network formation. Curr. Biol. 11, 370–374 (2001).
Weed, S.A., Du, Y. & Parsons, J.T. Translocation of cortactin to the cell periphery is mediated by the small GTPase Rac1. J. Cell. Sci. 111, 2433–2443 (1998).
Du, Y., Weed, S.A., Xiong, W.C., Marshall, T.D. & Parsons, J.T. Identification of a novel cortactin SH3 domain-binding protein and its localization to growth cones of cultured neurons. Mol. Cell. Biol. 18, 5838–5851 (1998).
Martin, T.F. Prime movers of synaptic vesicle exocytosis. Neuron 34, 9–12 (2002).
Chen, Y.A. & Scheller, R.H. SNARE-mediated membrane fusion. Nat. Rev. Mol. Cell. Biol. 2, 98–106 (2001).
Ahmari, S.E., Buchanan, J. & Smith, S.J. Assembly of presynaptic active zones from cytoplasmic transport packets. Nat. Neurosci. 3, 445–451 (2000).
Nakata, T., Terada, S. & Hirokawa, N. Visualization of the dynamics of synaptic vesicle and plasma membrane proteins in living axons. J. Cell Biol. 140, 659–674 (1998).
Jung, S. et al. Lanthanides potentiate TRPC5 currents by an action at extracellular sites close to the pore mouth. J. Biol. Chem. 278, 3562–3571 (2003).
Nadler, M.J.S. et al. LTRPC7 is a Mg–ATP-regulated divalent cation channel required for cell viability. Nature 411, 590–595 (2001).
Runnels, L.W., Yue, L. & Clapham, D.E. TRP-PLIK, a bifunctional protein with kinase and ion channel activities. Science 291, 1043–1047 (2001).
Ozon, S., Byk, T. & Sobel, A. SCLIP: a novel SCG10-like protein of the stathmin family expressed in the nervous system. J. Neurochem. 70, 2386–2396 (1998).
Gavet, O. et al. The stathmin phosphoprotein family: intracellular localization and effects on the microtubule network. J. Cell. Sci. 111, 3333–3346 (1998).
Stein, R., Mori, N., Matthews, K., Lo, L.C. & Anderson, D.J. The NGF-inducible SCG10 mRNA encodes a novel membrane-bound protein present in growth cones and abundant in developing neurons. Neuron 1, 463–476 (1988).
Ozon, S., Maucuer, A. & Sobel, A. The stathmin family—molecular and biological characterization of novel mammalian proteins expressed in the nervous system. Eur. J. Biochem. 248, 794–806 (1997).
Cassimeris, L. The oncoprotein 18/stathmin family of microtubule destabilizers. Curr. Opin. Cell Biol. 14, 18–24 (2002).
Antonsson, B. et al. Purification, characterization, and in vitro phosphorylation of the neuron-specific membrane-associated protein SCG10. Protein Expr. Purif. 9, 363–371 (1997).
Lutjens, R. et al. Localization and targeting of SCG10 to the trans-Golgi apparatus and growth cone vesicles. Eur. J. Neurosci. 12, 2224–2234 (2000).
Goshima, Y., Nakamura, F., Strittmatter, P. & Strittmatter, S.M. Collapsin-induced growth cone collapse mediated by an intracellular protein related to UNC-33. Nature 376, 509–514 (1995).
Takahashi, T., Nakamura, F., Jin, Z., Kalb, R.G. & Strittmatter, S.M. Semaphorins A and E act as antagonists of neuropilin-1 and agonists of neuropilin-2 receptors. Nat. Neurosci. 1, 487–493 (1998).
Belmont, L.D. & Mitchison, T.J. Identification of a protein that interacts with tubulin dimers and increases the catastrophe rate of microtubules. Cell 84, 623–631 (1996).
Song, H. & Poo, M. The cell biology of neuronal navigation. Nat. Cell Biol. 3, E81–88 (2001).
Bixby, J.L. & Spitzer, N.C. Early differentiation of vertebrate spinal neurons in the absence of voltage-dependent Ca2+ and Na+ influx. Dev. Biol. 106, 89–96 (1984).
Gu, X. & Spitzer, N.C. Distinct aspects of neuronal differentiation encoded by frequency of spontaneous Ca2+ transients. Nature 375, 784–787 (1995).
Kuhn, T.B., Brown, M.D. & Bamburg, J.R. Rac1-dependent actin filament organization in growth cones is necessary for beta1-integrin-mediated advance but not for growth on poly-D-lysine. J. Neurobiol. 37, 524–540 (1998).
Goslin, K. & Banker, G. Experimental observations on the development of polarity by hippocampal neurons in culture. J. Cell Biol. 108, 1507–1516 (1989).
Lorenz, E. et al. Evidence for direct physical association between a K+ channel (Kir6.2) and an ATP-binding cassette protein (SUR1) which affects cellular distribution and kinetic behavior of an ATP-sensitive K+ channel. Mol. Cell. Biol. 18, 1652–1659 (1998).
Acknowledgements
We thank G. Grenningloh (Institut de Biologie Cellulaire, Lausanne, Switzerland), A. Sobel (INSERM, Paris, France) and M. Gullberg (Umea University, Umea, Sweden) for stathmin reagents. We also thank C. Strübing for the DN-TRPC5 construct. We are indebted to M. Ericsson in the HMS Electron Microscopy Facility for technical assistance, and to S. Gapon for assistance with hippocampal cultures. We thank D. Corey, B. Bean and T. Schwarz for discussions and technical advice. This work was supported by a Howard Hughes Medical Institute Predoctoral Fellowship (A.G.), a Kaplan Fellowship (B.N.) and the Howard Hughes Medical Institute (D.E.C.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1.
Model of TRPC5 ’s role in growth cone morphology and motility. TRPC5 associates with stathmin 2 on VAMP-and synaptotagmin-containing transport packets. When packets arrive at the growth cone,VAMP and synaptotagmin mediate vesicle docking and fusion,thereby increasing functional TRPC5 channels in the plasma membrane.In response to extracellular growth and motility signals,G-protein-coupled receptors (GPCR)and growth factor receptors (RTK)activate TRPC5 channels in the plasma membrane through PLC-dependent mechanisms.TRPC5-mediated Ca 2+ influx affects growth cone morphology and motility through control of filopodial and neurite extension. (JPG 87 kb)
Rights and permissions
About this article
Cite this article
Greka, A., Navarro, B., Oancea, E. et al. TRPC5 is a regulator of hippocampal neurite length and growth cone morphology. Nat Neurosci 6, 837–845 (2003). https://doi.org/10.1038/nn1092
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn1092
This article is cited by
-
TRP Channels in Stroke
Neuroscience Bulletin (2023)
-
Upregulation of TRPC5 in hippocampal excitatory synapses improves memory impairment associated with neuroinflammation in microglia knockout IL-10 mice
Journal of Neuroinflammation (2021)
-
TRPC5 regulates axonal outgrowth in developing retinal ganglion cells
Laboratory Investigation (2020)
-
The versatile Kv channels in the nervous system: actions beyond action potentials
Cellular and Molecular Life Sciences (2020)
-
Na+ entry through heteromeric TRPC4/C1 channels mediates (−)Englerin A-induced cytotoxicity in synovial sarcoma cells
Scientific Reports (2017)