Abstract
Robo-Slit and Plexin-Semaphorin signaling participate in various developmental and pathogenic processes. During commissural axon guidance in the spinal cord, chemorepulsion by Semaphorin3B and Slits controls midline crossing. Slit processing generates an N-terminal fragment (SlitN) that binds to Robo1 and Robo2 receptors and mediates Slit repulsive activity, as well as a C-terminal fragment (SlitC) with an unknown receptor and bioactivity. We identified PlexinA1 as a Slit receptor and found that it binds the C-terminal Slit fragment specifically and transduces a SlitC signal independently of the Robos and the Neuropilins. PlexinA1–SlitC complexes are detected in spinal cord extracts, and ex vivo, SlitC binding to PlexinA1 elicits a repulsive commissural response. Analysis of various ligand and receptor knockout mice shows that PlexinA1-Slit and Robo-Slit signaling have complementary roles during commissural axon guidance. Thus, PlexinA1 mediates both Semaphorin and Slit signaling, and Slit processing generates two active fragments, each exerting distinct effects through specific receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Raper, J. & Mason, C. Cellular strategies of axonal pathfinding. Cold Spring Harb. Perspect. Biol. 2, a001933 (2010).
Bashaw, G.J. & Klein, R. Signaling from axon guidance receptors. Cold Spring Harb. Perspect. Biol. 2, a001941 (2010).
Dickson, B.J. & Gilestro, G.F. Regulation of commissural axon pathfinding by slit and its Robo receptors. Annu. Rev. Cell Dev. Biol. 22, 651–675 (2006).
Evans, T.A. & Bashaw, G.J. Axon guidance at the midline: of mice and flies. Curr. Opin. Neurobiol. 20, 79–85 (2010).
Nawabi, H. & Castellani, V. Axonal commissures in the central nervous system: how to cross the midline? Cell. Mol. Life Sci. 68, 2539–2553 (2011).
Zou, Y., Stoeckli, E., Chen, H. & Tessier-Lavigne, M. Squeezing axons out of the gray matter: a role for slit and semaphorin proteins from midline and ventral spinal cord. Cell 102, 363–375 (2000).
Nawabi, H. et al. A midline switch of receptor processing regulates commissural axon guidance in vertebrates. Genes Dev. 24, 396–410 (2010).
Charoy, C. et al. Gdnf activates midline repulsion by Semaphorin3B via NCAM during commissural axon guidance. Neuron 75, 1051–1066 (2012).
Kidd, T., Bland, K.S. & Goodman, C.S. Slit is the midline repellent for the robo receptor in Drosophila. Cell 96, 785–794 (1999).
Kidd, T., Russell, C., Goodman, C.S. & Tear, G. Dosage-sensitive and complementary functions of roundabout and commissureless control axon crossing of the CNS midline. Neuron 20, 25–33 (1998).
Seeger, M., Tear, G., Ferres-Marco, D. & Goodman, C.S. Mutations affecting growth cone guidance in Drosophila: genes necessary for guidance toward or away from the midline. Neuron 10, 409–426 (1993).
Long, H. et al. Conserved roles for Slit and Robo proteins in midline commissural axon guidance. Neuron 42, 213–223 (2004).
Brose, K. et al. Slit proteins bind Robo receptors and have an evolutionarily conserved role in repulsive axon guidance. Cell 96, 795–806 (1999).
Chen, J.H., Wen, L., Dupuis, S., Wu, J.Y. & Rao, Y. The N-terminal leucine-rich regions in Slit are sufficient to repel olfactory bulb axons and subventricular zone neurons. J. Neurosci. 21, 1548–1556 (2001).
Nguyen Ba-Charvet, K.T. et al. Diversity and specificity of actions of Slit2 proteolytic fragments in axon guidance. J. Neurosci. 21, 4281–4289 (2001).
Chédotal, A. Slits and their receptors. Adv. Exp. Med. Biol. 621, 65–80 (2007).
Jaworski, A., Long, H. & Tessier-Lavigne, M. Collaborative and specialized functions of Robo1 and Robo2 in spinal commissural axon guidance. J. Neurosci. 30, 9445–9453 (2010).
Toyofuku, T. et al. Dual roles of Sema6D in cardiac morphogenesis through region-specific association of its receptor, Plexin-A1, with off-track and vascular endothelial growth factor receptor type 2. Genes Dev. 18, 435–447 (2004).
Chen, Z., Gore, B.B., Long, H., Ma, L. & Tessier-Lavigne, M. Alternative splicing of the Robo3 axon guidance receptor governs the midline switch from attraction to repulsion. Neuron 58, 325–332 (2008).
Falk, J. et al. Dual functional activity of semaphorin 3B is required for positioning the anterior commissure. Neuron 48, 63–75 (2005).
Giordano, S. et al. The semaphorin 4D receptor controls invasive growth by coupling with Met. Nat. Cell Biol. 4, 720–724 (2002).
Swiercz, J.M., Kuner, R. & Offermanns, S. Plexin-B1/RhoGEF-mediated RhoA activation involves the receptor tyrosine kinase ErbB-2. J. Cell Biol. 165, 869–880 (2004).
Mitsui, N. et al. Involvement of Fes/Fps tyrosine kinase in semaphorin3A signaling. EMBO J. 21, 3274–3285 (2002).
Blom, N., Gammeltoft, S. & Brunak, S. Sequence and structure-based prediction of eukaryotic protein phosphorylation sites. J. Mol. Biol. 294, 1351–1362 (1999).
Toyofuku, T. et al. FARP2 triggers signals for Sema3A-mediated axonal repulsion. Nat. Neurosci. 8, 1712–1719 (2005).
Turner, L.J., Nicholls, S. & Hall, A. The activity of the plexin-A1 receptor is regulated by Rac. J. Biol. Chem. 279, 33199–33205 (2004).
Briançon-Marjollet, A. et al. Trio mediates netrin-1–induced Rac1 activation in axon outgrowth and guidance. Mol. Cell. Biol. 28, 2314–2323 (2008).
Yang, L. & Bashaw, G.J. Son of sevenless directly links the Robo receptor to rac activation to control axon repulsion at the midline. Neuron 52, 595–607 (2006).
Vikis, H.G., Li, W. & Guan, K.-L. The plexin-B1/Rac interaction inhibits PAK activation and enhances Sema4D ligand binding. Genes Dev. 16, 836–845 (2002).
Kruger, R.P., Aurandt, J. & Guan, K.-L. Semaphorins command cells to move. Nat. Rev. Mol. Cell Biol. 6, 789–800 (2005).
Unni, D.K. et al. Multiple Slits regulate the development of midline glial populations and the corpus callosum. Dev. Biol. 365, 36–49 (2012).
Cho, J.H., Kam, J.W.K. & Cloutier, J.-F. Slits and Robo-2 regulate the coalescence of subsets of olfactory sensory neuron axons within the ventral region of the olfactory bulb. Dev. Biol. 371, 269–279 (2012).
Johnson, K.G. et al. Axonal heparan sulfate proteoglycans regulate the distribution and efficiency of the repellent slit during midline axon guidance. Curr. Biol. 14, 499–504 (2004).
Xiao, T. et al. Assembly of lamina-specific neuronal connections by slit bound to type IV collagen. Cell 146, 164–176 (2011).
Wright, K.M. et al. Dystroglycan organizes axon guidance cue localization and axonal pathfinding. Neuron 76, 931–944 (2012).
Philipp, M. et al. RabGDI controls axonal midline crossing by regulating Robo1 surface expression. Neural Dev. 7, 36 (2012).
Whitford, K.L. et al. Regulation of cortical dendrite development by Slit-Robo interactions. Neuron 33, 47–61 (2002).
Mehlen, P., Delloye-Bourgeois, C. & Chédotal, A. Novel roles for Slits and netrins: axon guidance cues as anticancer targets? Nat. Rev. Cancer 11, 188–197 (2011).
Gu, C. & Giraudo, E. The role of semaphorins and their receptors in vascular development and cancer. Exp. Cell Res. 319, 1306–1316 (2013).
Marquardt, T. et al. Coexpressed EphA receptors and ephrin-A ligands mediate opposing actions on growth cone navigation from distinct membrane domains. Cell 121, 127–139 (2005).
Yoshida, Y., Han, B., Mendelsohn, M. & Jessell, T.M. PlexinA1 signaling directs the segregation of proprioceptive sensory axons in the developing spinal cord. Neuron 52, 775–788 (2006).
Plump, A.S. et al. Slit1 and Slit2 cooperate to prevent premature midline crossing of retinal axons in the mouse visual system. Neuron 33, 219–232 (2002).
Yuan, W. et al. A genetic model for a central (septum transversum) congenital diaphragmatic hernia in mice lacking Slit3. Proc. Natl. Acad. Sci. USA 100, 5217–5222 (2003).
Giger, R.J. et al. Neuropilin-2 is required in vivo for selective axon guidance responses to secreted semaphorins. Neuron 25, 29–41 (2000).
Rohm, B., Ottemeyer, A., Lohrum, M. & Püschel, A.W. Plexin/neuropilin complexes mediate repulsion by the axonal guidance signal semaphorin 3A. Mech. Dev. 93, 95–104 (2000).
Zhao, Y. et al. Automation of large scale transient protein expression in mammalian cells. J. Struct. Biol. 175, 209–215 (2011).
Creuzet, S., Couly, G., Vincent, C. & Le Douarin, N.M. Negative effect of Hox gene expression on the development of the neural crest-derived facial skeleton. Development 129, 4301–4313 (2002).
Blank, M.C., Chizhikov, V. & Millen, K.J. In ovo electroporations of HH stage 10 chicken embryos. J. Vis. Exp. 408 (2007).
Bechara, A. et al. FAK-MAPK–dependent adhesion disassembly downstream of L1 contributes to semaphorin3A-induced collapse. EMBO J. 27, 1549–1562 (2008).
Acknowledgements
V.C. is supported by grants from the French National Research Agency (ANR-2010-BLANC-1430-01) and the European Research Council under the European Union's Seventh Framework Programme (FP/2007-2013)/ERC Grant Agreement 281604-YODA. C.D.-B. is supported by a post-doc fellowship from the 'Ligue Contre le cancer'. This work was performed within the framework of the LABEX CORTEX and the LABEX DevWeCan of Université de Lyon within the program 'Investissements d'Avenir' (ANR-11-IDEX-0007) operated by the French National Research Agency (ANR). A.C. is supported by grants from the Fondation pour la Recherche Médicale (Programme 'équipe FRM') and the Agence Nationale de la Recherche (ANR-08-MNP-030) to A.C. We acknowledge M. Tessier-Lavigne (Laboratory of Brain Development and Repair, Rockefeller University) and A.B. Huber (Institute for Developmental Genetics, GSF-Research Center for Environment and Health) for sharing mouse lines. We thank T. Toyofuku (Osaka University) and A. Püschel (Abt. Molekularbiologie, Institut für Allgemeine Zoologie und Genetik, Westfälische Wilhelms-Universität) for sharing plasmids.
Author information
Authors and Affiliations
Contributions
C.D.-B. designed and conducted the majority of experiments. A.J. constructed expression vectors and participated in the experiments in Figure 7. C.C., F.R., H.N., K.T., K.K. and J.F. contributed to experiments shown in the figures and performed genotyping. Y.Y. provided Plxna1 embryos and mouse line. Y.K. and Y.E.J. contributed to the binding experiments and purified Slit2C protein. Y.Z. and A.C. provided various constructs and mouse lines and validated various antibodies. A.C. brought scientific input and advice for manuscript preparation. V.C. designed the experimental plan, supervised the project and wrote the manuscript with C.D.-B.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
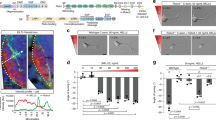
Supplementary Figure 2 In ovo electroporation of dominant-negative PlexinA1 in commissural neurons induces midline guidance defects.
a, b. Microphotographs of chick open-books (a) and quantitative analysis (b) after control/GFP or PlexinA-DN/GFP co-electroporations (Ctl; n = 8 embryos, 107 fields; PlexA-DN; n = 8 embryos, 107 fields). Abnormal turn (*) and recrossing (arrowhead) phenotypes are outlined. Scale bar: 25 μM. c, d. Immunofluorescent labeling (c) and quantification of grey matter entry phenotype in chick open books (d) co-electroporated with Ctl/GFP (n = 7 embryos) or PlexA-DN/GFP (n = 8 embryos). c shows GFP fluorescence, Robo3 and Hoechst staining. Lower panels are grayscale enlargements of GFP fluorescence. Arrowheads point axons abnormally entering the grey matter. Scale bar: 100 μM. For b and d, error bars indicate mean + SEM; *p<0.05; **p<0.01; ***p<0.001; Student t-test.
Supplementary Figure 3 SlitC interacts with PlexinA1 in vitro and in vivo.
a. Co-immunoprecipitation performed in COS7 cells transfected with Flag-tagged PlexinA1 construct together with Myc-tagged Slit1 or Slit2. b. Scheme of PlexinA1 protein, and PlexinA1 fragment used to map interaction domain with Slit PlexinA1IPT1-4 and PlexinA1IPT3-4. Sema: Semaphorin domain; PSI: Plexin, Semaphorin and Integrin domain; IPT: Ig-like, Plexin and Transcription factor domain; TM: Transmembrane domain; C1/2: Segmented GAP domains. c. Co-immunoprecipitations in COS7 cells transfected with Flag-tagged PlexinA1, PlexinA1IPT1-4 or PlexinA1IPT3-4 or together with Myc-tagged Slit2C-AP construct. d. Co-immunoprecipitation performed in E12.5 spinal cords using a Slit2C immunoprecipitating antibody. 2 samples for each condition are presented (s1 and s2). In Slit2C immunoprecipitate, the lower band corresponds to the immunoglobulin heavy chain. e. Co-immunoprecipitation performed in spinal cords from E12.5 Robo1/2+/+ and Robo1/2—/— embryos using a PlexinA1 immunoprecipitating antibody. PlexinA1 and Slit1 are detected using antibodies directed against PlexinA1 and a C-terminal Slit1 epitope respectively. For Slit1 immunoblot, two different exposures are shown, to detect both the full-length protein (10’) and the C-terminal cleavage fragment (10’’), pointed by arrowhead.
Supplementary Figure 4 Experimental settings and PlexinA1 status at the growth cone surface in collapse assays.
Schemes of the experimental settings (left panels) and PlexinA1 surface status. Right panels: microphotographs of Hoechst and phalloidin labeling of cultured commissural neurons, with enlarged panels illustrating collapsed and not collapsed observations. Scale bar: 15 μm.
Supplementary Figure 5 PlexinA1 mediates a SlitC collapse response independently of the Robos.
a. In vitro validation of PlexinA1 siRNA efficiency. Western Blot analysis of PlexinA1 protein expression was performed on COS7 cells transfected with PlexinA1-Flag expressing construct together with scramble siRNA or PlexinA1 siRNA. b. Collapse assay on E12.5 PlexinA1 wild-type (+/+), heterozygous (+/—), and null (—/—) commissural neurons incubated with Slit2-FL supernatant or Slit2C supernatant alone or together with GDNF. c. Collapse assay performed on E12.5 Robo1/2 wild-type (+/+) and null (—/—) commissural neurons incubated with Slit1C supernatant alone or together with GDNF. For b and c, n = 4 experiments per condition; number of analyzed growth cones per condition > 200; graphs show mean collapse rate + SD; ***p<0.001; Student t-test. d. Collapse assay on E12.5 commissural neurons incubated with increasing concentrations of Slit2 alone or together with GDNF (n = 3 experiments per condition; number of analyzed growth cones per condition > 100). Graph shows mean collapse rate + SD; *p<0.05, Mann-Whitney U-test.
Supplementary Figure 6 SlitC triggers activation of Rac1 and phosphorylation of ERK.
a. Rac1 activation assay performed on HEK293T cells transfected with PlexinA1 and incubated with indicated supernatants for 15 min. Rac1 active form (bound to GTP) is pulled-down with a GST-CRIB (Cdc42/Rac Interactive Binding) fusion protein. Lower diagram shows quantification of Rac1-GTP/total Rac1 ratio. Graph shows mean + SD; *p<0.05; Mann-Whitney test; 3 independent experiments. b. Time course analysis of ERK phosphorylation in HEK293T cells transfected with PlexinA1 and incubated with supernatants from cells transfected with vectors encoding Slit1-FL, Slit2-FL, Slit2N or Slit2C. Western Blot analysis shows detection of both phosphorylated and total p42 and p44 forms.
Supplementary Figure 7 Model of spatiotemporal engagement of receptors for midline repellents during floor plate crossing.
In the FP Slit is processed into N-terminal (SlitN) and C-terminal (SlitC) fragments. PlexinA1 is processed by calpain in precrossing commissural growth cones. Upon crossing, FP-released GDNF suppresses calpain1 activity, triggering PlexinA1 availability at the growth cone surface. Thus, from the crossing stage, two independent Slit signaling, PlexA1/SlitC and Robo/SlitN are triggered to mediate complementary effects of Slits midline repellents. In parallel, PlexinA1/Nrp2 complexes process the repulsive Sema3B signaling.
Supplementary Figure 9 Full-length pictures of the blots presented as cropped panels in main-text Figure 7.
In Fig. 7h, the same membrane was used to detect PlexinA1 (α-Flag) and Nrp2 (α-HA) successively,
Supplementary Figure 10 Western blot validation of antibodies used in the study.
a-d: Western blot analysis of E12.5 embryos tissue lysates from wild-type, Slit2—/— (a), Robo1/2—/— (b, c) and Nrp2—/— (d) using Slit2C (a, Epitomics 2864-1), Robo1 (b, abcam ab85312), Robo2 (c, abcam ab64158) and Nrp2 (d, R&D AF567) antibodies. Arrowheads point predicted endogenous protein size.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10 (PDF 3069 kb)
Rights and permissions
About this article
Cite this article
Delloye-Bourgeois, C., Jacquier, A., Charoy, C. et al. PlexinA1 is a new Slit receptor and mediates axon guidance function of Slit C-terminal fragments. Nat Neurosci 18, 36–45 (2015). https://doi.org/10.1038/nn.3893
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3893
This article is cited by
-
Guidance landscapes unveiled by quantitative proteomics to control reinnervation in adult visual system
Nature Communications (2022)
-
The behavior and functions of embryonic microglia
Anatomical Science International (2022)
-
Manipulating oligodendrocyte intrinsic regeneration mechanism to promote remyelination
Cellular and Molecular Life Sciences (2021)
-
Transient microglial absence assists postmigratory cortical neurons in proper differentiation
Nature Communications (2020)
-
JNK-mediated Slit-Robo signaling facilitates epithelial wound repair by extruding dying cells
Scientific Reports (2019)