Abstract
The contribution of basal ganglia outputs to consummatory behavior remains poorly understood. We recorded from the substantia nigra pars reticulata (SNR), the major basal ganglia output nucleus, during self-initiated drinking in mice. The firing rates of many lateral SNR neurons were time-locked to individual licks. These neurons send GABAergic projections to the deep layers of the orofacial region of the lateral tectum (superior colliculus, SC). Many tectal neurons were also time-locked to licking, but their activity was usually in antiphase with that of SNR neurons, suggesting inhibitory nigrotectal projections. We used optogenetics to selectively activate the GABAergic nigrotectal afferents in the deep layers of the SC. Photo-stimulation of the nigrotectal projections transiently inhibited the activity of the lick-related tectal neurons, disrupted their licking-related oscillatory pattern and suppressed self-initiated drinking. These results demonstrate that GABAergic nigrotectal projections have a crucial role in coordinating drinking behavior.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Fowler, S.C. & Mortell, C. Low doses of haloperidol interfere with rat tongue extensions during licking: a quantitative analysis. Behav. Neurosci. 106, 386–395 (1992).
Nakamura, S., Muramatsu, S. & Yoshida, M. Role of the basal ganglia in manifestation of rhythmical jaw movement in rats. Brain Res. 535, 335–338 (1990).
Travers, J.B., Dinardo, L.A. & Karimnamazi, H. Motor and premotor mechanisms of licking. Neurosci. Biobehav. Rev. 21, 631–647 (1997).
Alexander, G.E. & Crutcher, M.D. Functional architecture of basal ganglia circuits: neural substrates of parallel processing. Trends Neurosci. 13, 266–271 (1990).
Yin, H.H., Ostlund, S.B. & Balleine, B.W. Reward-guided learning beyond dopamine in the nucleus accumbens: the integrative functions of cortico-basal ganglia networks. Eur. J. Neurosci. 28, 1437–1448 (2008).
Takakusaki, K., Saitoh, K., Harada, H. & Kashiwayanagi, M. Role of basal ganglia-brainstem pathways in the control of motor behaviors. Neurosci. Res. 50, 137–151 (2004).
Deniau, J.M. & Chevalier, G. The lamellar organization of the rat substantia nigra pars reticulata: distribution of projection neurons. Neuroscience 46, 361–377 (1992).
Beckstead, R.M. Long collateral branches of substantia nigra pars reticulata axons to thalamus, superior colliculus and reticular formation in monkey and cat. Multiple retrograde neuronal labeling with fluorescent dyes. Neuroscience 10, 767–779 (1983).
Taha, E.B., Dean, P. & Redgrave, P. Oral behaviour induced by intranigral muscimol is unaffected by haloperidol but abolished by large lesions of superior colliculus. Psychopharmacology (Berl.) 77, 272–278 (1982).
Redgrave, P., Dean, P., Donohoe, T.P. & Pope, S.G. Superior colliculus lesions selectively attenuate apomorphine-induced oral stereotypy: a possible role for the nigrotectal pathway. Brain Res. 196, 541–546 (1980).
Wang, S. & Redgrave, P. Microinjections of muscimol into lateral superior colliculus disrupt orienting and oral movements in the formalin model of pain. Neuroscience 81, 967–988 (1997).
Li, N., Chen, T.-W., Guo, Z.V., Gerfen, C.R. & Svoboda, K. A motor cortex circuit for motor planning and movement. Nature 519, 51–56 (2015).
Bjursten, L.M., Norrsell, K. & Norrsell, U. Behavioural repertory of cats without cerebral cortex from infancy. Exp. Brain Res. 25, 115–130 (1976).
Bignall, K.E. & Schramm, L. Behavior of chronically decerebrated kittens. Exp. Neurol. 42, 519–531 (1974).
Grill, H.J. Production and regulation of ingestive consummatory behavior in the chronic decerebrate rat. Brain Res. Bull. 5, 79–87 (1980).
Rossi, M.A. & Yin, H.H. Elevated dopamine alters consummatory pattern generation and increases behavioral variability during learning. Front. Integr. Neurosci. 9, 37 (2015).
Spector, A.C., Klumpp, P.A. & Kaplan, J.M. Analytical issues in the evaluation of food deprivation and sucrose concentration effects on the microstructure of licking behavior in the rat. Behav. Neurosci. 112, 678–694 (1998).
Weijnen, J.A. Licking behavior in the rat: measurement and situational control of licking frequency. Neurosci. Biobehav. Rev. 22, 751–760 (1998).
Gulley, J.M., Kosobud, A.E. & Rebec, G.V. Behavior-related modulation of substantia nigra pars reticulata neurons in rats performing a conditioned reinforcement task. Neuroscience 111, 337–349 (2002).
Kim, I.H. et al. Spine pruning drives antipsychotic-sensitive locomotion via circuit control of striatal dopamine. Nat. Neurosci. 18, 883–891 (2015).
Redgrave, P., Marrow, L. & Dean, P. Topographical organization of the nigrotectal projection in rat: evidence for segregated channels. Neuroscience 50, 571–595 (1992).
Chevalier, G., Deniau, J.M., Thierry, A.M. & Feger, J. The nigro-tectal pathway. An electrophysiological reinvestigation in the rat. Brain Res. 213, 253–263 (1981).
Hikosaka, O. & Wurtz, R.H. Visual and oculomotor functions of monkey substantia nigra pars reticulata. IV. Relation of substantia nigra to superior colliculus. J. Neurophysiol. 49, 1285–1301 (1983).
DeLong, M.R., Crutcher, M.D. & Georgopoulos, A.P. Relations between movement and single cell discharge in the substantia nigra of the behaving monkey. J. Neurosci. 3, 1599–1606 (1983).
Dräger, U.C. & Hubel, D.H. Topography of visual and somatosensory projections to mouse superior colliculus. J. Neurophysiol. 39, 91–101 (1976).
Tsumori, T., Ono, K., Kishi, T. & Yasui, Y. Demonstration of the corticotectobulbar pathway from the orofacial motor cortex to the parvicellular reticular formation in the rat. Brain Res. 755, 151–155 (1997).
Komiyama, T. et al. Learning-related fine-scale specificity imaged in motor cortex circuits of behaving mice. Nature 464, 1182–1186 (2010).
Rossi, M.A., Fan, D., Barter, J.W. & Yin, H.H. Bidirectional modulation of substantia nigra activity by motivational state. PLoS One 8, e71598 (2013).
Yasui, Y. et al. Descending projections from the superior colliculus to the reticular formation around the motor trigeminal nucleus and the parvicellular reticular formation of the medulla oblongata in the rat. Brain Res. 656, 420–426 (1994).
Tsumori, T. & Yasui, Y. Organization of the nigro-tecto-bulbar pathway to the parvicellular reticular formation: a light- and electron-microscopic study in the rat. Exp. Brain Res. 116, 341–350 (1997).
Travers, J.B., DiNardo, L.A. & Karimnamazi, H. Medullary reticular formation activity during ingestion and rejection in the awake rat. Exp. Brain Res. 130, 78–92 (2000).
Stanek, E. IV, Cheng, S., Takatoh, J., Han, B.X. & Wang, F. Monosynaptic premotor circuit tracing reveals neural substrates for oro-motor coordination. Elife 3, e02511 (2014).
Wiesenfeld, Z., Halpern, B.P. & Tapper, D.N. Licking behavior: evidence of hypoglossal oscillator. Science 196, 1122–1124 (1977).
Nakamura, Y., Enomoto, S. & Kato, M. The role of medial bulbar reticular neurons in the orbital cortically induced masticatory rhythm in cats. Brain Res. 202, 207–212 (1980).
Barter, J.W. et al. Basal ganglia outputs map instantaneous position coordinates during behavior. J. Neurosci. 35, 2703–2716 (2015).
Vong, L. et al. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71, 142–154 (2011).
Sparta, D.R. et al. Construction of implantable optical fibers for long-term optogenetic manipulation of neural circuits. Nat. Protoc. 7, 12–23 (2012).
Faingold, C.L. & Randall, M.E. Neurons in the deep layers of superior colliculus play a critical role in the neuronal network for audiogenic seizures: mechanisms for production of wild running behavior. Brain Res. 815, 250–258 (1999).
Faingold, C. & Casebeer, D. Modulation of the audiogenic seizure network by noradrenergic and glutamatergic receptors of the deep layers of superior colliculus. Brain Res. 821, 392–399 (1999).
Rossi, M.A., Sukharnikova, T., Hayrapetyan, V.Y., Yang, L. & Yin, H.H. Operant self-stimulation of dopamine neurons in the substantia nigra. PLoS One 8, e65799 (2013).
Kato, S. et al. A lentiviral strategy for highly efficient retrograde gene transfer by pseudotyping with fusion envelope glycoprotein. Hum. Gene Ther. 22, 197–206 (2011).
Nelson, A. et al. A circuit for motor cortical modulation of auditory cortical activity. J. Neurosci. 33, 14342–14353 (2013).
Rossi, M.A. et al. Prefrontal cortical mechanisms underlying delayed alternation in mice. J. Neurophysiol. 108, 1211–1222 (2012).
Paxinos, G. & Franklin, K. The Mouse Brain in Stereotaxic Coordinates (Academic, 2003).
Acknowledgements
We would like to thank F. Wang, L. Glickfeld, and G. Stuber for help with the head-fixed setup. This research is supported by US National Institutes of Health grant MH103374 (S.H.S.), AA021074 (H.H.Y.), and National Science Foundation graduate fellowships (M.A.R. and J.W.B.).
Author information
Authors and Affiliations
Contributions
M.A.R. and H.H.Y. conceived the study, analyzed the data and wrote the manuscript. M.A.R., H.E.L., D.L., R.A.B., E.G., J.W.B., N.K., and M.T.C. performed the behavioral experiments. M.A.R. and H.E.L. performed the electrophysiological recordings. I.H.K. and S.H.S. performed the viral tracing and imaging.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
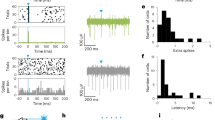
Supplementary Figure 1 Recording from SNR and SC.
(a-b) Photomicrographs showing electrode placement within the SNR (a) and SC (b) (left). Scale bars are 1 mm. Numbers indicate location relative to Bregma. An example of an isolated single unit (top right) and the population average (± s.e.m.) of all recorded units (bottom right). Arrowheads indicate electrode placement. The spread of electrode placements in SNR (c) and SC (d) is indicated by a single rectangle corresponding to each mouse. Scale bars are 1 mm. (e-f) Licking behavior is similar between SNR and SC mice. (e) Cumulative probability of lick durations. Inset is median lick duration (SNR: 4 mice two sessions each, 2 mice 3 sessions each; SC: 3 mice 3 sessions each, 1 mouse 2 sessions; Mann-Whitney U = 65.00, p = 0.53). (f) Cumulative probability of lick period. Inset is median period (SNR: 4 mice two sessions each, 2 mice 3 sessions each; SC: 3 mice 3 sessions each, 1 mouse 2 sessions; Mann-Whitney U = 72.50, p = 0.38). Values are mean ±sem.
Supplementary Figure 2 SNR and SC units reflect the boundaries of licking bouts.
(a) Spike density functions of bout-related activity. Neurons that decreased firing during a licking bout increased firing after the bout ended in both SNR and SC. Neurons that increased firing during a licking bout decreased firing after the bout ended. (b) Average (mean±s.e.m.) response of 'increasing' neurons. (c) Average (± s.e.m.) response of 'decreasing' neurons.
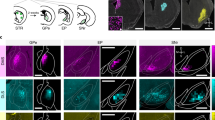
Supplementary Figure 3 Excitotoxic lesions of lateral SC disrupt lick patterning.
NMDA (n = 6 mice) or PBS (n = 6 mice) were injected into the lateral SC. (a) Representative photomicrograph a lesioned mouse. The white line indicates the SC, the black line indicates the lesioned area. Lesions were observed in all mice of the lesion group. (b) The extent of the largest lesion is shown in gray and the smallest in black. Numbers indicate location relative to Bregma. Scale is 1 mm. (c) Example lickometer traces of NMDA- and sham-lesioned mice. Upward deflections represent licks. (d) The period was unchanged (t(10) = 0.79, p = 0.45). (e) SC lesions increased the duration of licks (t(10) = 2.77, * p = 0.02). Values are mean ± s.e.m.
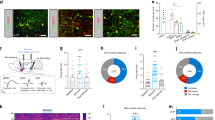
Supplementary Figure 4 Vgat::ChR2SNR→SC fiber and injection placements.
(a) Fiber placements (circles) and virus injection sites (triangles) are shown for ChR2 (blue symbols) and eYFP (green symbols) mice used in optogenetics experiments. Scale bar is 1 mm. (b) Fluorescence intensity was quantified within the SNR, lateral SC, and medial SC (one way ANOVA (n = 7 mice, 2 hemispheres each): F(2,26) = 22.24, p = 6.36 x 10-5, Tukey's multiple comparison test medial SC vs. SNR: *** p = 8.92 x 10-4; lateral SC vs medial SC: ** p = 2.83 x 10-4). Values are mean ±sem.
Supplementary Figure 5 Vgat::ChR2SNR→SC stimulation disrupts lick bouts without affecting overall lick rate.
(a-c) Vgat::ChR2SNR→SC changes bout characteristics without altering overall lick rate. (a) The rate of licking was not affected by stimulation (one-way ANOVA: F(6,36) = 1.50, p = 0.21). (b) The number of bouts was increased by constant illumination (F(6,36) = 5.91, p = 0.0002). (c) The number of licks per bout was decreased by constant illumination (F(6,36) = 2.45, p = 0.04). (d) Stimulation did not affect lick duration (F(6,36) = 1.13, p = 0.36). (e) Stimulation increased the coefficient of variation (CV) of lick duration (F(6,36) = 3.86, p = 0.0045). (f) Probability of pause for early and late trails (two-way ANOVA: main effect of Frequency, F(6,42) = 8.18, p = 6.90 x 10-6; main effect of Motivation, F(1,42) = 19.23, p = 7.62 x 10-5, interaction between Frequency and Motivation, F(6,42) = 3.97, p = 0.0031); Bonferroni post test comparing early vs late trials: 1 s On * p = 0.026, and 2 s On ** p = 0.0015 conditions). Values are mean ± sem. n = 7 mice.
Supplementary Figure 6 Vgat::YFPSNR→SC stimulation does not affect consummatory behavior.
(a) Population lick rate (mean±s.e.m.) aligned to the onset of stimulation for Vgat::YFPSNR→SC control mice (n = 6). (b) Lick rate is unaffected by stimulation (one-way ANOVA: F(6,30) = 1.59, p = 0.18). Values are mean and s.e.m. (c) Example lick rasters from a single Vgat::YFPSNR→SC mouse under no stimulation (left), 1 s (middle) or 2 s (right) constant illumination. Each row represents one trial. Average histogram is on the bottom.
Supplementary Figure 7 Vgat::ChR2SNR→SC stimulation does not cause significant head movements.
The speed of the mouse's head was tracked at 100 frames/s using high-resolution motion tracking. (a-b) Normalized head speed under different stimulation frequencies for (a) Vgat::ChR2SNR→SC (n = 7 mice) and (b) Vgat::YFPSNR→SC (n = 4) aligned to onset of stimulation. (c) Stimulation did not significantly affect head speed (two-way repeated measures ANOVA: no main effect of Group, F(1,36) = 2.66, p = 0.13; no main effect of Frequency, F(4,36) = 1.56, p = 0.20, no interaction between Group and Frequency F(4,36) = 0.71, p = 0.59). Values are mean ±s.e.m.
Supplementary Figure 8 Vgat::ChR2SNR→SC stimulation does not affect whisking or blinking.
(a) Stimulation reduced the lick rate in a frequency dependent manner in headfixed mice (n = 5) but had no effect on voluntary licking in control mice (n = 3). (b) Lick rate during stimulation (two-way ANOVA [Group x Stimulation Frequency]: main effect of Group, F (1, 30) = 32.42, p = 0.0013; main effect of Stimulation Frequency, F (5, 30) = 3.01, p = 0.0256; no interaction between Group and Stimulation Frequency, F (5, 30) = 1.669, p = 0.1726. (c) The probability of whisking was unaffected by stimulation: no main effect of Group, F (1, 30) = 0.7483, p = 0.4203; no main effect of Stimulation Frequency, F (5, 30) = 2.35, p = 0.065; no interaction between Group and Stimulation Frequency, F (5, 30) = 1.395, p = 0.2542. (d) Blink rate was unaffected by Vgat::ChR2SNR→SC stimulation: no main effect of Group, F (1, 30) = 0.3818, p = 0.5594; no main effect of Stimulation Frequency, F (5, 30) = 0.2852, p = 0.9175; no interaction between Group and Stimulation Frequency, F (5, 30) = 0.5059, p = 0.7694. Values are mean and s.e.m.
Supplementary Figure 9 Histological verification of nigro-mRF stimulation.
(a) YFP-expressing neurons do not overlap with TH-expressing neurons. Results were replicated with 5 mice. (b) Fiber placements within the mesencephalic reticular formation. (c) ChR2 injection sites within the SNR. Blue circles represent ChR2-YFP mice (n = 6), green circles represent YFP control mice (n = 5). Scale bars are 1 mm. Approximate coordinates are given in mm relative to Bregma.
Supplementary Figure 10 Optogenetic activation of nigro-mRF projections does not significantly alter consummatory behavior.
(a) Population lick rate (mean±s.e.m.) aligned to the onset of stimulation for Vgat::ChR2SNR→mRF mice (n = 6). (b) Vgat::ChR2SNR→mRF stimulation caused a marginally significant reduction in the lick rate (one way ANOVA: F(6,30) = 2.26, p = 0.06). Values are mean and s.e.m. (c) Example lick rasters from a single Vgat::ChR2SNR→mRF mouse under no stimulation (left), 1 s (middle) or 2 s (right) constant illumination. Each row represents one trial. (d-f) Vgat::YFPSNR→mRF (n = 5) stimulation did not affect consummatory drinking. (d) Population lick rate (mean±s.e.m.) aligned to the onset of stimulation for Vgat::YFPSNR→mRF mice. (e) Vgat::YFPSNR→mRF stimulation did not alter the lick rate (F(6,24) = 1.46, p = 0.23). Values are mean and s.e.m. (f) Example lick rasters from a single Vgat::YFPSNR→mRF mouse under no stimulation (left), 1 s (middle) or 2 s (right) constant illumination. Each row represents one trial. Average histogram is on the bottom.
Supplementary Figure 11 Vgat::ChR2SNR→SC stimulation inhibits SC firing during non-consummatory behavior.
(a) Example rasters of a single unit recorded from SC during stimulation when mice were not licking. 11-12 trials per condition in randomized order. Each row represents one trial, average response is on the bottom. Shaded region indicated stimulation epoch. (b) Population activity (mean and s.e.m.) of 18 single units recorded from 3 mice during stimulation (one way ANOVAL F(6,102) = 9.14, p = 5.36 x 10-8; Dunnett's multiple comparison test vs. Baseline: SC activity is suppressed by 50 Hz (*p = 0.033), 100 Hz (**p = 0.021), 1 s On (***p = 0.012), and 2 s On (****p = 0.025) conditions).
Supplementary Figure 12 Vgat::ChR2SNR→SC activation disrupts SC oscillatory activity.
(a) Example neuron that shows oscillatory activity that is antiphase to the lick cycle during the baseline period. When 1 s of constant illumination occurs during licking, consequently disrupting the pattern of consummatory behavior, the oscillatory activity is suppressed. Waveforms recorded during baseline and stimulation are shown at the right (median waveform is in red). The waveform shape was not affected by stimulation. (b) Population response (7 single units from 3 mice) of neurons that paused at the time of spout contact during the baseline period. During stimulation trials, the oscillatory activity is suppressed. Values are mean and s.e.m.
Supplementary Figure 13 SC oscillatory activity is disrupted by Vgat::ChR2SNR→SC stimulation.
(a-g) Spike density functions of neurons recorded in the lateral SC. Activity is aligned to lick start. Each row represents the activity of one neuron. (a) Antiphase oscillatory activity is present under baseline conditions in which no stimulation is delivered (n = 28 neurons from 3 mice). Photo-stimulation of the nigrotectal projections disrupted the oscillatory activity. (b-g) These neurons showed antiphase oscillatory activity during baseline conditions. (h) The amplitude of oscillations (the difference between the maximum and minimum firing rates within the lick cycle) was reduced by Vgat::ChR2SNR→SC stimulation. Planned comparison Mann-Whitney tests were performed between Baseline and 3 Hz (U = 100.00, p = 0.66, 8 neurons from 3 mice), 10 Hz (U = 67.00, p = 0.09, 8 neurons from 3 mice), 50 Hz (U = 96.00, p = 0.56, 8 neurons from 3 mice), 100 Hz (U = 52.00, *p = 0.02, 8 neurons from 3 mice), 1 s On (U = 42.00, **p = 0.02, 7 neurons from 3 mice), and 2 s On (U = 67.00, p = 0.90, 5 neurons from 3 mice). Values are mean and s.e.m. (i-k) Vgat::ChR2SNR→SC stimulation disrupts the phase relationship between licking and SC activity. (i) Peak response within the lick cycle as a function of stimulation. Values are median. (j) Distribution of peak responses under no stimulation (BL) and stimulation conditions in which the lick rate was significantly affected. Responses from 50 Hz, 100 Hz, 1 s On, and 2 s On conditions are pooled. (k) Proportion of neurons showing peak activity at 90 ͦ and 180 ͦ in the lick cycle. Under baseline conditions more neurons are 180 ͦ out of phase (46%, 13/28 from 3 mice) than 90 ͦ out of phase (25%, 7/2 from 3 mice 8). In contrast, during stimulation more neurons are 90 ͦ out of phase (50%, 14/28 from 3 mice) than 180 ͦ out of phase (21%, 6/28 neurons from 3 mice; χ2 = 4.91, p = 0.03).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13 (PDF 3303 kb)
Muscimol suppresses licking.
Following infusion of 1.0 μg/μl muscimol into the lateral SC, mice cannot initiate licking. (MP4 1187 kb)
Vehicle control.
Following infusion of vehicle into the lateral SC, mice readily lick for sucrose solution. (MP4 3196 kb)
Activation of nigrotectal pathway suppresses drinking.
Mice are unable to lick for sucrose solution during optogenetic activation of the nigrotectal pathway. Constant laser illumination indicated by 'Laser' in the top left corner. (MP4 7561 kb)
Activation of nigrotectal pathway does not affect other orofacial behaviors.
Optogenetic activation of the nigrotectal pathway does not influence whisking or blinking. This is the same mouse as Video 3. Constant laser illumination indicated by 'Laser' in the top left corner. (MP4 5076 kb)
Rights and permissions
About this article
Cite this article
Rossi, M., Li, H., Lu, D. et al. A GABAergic nigrotectal pathway for coordination of drinking behavior. Nat Neurosci 19, 742–748 (2016). https://doi.org/10.1038/nn.4285
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.4285
This article is cited by
-
Basal ganglia–spinal cord pathway that commands locomotor gait asymmetries in mice
Nature Neuroscience (2024)
-
Superior colliculus bidirectionally modulates choice activity in frontal cortex
Nature Communications (2023)
-
Activity map of a cortico-cerebellar loop underlying motor planning
Nature Neuroscience (2023)
-
Influence of Recent Trial History on Interval Timing
Neuroscience Bulletin (2023)
-
A behavioural correlate of the synaptic eligibility trace in the nucleus accumbens
Scientific Reports (2022)