Abstract
As an essential means of resolving conflicts, aggression is expressed by both sexes but often at a higher level in males than in females. Recent studies suggest that cells in the ventrolateral part of the ventromedial hypothalamus (VMHvl) that express estrogen receptor-α (Esr1) and progesterone receptor are essential for male but not female mouse aggression. In contrast, here we show that VMHvlEsr1+ cells are indispensable for female aggression. This population was active when females attacked naturally. Inactivation of these cells reduced female aggression whereas their activation elicited attack. Additionally, we found that female VMHvl contains two anatomically distinguishable subdivisions that showed differential gene expression, projection and activation patterns after mating and fighting. These results support an essential role of the VMHvl in both male and female aggression and reveal the existence of two previously unappreciated subdivisions in the female VMHvl that are involved in distinct social behaviors.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Darwin, C. The Descent of Man, and Selection in Relation to Sex (D. Appleton and Company, 1871).
Lindenfors, P. & Tullberg, B.S. Evolutionary aspects of aggression the importance of sexual selection. Adv. Genet. 75, 7–22 (2011).
Kruk, M.R. et al. Discriminant analysis of the localization of aggression-inducing electrode placements in the hypothalamus of male rats. Brain Res. 260, 61–79 (1983).
Olivier, B. & Wiepkema, P.R. Behaviour changes in mice following electrolytic lesions in the median hypothalamus. Brain Res. 65, 521–524 (1974).
Siegel, A., Roeling, T.A.P., Gregg, T.R. & Kruk, M.R. Neuropharmacology of brain-stimulation-evoked aggression. Neurosci. Biobehav. Rev. 23, 359–389 (1999).
Lee, H. et al. Scalable control of mounting and attack by Esr1+ neurons in the ventromedial hypothalamus. Nature 509, 627–632 (2014).
Lin, D. et al. Functional identification of an aggression locus in the mouse hypothalamus. Nature 470, 221–226 (2011).
Falkner, A.L., Grosenick, L., Davidson, T.J., Deisseroth, K. & Lin, D. Hypothalamic control of male aggression-seeking behavior. Nat. Neurosci. 19, 596–604 (2016).
Yang, C.F. et al. Sexually dimorphic neurons in the ventromedial hypothalamus govern mating in both sexes and aggression in males. Cell 153, 896–909 (2013).
Falkner, A.L., Dollar, P., Perona, P., Anderson, D.J. & Lin, D. Decoding ventromedial hypothalamic neural activity during male mouse aggression. J. Neurosci. 34, 5971–5984 (2014).
Falkner, A.L. & Lin, D. Recent advances in understanding the role of the hypothalamic circuit during aggression. Front. Syst. Neurosci. 8, 168 (2014).
Kruk, M.R. et al. Comparison of aggressive behaviour induced by electrical stimulation in the hypothalamus of male and female rats. Prog. Brain Res. 61, 303–314 (1984).
Ogawa, S., Lubahn, D.B., Korach, K.S. & Pfaff, D.W. Behavioral effects of estrogen receptor gene disruption in male mice. Proc. Natl. Acad. Sci. USA 94, 1476–1481 (1997).
Spiteri, T. et al. The role of the estrogen receptor alpha in the medial amygdala and ventromedial nucleus of the hypothalamus in social recognition, anxiety and aggression. Behav. Brain Res. 210, 211–220 (2010).
Sano, K., Tsuda, M.C., Musatov, S., Sakamoto, T. & Ogawa, S. Differential effects of site-specific knockdown of estrogen receptor α in the medial amygdala, medial pre-optic area, and ventromedial nucleus of the hypothalamus on sexual and aggressive behavior of male mice. Eur. J. Neurosci. 37, 1308–1319 (2013).
Aou, S., Oomura, Y. & Yoshimatsu, H. Neuron activity of the ventromedial hypothalamus and the medial preoptic area of the female monkey during sexual behavior. Brain Res. 455, 65–71 (1988).
Oomura, Y., Aou, S., Koyama, Y., Fujita, I. & Yoshimatsu, H. Central control of sexual behavior. Brain Res. Bull. 20, 863–870 (1988).
Pfaff, D.W. & Sakuma, Y. Deficit in the lordosis reflex of female rats caused by lesions in the ventromedial nucleus of the hypothalamus. J. Physiol. (Lond.) 288, 203–210 (1979).
Pfaff, D.W. & Sakuma, Y. Facilitation of the lordosis reflex of female rats from the ventromedial nucleus of the hypothalamus. J. Physiol. (Lond.) 288, 189–202 (1979).
Musatov, S., Chen, W., Pfaff, D.W., Kaplitt, M.G. & Ogawa, S. RNAi-mediated silencing of estrogen receptor alpha in the ventromedial nucleus of hypothalamus abolishes female sexual behaviors. Proc. Natl. Acad. Sci. USA 103, 10456–10460 (2006).
Yang, C.F. & Shah, N.M. Representing sex in the brain, one module at a time. Neuron 82, 261–278 (2014).
Bayless, D.W. & Shah, N.M. Genetic dissection of neural circuits underlying sexually dimorphic social behaviours. Phil. Trans. R. Soc. Lond. B 371, 20150109 (2016).
Yang, T. & Shah, N.M. Molecular and neural control of sexually dimorphic social behaviors. Curr. Opin. Neurobiol. 38, 89–95 (2016).
Lonstein, J.S. & Gammie, S.C. Sensory, hormonal, and neural control of maternal aggression in laboratory rodents. Neurosci. Biobehav. Rev. 26, 869–888 (2002).
Parmigiani, S., Palanza, P., Rogers, J. & Ferrari, P.F. Selection, evolution of behavior and animal models in behavioral neuroscience. Neurosci. Biobehav. Rev. 23, 957–969 (1999).
Hurst, J.L. & Barnard, C.J. Kinship and social tolerance among female and juvenile wild house mice - kin bias but not kin discrimination. Behav. Ecol. Sociobiol. 36, 333–342 (1995).
Koyama, Y., Fujita, I., Aou, S. & Oomura, Y. Proceptive presenting elicited by electrical stimulation of the female monkey hypothalamus. Brain Res. 446, 199–203 (1988).
Gunaydin, L.A. et al. Natural neural projection dynamics underlying social behavior. Cell 157, 1535–1551 (2014).
Cui, G. et al. Concurrent activation of striatal direct and indirect pathways during action initiation. Nature 494, 238–242 (2013).
Chen, T.W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013).
Armbruster, B.N., Li, X., Pausch, M.H., Herlitze, S. & Roth, B.L. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc. Natl. Acad. Sci. USA 104, 5163–5168 (2007).
Dong, S., Rogan, S.C. & Roth, B.L. Directed molecular evolution of DREADDs: a generic approach to creating next-generation RASSLs. Nat. Protoc. 5, 561–573 (2010).
Boyden, E.S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Bosch, O.J., Meddle, S.L., Beiderbeck, D.I., Douglas, A.J. & Neumann, I.D. Brain oxytocin correlates with maternal aggression: link to anxiety. J. Neurosci. 25, 6807–6815 (2005).
Nomoto, K. & Lima, S.Q. Enhanced male-evoked responses in the ventromedial hypothalamus of sexually receptive female mice. Curr. Biol. 25, 589–594 (2015).
Wong, L.C. et al. Effective modulation of male aggression through lateral septum to medial hypothalamus projection. Curr. Biol. 26, 593–604 (2016).
Guzowski, J.F., McNaughton, B.L., Barnes, C.A. & Worley, P.F. Environment-specific expression of the immediate-early gene Arc in hippocampal neuronal ensembles. Nat. Neurosci. 2, 1120–1124 (1999).
Vong, L. et al. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71, 142–154 (2011).
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 13, 133–140 (2010).
Sapin, E. et al. A very large number of GABAergic neurons are activated in the tuberal hypothalamus during paradoxical (REM) sleep hypersomnia. PLoS One 5, e11766 (2010).
Jennings, J.H. et al. Visualizing hypothalamic network dynamics for appetitive and consummatory behaviors. Cell 160, 516–527 (2015).
Lein, E.S. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007).
Espina, V., Milia, J., Wu, G., Cowherd, S. & Liotta, L.A. Laser capture microdissection. Methods Mol. Biol. 319, 213–229 (2006).
Garfield, A.S. et al. A neural basis for melanocortin-4 receptor-regulated appetite. Nat. Neurosci. 18, 863–871 (2015).
Fang, J. & Clemens, L.G. Contextual determinants of female-female mounting in laboratory rats. Anim. Behav. 57, 545–555 (1999).
Xu, X. et al. Modular genetic control of sexually dimorphic behaviors. Cell 148, 596–607 (2012).
Tinbergen, N. The Study of Instinct xii edn., 228 (Clarendon, Oxford, 1951).
Unger, E.K. et al. Medial amygdalar aromatase neurons regulate aggression in both sexes. Cell Rep. 10, 453–462 (2015).
Wu, Z., Autry, A.E., Bergan, J.F., Watabe-Uchida, M. & Dulac, C.G. Galanin neurons in the medial preoptic area govern parental behaviour. Nature 509, 325–330 (2014).
Kimchi, T., Xu, J. & Dulac, C. A functional circuit underlying male sexual behaviour in the female mouse brain. Nature 448, 1009–1014 (2007).
Kuchiiwa, S. & Kuchiiwa, T. A novel semi-automated apparatus for measurement of aggressive biting behavior in mice. J. Neurosci. Methods 228, 27–34 (2014).
Chan, E., Kovacevíc, N., Ho, S.K., Henkelman, R.M. & Henderson, J.T. Development of a high resolution three-dimensional surgical atlas of the murine head for strains 129S1/SvImJ and C57Bl/6J using magnetic resonance imaging and micro-computed tomography. Neuroscience 144, 604–615 (2007).
Wang, L., Chen, I.Z. & Lin, D. Collateral pathways from the ventromedial hypothalamus mediate defensive behaviors. Neuron 85, 1344–1358 (2015).
Acknowledgements
We thank D. Anderson (California Institute of Technology) for providing Esr1-2A-Cre mice and B. Lowell (Harvard Medical School) for providing Vglut2-ires-Cre and Vgat-ires-Cre mice and AAV-DIO-synaptophysin-mCherry for the pilot experiments. We thank R. Machold and M. Baek for technical support on in situ hybridization and RNA-seq, C. Loomis at the NYULMC Experimental Pathology Research Laboratory for help on laser capture microdissection, A. Heguy and Y. Zhang at the NYULMC Genome Technology Center for help on RNA-seq and T. Lhakhang at the NYULMC Bioinformatics Laboratory for help with sequence alignment. We thank A.L. Falkner, M. Halassa, G. Stuber and G. Suh for critical comments on the manuscript. This research was supported by a JSPS oversea fellowship (K.H.), Uehara postdoctoral fellowship (K.H.), National Natural Science Foundation of China 81471630 (J.Z.), NIH 1K99NS074077 (H.L.), NIH R21NS093987 (B.R.), NIH P01NS074972 (B.R.), NIH 1R01MH101377 (D. L.), NIH 1R21MH105774-01A1 (D. L.), the Mathers Foundation (D.L.), an Irma T. Hirschl Career Scientist Award (D.L.), a Sloan Research Fellowship (D.L.), a McKnight Scholar Award (D.L.), a Whitehall Fellowship (D.L.) and a Klingenstein Fellowship Award (D.L.).
Author information
Authors and Affiliations
Contributions
D.L. supervised the project. D.L. and K.H. conceived the project, designed experiments and wrote the manuscript. K.H. and Y.H. conducted most experiments and analyzed data. R.T. conducted in vitro slice physiology. J.Z. conducted pilot pharmacological and c-Fos experiments. J.E.F. optimized in vivo single-unit recording. W.T.P. helped with pilot optogenetic experiments. A.S. helped with fiber photometry and optogenetic experiments. H.L. generated Esr1-2A-Cre mice. B.R. supervised slice physiology.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
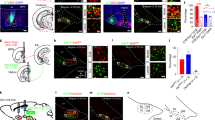
Supplementary Figure 1 The VMHvl is activated by aggression in both virgin and lactating females.
(a) Representative images showing fighting induced c-Fos (green) in the VMHvl in virgin SW female (1st row), lactating SW female (2nd and 3rd rows) and lactating C57 female (4th row) mice. Scale bar: 150 μm. (b) Average number of c-Fos+ cells per section in the VMHvl following various test conditions. Inves.: Investigate. Unpaired t-test or one way ANOVA followed by Tukey test. *p < 0.05, **p < 0.01, ***p < 0.001. Data are presented as mean ± s.e.m. See also Supplementary Table 1.
Supplementary Figure 2 Inhibiting the VMHvl reduces female aggression in wild-type animals.
(a) Viral constructs and schematics of the experiment. (b) Left: bilateral hM4Di-mCherry expression (red) in the VMHvl of a wild type SW female mouse. Scale bar: 300 μm. Right: percentage of infected cells among all DAPI stained cells in the VMHvl and its surrounding areas. (c) Timeline of the experiment. (d) Whole-cell patch clamp recording from a hM4Di-mCherry+ VMHvl neuron on a brain slice. Scale bars: 10 μm (left); 30 s and 20 mV (right). (e) Representative raster plots illustrating the effect of CNO injection on aggression (red) and investigation (green) towards an adult male intruder from animals in hM4Di group (top) and control group (bottom). Scale bar: 60 s. (f) Attack duration on each day after saline (S) and CNO (C) injection. (g-h) In comparison to saline injection, CNO injection (g) reduced attack duration (h) but did not change latency to attack. (i) Investigation duration and (j) time required to retrieve four pups were not changed after CNO injection. f-j only includes animals with over 10% of infected cells in the VMHvl and less than 5% of infected cells in regions outside of the VMHvl. (k) Changes in attack duration after CNO injection was correlated with the percentage of infected cells in the posterior VMHvl (middle) but not that in the anterior VMHvl (left) or Arc (right). k includes all animals with attack duration over 20 s on saline days regardless of the percentage of infected cells. g, h, j: paired t-test; f, i: Two-way repeated measure ANOVA followed by Holm-Sidak post-hoc multiple comparisons. k: Pearson product-moment correlation. *p < 0.05, **p < 0.01.
Supplementary Figure 3 Responses of VMHvl Esr1+ neurons to conspecific olfactory cues.
(a, b, d, e) (left): Normalized GCaMP6f (black) and mCherry (red) traces when a virgin or a lactating SW female resident investigated juvenile or adult male urine. Green shades mark periods of investigation; (right): PETHs of GCaMP6f signal aligned to the onset of investigation. (c, f) The peak ∆F/F of GCaMP6f signals during investigating urine in (c) virgin females or (f) lactating females. Note that adult male urine elicited significantly higher VMHvl responses in lactating but not virgin females. Paired t-test. *p < 0.05. Data are presented as means ± s.e.m.
Supplementary Figure 4 VMHvl Esr1+ neurons are activated during fighting or mating in female C57BL/6 mice.
(a) (left): Representative normalized GCaMP6f (black) and mCherry (red) traces during interaction with an object or various social stimuli introduced into the home cage of the test female. Colored shades mark behavioral episodes. Green: investigation; Blue: mounted; Red: attack. (right) PETHs of GCaMP6f signal aligned to the onset of various behaviors. (b) The peak ∆F/F of GCaMP6f (black) and mCherry (red) signals during various behaviors in virgin and lactating C57BL/6 female. Paired t-test. *p < 0.05, ***p < 0.001. Data are presented as means ± s.e.m.
Supplementary Figure 5 Control fluorophores show minimal fluctuation in fluorescence during social interactions.
(a) Experimental scheme. YFP and mCherry were conditionally expressed in Esr1+ neurons in the VMHvl. (b) Representative images showing mCherry (red), YFP (green), Nissl (blue) and optical fiber tract (yellow dashed line). Scale bar: 300 μm. (c) Coefficient of variation of GCamp6f, YFP and mcherry signals during encounters with a juvenile or an adult male intruder. (d) Representative normalized simultaneously recorded YFP (black) and mCherry (red) traces during interaction with various social stimuli introduced into the home cage of the test female. Colored shades mark behavioral episodes. Green: investigation; Blue: mounted; Red: attack. (e) PETHs of YFP (left) and mCherry (right) signals aligned to the onset of various behaviors. (f) Comparison of the peak ∆F/F of YFP and mCherry signals during various behaviors. Paired t-test. Data are presented as means ± s.e.m.
Supplementary Figure 6 Inactivation of Esr1+ neurons in the VMHvl reduced aggression in virgin Esr1-2A-Cre mice with a SW background and in lactating Esr1-2A-Cre mice with a C57 background.
(a) Experimental schedule. (b) A representative image showing bilateral hM4Di-mCherry expression (red) in the VMHvl. Blue: Nissl. Scale bar: 300 μm. (c) Representative raster plots illustrating effect of CNO injection on aggression (red) and investigation (green) towards a juvenile male intruder. Scale bar: 60 s. (d) CNO injection significantly reduced attack duration (left) and increased latency to attack a juvenile male intruder (right) in hM4Di group (red), but not in control group (black). (e) The investigation duration after CNO injection did not change in comparison to that after saline injection. Results from all CNO (saline) days were first averaged and then compared. (f) Experimental schedule. (g) A representative image showing bilateral hM4Di-mCherry expression (red) in the VMHvl. Blue: Nissl. Scale bar: 300 μm. (h) Representative raster plots illustrating effect of CNO injection on aggression (red) and investigation (green) towards a juvenile male intruder. Scale bar: 60 s. (i) CNO injection significantly reduced attack duration (left) and increased latency to attack a juvenile male intruder (right). (j) The investigation duration (left) and the time spent to retrieve four pups (right) after CNO injection did not change in comparison to that after saline injection. d, e, i and j: paired t-test, *p < 0.05, **p < 0.01. Data are presented as means ± s.e.m.
Supplementary Figure 7 Esr1+ neurons in the posterior VMHvl are critical for eliciting attack in female mice.
(a) A representative image showing tracks of optic fibers over the VMHvl. Scale bar: 300 μm. (b) The ending points of the optic fibers in all tested sites with >10% light induced c-Fos. Color indicates the type of light induced behavior. (c) Representative images illustrating light induced c-Fos (red), ChR2-EYFP (green), and Nissl (blue) in the anterior and posterior VMHvl of an attack-inducible site (left) and a site that no social behavior could be induced (right). Scale bars: 300 μm.
Supplementary Figure 8 Activation of Esr1+ neurons in the VMHvl induces mounting in virgin female mice with a C57BL/6 background.
(a) Experimental scheme. (b) ChR2-EYFP (green) and light-induced c-Fos (red) at an injection site. Scale bar: 300 μm. (c) Experimental schedule. (d) Representative raster plots illustrating light-induced behaviors towards a female intruder. Scale bar: 60 s. (e) Across all stimulated sites, light activation significantly increased the duration of investigation (left) and mounting (right) towards either an adult female or an adult male intruder. Paired t-test. (f) Left: representative images showing c-Fos expression in the VMHvl of virgin C57BL/6 mice after various testing conditions. Scale bar: 150 μm. Right: average number of c-Fos cells per section. Tukey test after one-way ANOVA. (g) Left: representative normalized GCaMP6f (black) and mCherry (red) traces during interaction with an adult female introduced into the home cage of the test female. Colored shades mark behavioral episodes. Green: investigation; purple: mount. Right: PETHs of GCaMP6f signal aligned to the onset of various behaviors. *p < 0.05, ***p < 0.001. Data are presented as means ± s.e.m.
Supplementary Figure 9 Activation of Esr1+ neurons in the VMHvl induces attack in lactating C57BL/6 female mice.
(a) Experimental scheme. (b) Representative histological pictures showing the ChR2-EYFP expression (green) and c-Fos (red) at an injection site. Scale bar: 300 μm. (c) Experimental schedule. (d) The percentage of VMHvl cells expressing light-induced c-Fos in the anterior vs. posterior VMHvl of all tested sites. (e) Representative raster plots illustrating light-induced behaviors towards a female intruder. Scale bar: 60 s. (f) Light activation of posterior VMHvl sites increased investigation (left) and attack (right) duration towards either an adult female or male intruder. Paired t-test, *p < 0.05, **p < 0.01. Data are presented as means ± s.e.m.
Supplementary Figure 10 Optogenetic activation of VMHvl Esr1+ cells does not affect anxiety state.
(a, e) Experimental scheme. (b, f) Experimental schedule. EPM: elevated plus maze. (c, d, g, h) No difference was found in (c, g) the time spent in in open arm or (d, h) the number of entry into the open arm during pre-stimulation, sham-on, light-on, and post-stimulation periods in (c, d) SW virgin female and (g, h) C57BL/6 lactating females. One-way ANOVA followed by Holm-Sidak post-hoc multiple comparisons. Data are presented as means ± s.e.m.
Supplementary Figure 11 Comparing responses of VMHvl neurons to juveniles and adult males in female mice.
(a) Firing rates of juvenile-excited cells before adult male introduction (base) and during investigating or staying away (no contact) from the adult male. (b) Firing rates of male-excited cells before juvenile male introduction (base) and when the recorded animal investigated, attacked or stayed away from the juvenile male. Only male-excited cells with attacking juvenile episodes are shown. (c-e) Distributions of the firing rate changes during investigation of a juvenile male, a juvenile female and two strains of adult males in all 103 recorded cells. a and b: One-way ANOVA followed by Holm-Sidak post-hoc multiple comparisons; *p <0.05; c-e: Pearson product-moment correlation.
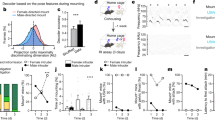
Supplementary Figure 12 Fighting- and mating-related populations are topographically organized in the VMHvl in both SW and C57 females.
(a) Schematic illustration of how the mediolateral distances of c-Fos+ cells were measured. Scale bar: 300 μm. (b, c) (left, middle) Representative images showing fighting (left) or mating (middle) induced c-Fos. (right) Distributions of the mediolateral distances of c-Fos+ cells after fighting (red) or mating (blue) in females (b, SW; c, C57BL/6N) at Bregma levels −1.4, −1.6, −1.8 and −2.0 mm (N = 3-7 animals). Dashed line indicates the median value of the mediolateral distance of each group. (d) The number of c-Fos+ cells per section in the VMHvl along the anterior-posterior axis (N = 3-7 animals). (e) The number of c-Fos+ cells in the anterior (Bregma level: −1.4 mm) and posterior VMHvl (Bregma level: −1.6mm to −2.0mm) after mating (blue, left) and fighting (red, right). Sections were occasionally not successfully collected resulting in different sample size at different Bregma level. Data are presented as means ± s.e.m. b, c, d: unpaired t-tests. e: paired t-test. *p < 0.05, **p < 0.01, ***p < 0.001.
Supplementary Figure 13 Neuronal staining revealed subcompartments in the female VMHvl.
The left four columns showing Nissl (white), Esr1 (red), and ZsGreen (green) of female Vgat-ires-Cre × Ai6 mice in the hypothalamus from Bregma level −1.2 to −2.0 mm. The rightmost column shows ZsGreen of a female Vglut2-ires-Cre × Ai6 mouse. The outlines of the VMHpvlm and VMHpvll are determined based solely on Nissl and then overlaid onto the Esr1 and ZsGreen images. Yellow arrows indicate the gap between the VMHpvlm and VMHpvll. Note that the VMHpvlm and pvll match the expression of Esr1 and Vglut2. Scale bars: 300 μm (first column) and 150 μm (second column).
Supplementary Figure 14 Projections of Esr1+ cells in the female VMHvl.
(a) Experimental scheme. (b) mCherry (red) expressing cells in the VMHvl and major terminal fields of VMHvl Esr1+ neurons at various brain regions. LS: lateral septum, AVPV: anteroventral periventricular nucleus, MPN: medial preoptic nucleus, PVH: paraventricular nucleus of hypothalamus, AHN: anterior hypothalamic nucleus, PVT: paraventricular thalamus, AMY: amygdala, SPFp: subparafascicular nucleus thalamus, parvicellular part. PAG: periaqueductal gray. Coronal brain atlas is from Allen Brain Atlas (http://www.brain-map.org). Scale bars: 300 μm
Supplementary Figure 15 The sexually dimorphic organization of aggression-related and mating-related populations in the VMHvl.
(a) Summary of all the functional manipulation experiments in our study. (b) Schematic illustrating topographic organization of aggression and mating related populations in the VMHvl of male and female mice. (c) Representative images showing fighting or mating induced c-Fos (green) in the VMHvl of SW male mice. Scale bar: 150 μm. (d) Spatial distributions of fighting and mating induced c-Fos in the male VMHvl are similar. The distributions of the mediolateral distances of c-Fos+ cells at different Bregma levels after mating (blue) or fighting (red) in males. Dashed line indicates the median value of the mediolateral distance of each group. Unpaired t-test. All p values above 0.1.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15, Supplementary Tables 1 and 2, and Supplementary Note 1 (PDF 5654 kb)
The VMHvl Esr1+ population in a virgin SW female did not respond to an object.
The GCaMP6f signal of Esr1+ neurons in the VMHvl of a female mouse was recorded using fiber photometry. There was little change in GCaMP6f signal when the female investigated a toy mouse. (WMV 4767 kb)
The VMHvl Esr1+ population in a virgin female responded during aggressive encounters with a juvenile male mouse.
GCaMP6f signal increased when the same female as shown in Supplementary Video 1 investigated or attacked a juvenile mouse. (WMV 3681 kb)
The VMHvl Esr1+ population in a virgin SW female responded during a sexual interaction with an adult male mouse.
GCaMP6f signal increased when the same female as shown in Supplementary Video 1 investigated or was engaged in sexual intercourse with an adult male mouse. (WMV 8611 kb)
The VMHvl Esr1+ population in a lactating SW female responded during aggressive encounters with an adult male mouse.
The GCaMP6f signal of Esr1+ neurons in the VMHvl of a lactating female mouse increased when the female attacked an adult male mouse. (WMV 9861 kb)
Optogenetic activation of VMHvl Esr1+ neurons elicited social investigation of and attack on an adult female in a virgin SW female mouse.
A SW female mouse that expressed ChR2 in Esr1+ neurons of the VMHvl was photostimulated through an optic fiber at 0.1, 0.4 or 1.5 mW, 20 Hz, 20-ms pulses. Light-induced behaviors progressed from investigation to attack as the light intensity increased. (WMV 7007 kb)
Optogenetic activation of VMHvl Esr1+ neurons elicited mounting towards an adult female and an adult male in a virgin C57 female mouse.
A C57 female mouse that expressed ChR2 in Esr1+ neurons of the VMHvl was photostimulated through an optic fiber at 20 Hz, 20 ms, 0.12 mW. The light induced mounting towards a female intruder, as well as a male intruder. (WMV 9067 kb)
Optogenetic activation of VMHvl Esr1+ neurons elicited attack on an adult female in a lactating C57 female mouse.
A lactating C57 female mouse (postpartum day 3) that expressed ChR2 in Esr1+ neurons of the VMHvl was photostimulated through an optic fiber at 20 Hz, 20 ms, 0.35 mW. Light induced an attack on a female intruder. (WMV 5098 kb)
Fight and mate populations in the VMHvl of a female mouse are largely distinct.
The 3D reconstruction of fighting- and mating-related populations in the VMHvl at all bregma levels of a female mouse used in a Fos catFISH experiment. Red, blue, and yellow dots indicate cells activated by fighting, mating and both, respectively. (WMV 4192 kb)
Rights and permissions
About this article
Cite this article
Hashikawa, K., Hashikawa, Y., Tremblay, R. et al. Esr1+ cells in the ventromedial hypothalamus control female aggression. Nat Neurosci 20, 1580–1590 (2017). https://doi.org/10.1038/nn.4644
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4644
This article is cited by
-
Unique transcriptional signatures correlate with behavioral and psychological symptom domains in Alzheimer’s disease
Translational Psychiatry (2024)
-
A dedicated hypothalamic oxytocin circuit controls aversive social learning
Nature (2024)
-
Independent inhibitory control mechanisms for aggressive motivation and action
Nature Neuroscience (2024)
-
Substance P in the medial amygdala regulates aggressive behaviors in male mice
Neuropsychopharmacology (2024)
-
Stress during pubertal development affects female sociosexual behavior in mice
Nature Communications (2024)