Abstract
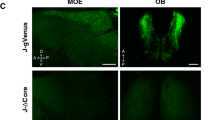
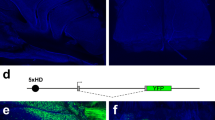
To study the mutually exclusive expression of odorant receptor (OR) genes, we generated transgenic mice that carried the murine OR gene MOR28. Expression of the transgene and the endogenous MOR28 was distinguished by using two different markers, β-galactosidase and green fluorescent protein (GFP), respectively. Double staining of the olfactory epithelium revealed that the two genes were rarely expressed simultaneously in individual olfactory neurons. A similar exclusion was also observed between differently tagged but identical transgenes integrated into the same locus of one particular chromosome. Although allelic inactivation has been reported for the choice between the maternal and paternal alleles, this is the first demonstration of mutually exclusive activation among non-allelic OR gene members with identical coding and regulatory sequences. Such an unusual mode of gene expression, monoallelic and mutually exclusive, has previously been shown only for the antigen-receptor genes of the immune system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Buck, L. & Axel, R. A novel multigene family may encode odorant receptors: a molecular basis for odorant recognition. Cell 65, 175–187 ( 1991).
Parmentier, M. et al. Expression of members of the putative olfactory receptor gene family in mammalian germ cells. Nature 355, 453–455 (1992).
Sullivan, S. L., Adamson, M. C., Ressler, K. J., Kozak, C. A. & Buck, L. B. The chromosomal distribution of mouse odorant receptor genes. Proc. Natl. Acad. Sci. USA 93, 884–888 (1996).
Ben-Arie, N. et al. Olfactory receptor gene cluster on human chromosome 17: possible duplication of an ancestral receptor repertoire. Hum. Mol. Genet. 3, 229–235 ( 1994).
Rouquier, S. et al. Distribution of olfactory receptor genes in the human genome . Nat. Genet. 18, 243–250 (1998).
Ressler, K. J., Sullivan, S. L. & Buck, L. B. A zonal organization of odorant receptor gene expression in the olfactory epithelium. Cell 73, 597 –609 (1993).
Vassar, R., Ngai, J. & Axel, R. Spatial segregation of odorant receptor expression in the mammalian olfactory epithelium. Cell 74, 309– 318 (1993).
Ngai, J. et al. Coding of olfactory information: topography of odorant receptor expression in the catfish olfactory epithelium. Cell 72, 667–680 (1993).
Malnic, B., Hirono, J., Sato, T. & Buck, L. B. Combinatorial receptor codes for odors. Cell 96, 713– 723 (1999).
Chess, A., Simon, I., Cedar, H. & Axel, R. Allelic inactivation regulates olfactory receptor gene expression. Cell 78, 823–834 (1994).
Belluscio, L., Koentges, G., Axel, R. & Dulac, C. A map of pheromone receptor activation in the mammalian brain. Cell 97 , 209–220 (1999).
Rodriguez, I., Feinstein, P. & Mombaerts, P. Variable patterns of axonal projections of sensory neurons in the mouse vomeronasal system. Cell 97, 199–208 (1999).
Ressler, K. J., Sullivan, S. L. & Buck, L. B. Information coding in the olfactory system: evidence for a stereotyped and highly organized epitope map in the olfactory bulb. Cell 79, 1245–1255 ( 1994).
Vassar, R. et al. Topographic organization of sensory projections to the olfactory bulb. Cell 79, 981–991 (1994).
Mombaerts, P. et al. Visualizing an olfactory sensory map. Cell 87, 675–686 (1996).
Wang, F., Nemes, A., Mendelsohn, M. & Axel, R. Odorant receptors govern the formation of a precise topographic map. Cell 93, 47–60 ( 1998).
Tsuboi, A. et al. Olfactory neurons expressing closely linked and homologous odorant receptor genes tend to project their axons to neighboring glomeruli on the olfactory bulb. J. Neurosci. 19, 8409–8418 (1999).
Qasba, P. & Reed, R. R. Tissue and zonal-specific expression of an olfactory receptor transgene. J. Neurosci. 18 , 227–236 (1998).
Ebrahimi, F. A. W., Edmondson, J., Rothstein, R. & Chess, A. YAC transgene-mediated olfactory receptor gene choice. Dev. Dyn. 217, 225–231 ( 2000).
Tonegawa, S. Somatic generation of antibody diversity. Nature 302 , 575–581 (1983).
Strathern, J. N. et al. Homothallic switching of yeast mating type cassettes is initiated by a double-stranded cut in the MAT locus. Cell 31, 183–192 (1982).
Borst, P. Molecular genetics and antigenic variation. Immunol. Today 12, 29–33 (1991).
Krautwurst, D., Yau, K. W. & Reed, R. R. Identification of ligands for olfactory receptors by functional expression of a receptor library. Cell 95 , 917–926 (1998).
Callahan, C. A. & Thomas, J. B. Tau-beta-galactosidase, an axon-targeted fusion protein. Proc. Natl. Acad. Sci. USA 91, 5972–5976 (1994).
Moriyoshi, K., Richards, L. J., Akazawa, C., O'Leary, D. D. M. & Nakanishi, S. Labeling neural cells using adenoviral gene transfer of membrane-targeted GFP. Neuron 16, 255–260 ( 1996).
Gu, H., Zou, Y. R. & Rajewsky, K. Independent control of immunoglobulin switch recombination at individual switch regions evidenced through Cre-loxP-mediated gene targeting . Cell 73, 1155–1164 (1993).
Soriano, P., Montogomery, C., Geske, R. & Bradley, A. Targeted disruption of the c-src proto-oncogene leads to osteopetrosis in mice. Cell 64, 693–702 (1991).
Yagi, T. et al. A novel negative selection for homologous recombination using diphtheria toxin A fragment gene. Anal. Biochem. 214 , 77–86 (1993).
Bungert, J. et al. Synergistic regulation of human beta-globin gene switching by locus control region elements HS3 and HS4. Genes Dev. 9, 3083–3096 (1995).
Wutz, A. et al. Imprinted expression of the Igf2r gene depends on an intronic CpG island. Nature 389, 745– 749 (1997).
Asano, M. et al. Growth retardation and early death of β-1,4-galactosyltransferase knockout mice with augmented proliferation and abnormal differentiation of epithelial cells. EMBO J. 16, 1850– 1857 (1997).
Shibata, H. et al. Rapid colorectal adenoma formation inhibited by conditional targeting of the apc gene. Science 278, 120–123 (1997).
Nagy, A., Rossant, J., Nagy, R., Abramow-Newerly, W. & Roder, J. C. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc. Natl. Acad. Sci. USA 90, 8424–8428 ( 1993).
Acknowledgements
We thank the following people for providing us with plasmid samples or cell lines: H. Gu (pGH-1), P. Soriano (pPGKneobpA), T. Yagi (DT-A cassette), J. Thomas (tau–lacZ cassette), K. Moriyoshi (pGAP–GFP), A. Nagy (R1 cells) and I. Saito (adenovirus vector). This work was supported by the Special Promotion Research Grants from the Ministry of Education and Culture of Japan and by grants from Toray Science Foundation, Nissan Science Foundation and Mitsubishi Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Serizawa, S., Ishii, T., Nakatani, H. et al. Mutually exclusive expression of odorant receptor transgenes. Nat Neurosci 3, 687–693 (2000). https://doi.org/10.1038/76641
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/76641
This article is cited by
-
Future therapeutic strategies for olfactory disorders: electrical stimulation, stem cell therapy, and transplantation of olfactory epithelium—an overview
HNO (2023)
-
Immature olfactory sensory neurons provide behaviourally relevant sensory input to the olfactory bulb
Nature Communications (2022)
-
C. elegans-based chemosensation strategy for the early detection of cancer metabolites in urine samples
Scientific Reports (2021)
-
Zukunftsweisende Therapieansätze bei Riechstörungen: elektrische Stimulation, Stammzelltherapie und Transplantation von Riechepithel – eine Übersicht
HNO (2021)
-
Primary dendrites of mitral cells synapse unto neighboring glomeruli independent of their odorant receptor identity
Communications Biology (2019)