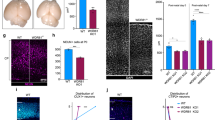
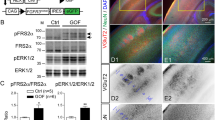
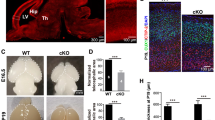
Abstract
Neuronal migration disorders such as lissencephaly and subcortical band heterotopia are associated with epilepsy and intellectual disability. DCX, PAFAH1B1 and TUBA1A are mutated in these disorders; however, corresponding mouse mutants do not show heterotopic neurons in the neocortex. In contrast, spontaneously arisen HeCo mice display this phenotype, and our study revealed that misplaced apical progenitors contribute to heterotopia formation. While HeCo neurons migrated at the same speed as wild type, abnormally distributed dividing progenitors were found throughout the cortical wall from embryonic day 13. We identified Eml1, encoding a microtubule-associated protein, as the gene mutated in HeCo mice. Full-length transcripts were lacking as a result of a retrotransposon insertion in an intron. Eml1 knockdown mimicked the HeCo progenitor phenotype and reexpression rescued it. We further found EML1 to be mutated in ribbon-like heterotopia in humans. Our data link abnormal spindle orientations, ectopic progenitors and severe heterotopia in mouse and human.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
Primary accessions
Gene Expression Omnibus
NCBI Reference Sequence
Referenced accessions
NCBI Reference Sequence
References
Harding, B. Gray matter heterotopia. in Dysplasias of Cerebral Cortex and Epilepsy, (eds. Guerrini, R. et al.) 81–88 (Lippincott-Raven, Philadelphia, 1996).
Jaglin, X.H. & Chelly, J. Tubulin-related cortical dysgeneses: microtubule dysfunction underlying neuronal migration defects. Trends Genet. 25, 555–566 (2009).
Hirotsune, S. et al. Graded reduction of Pafah1b1 (Lis1) activity results in neuronal migration defects and early embryonic lethality. Nat. Genet. 19, 333–339 (1998).
Corbo, J.C. et al. Doublecortin is required in mice for lamination of the hippocampus but not the neocortex. J. Neurosci. 22, 7548–7557 (2002).
Kappeler, C. et al. Magnetic resonance imaging and histological studies of corpus callosal and hippocampal abnormalities linked to doublecortin deficiency. J. Comp. Neurol. 500, 239–254 (2007).
Keays, D.A. et al. Mutations in alpha-tubulin cause abnormal neuronal migration in mice and lissencephaly in humans. Cell 128, 45–57 (2007).
Bai, J. et al. RNAi reveals doublecortin is required for radial migration in rat neocortex. Nat. Neurosci. 6, 1277–1283 (2003).
Lee, K.S. et al. A genetic animal model of human neocortical heterotopia associated with seizures. J. Neurosci. 17, 6236–6242 (1997).
Munji, R.N., Choe, Y., Li, G., Siegenthaler, J.A. & Pleasure, S.J. Wnt signaling regulates neuronal differentiation of cortical intermediate progenitors. J. Neurosci. 31, 1676–1687 (2011).
Bilasy, S.E. et al. Dorsal telencephalon–specific RA-GEF-1 knockout mice develop heterotopic cortical mass and commissural fiber defect. Eur. J. Neurosci. 29, 1994–2008 (2009).
Cappello, S. et al. A radial glia–specific role of RhoA in double cortex formation. Neuron 73, 911–924 (2012).
Croquelois, A. et al. Characterization of the HeCo mutant mouse: a new model of subcortical band heterotopia associated with seizures and behavioral deficits. Cereb. Cortex 19, 563–575 (2009).
Rosen, G.D. et al. Bilateral subcortical heterotopia with partial callosal agenesis in a mouse mutant. Cereb. Cortex 23, 859–872 (2013).
Barkovich, A.J. et al. A developmental and genetic classification for malformations of cortical development: update 2012. Brain 135, 1348–1369 (2012).
Konno, D. et al. Neuroepithelial progenitors undergo LGN-dependent planar divisions to maintain self-renewability during mammalian neurogenesis. Nat. Cell Biol. 10, 93–101 (2008).
Chenn, A. & Walsh, C.A. Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science 297, 365–369 (2002).
Baust, C. et al. Structure and expression of mobile ETnII retroelements and their coding-competent MusD relatives in the mouse. J. Virol. 77, 11448–11458 (2003).
Welker, E. et al. Altered sensory processing in the somatosensory cortex of the mouse mutant barrelless. Science 271, 1864–1867 (1996).
Abdel-Majid, R.M. et al. Loss of adenylyl cyclase I activity disrupts patterning of mouse somatosensory cortex. Nat. Genet. 19, 289–291 (1998).
Hansen, D.V., Lui, J.H., Parker, P.R. & Kriegstein, A.R. Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature 464, 554–561 (2010).
Fietz, S.A. et al. OSVZ progenitors of human and ferret neocortex are epithelial-like and expand by integrin signaling. Nat. Neurosci. 13, 690–699 (2010).
Reillo, I., de Juan Romero, C., García-Cabezas, M.A. & Borrell, V. A role for intermediate radial glia in the tangential expansion of the mammalian cerebral cortex. Cereb. Cortex 21, 1674–1694 (2011).
Kelava, I. et al. Abundant occurrence of basal radial glia in the subventricular zone of embryonic neocortex of a lissencephalic primate, the common marmoset Callithrix jacchus. Cereb. Cortex 22, 469–481 (2012).
Suprenant, K.A., Dean, K., McKee, J. & Hake, S. EMAP, an echinoderm microtubule-associated protein found in microtubule-ribosome complexes. J. Cell Sci. 104, 445–450 (1993).
Eichenmüller, B., Everley, P., Palange, J., Lepley, D. & Suprenant, K.A. The human EMAP-like protein-70 (ELP70) is a microtubule destabilizer that localizes to the mitotic apparatus. J. Biol. Chem. 277, 1301–1309 (2002).
Brisch, E., Daggett, M.A. & Suprenant, K.A. Cell cycle–dependent phosphorylation of the 77 kDa echinoderm microtubule-associated protein (EMAP) in vivo and association with the p34cdc2 kinase. J. Cell Sci. 109, 2885–2893 (1996).
Pollmann, M. et al. Human EML4, a novel member of the EMAP family, is essential for microtubule formation. Exp. Cell Res. 312, 3241–3251 (2006).
Hueston, J.L. et al. The C. elegans EMAP-like protein, ELP-1 is required for touch sensation and associates with microtubules and adhesion complexes. BMC Dev. Biol. 8, 110 (2008).
Bechstedt, S. et al. A doublecortin containing microtubule-associated protein is implicated in mechanotransduction in Drosophila sensory cilia. Nat. Commun. 1, 11 (2010).
Tegha-Dunghu, J. et al. EML3 is a nuclear microtubule-binding protein required for the correct alignment of chromosomes in metaphase. J. Cell Sci. 121, 1718–1726 (2008).
Gromley, A. et al. Centriolin anchoring of exocyst and SNARE complexes at the midbody is required for secretory-vesicle-mediated abscission. Cell 123, 75–87 (2005).
Hagemeijer, A. & Graux, C. ABL1 rearrangements in T-cell acute lymphoblastic leukemia. Genes Chromosom. Cancer 49, 299–308 (2010).
Van Karnebeek, C.D. et al. Further delineation of the chromosome 14q terminal deletion syndrome. Am. J. Med. Genet. 110, 65–72 (2002).
Ravnan, J.B. et al. Subtelomere FISH analysis of 11688 cases: an evaluation of the frequency and pattern of subtelomere rearrangements in individuals with developmental disabilities. J. Med. Genet. 43, 478–489 (2006).
Richards, M.W. et al. Crystal structure of EML1 reveals the basis for Hsp90 dependence of oncogenic EML4-ALK by disruption of an atypical β-propeller domain. Proc. Natl. Acad. Sci. USA 111, 5195–5200 (2014).
Sarkisian, M.R. et al. Trouble making the first move: interpreting arrested neuronal migration in the cerebral cortex. Trends Neurosci. 31, 54–61 (2008).
Cappello, S. et al. Mutations in genes encoding the cadherin receptor-ligand pair DCHS1 and FAT4 disrupt cerebral cortical development. Nat. Genet. 45, 1300–1308 (2013).
Takahashi, T., Goto, T., Miyama, S., Nowakowski, R.S. & Caviness, V.S. Jr. Sequence of neuron origin and neocortical laminar fate: relation to cell cycle of origin in the developing murine cerebral wall. J. Neurosci. 19, 10357–10371 (1999).
Stoykova, A., Treichel, D., Hallonet, M. & Gruss, P. Pax6 modulates the dorsoventral patterning of the mammalian telencephalon. J. Neurosci. 20, 8042–8050 (2000).
Götz, M., Stoykova, A. & Gruss, P. Pax6 controls radial glia differentiation in the cerebral cortex. Neuron 21, 1031–1044 (1998).
Wang, X., Tsai, J.W., Lamonica, B. & Kriegstein, A.R. A new subtype of progenitor cell in the mouse embryonic neocortex. Nat. Neurosci. 14, 555–561 (2011).
Shitamukai, A., Konno, D. & Matsuzaki, F. Oblique radial glial divisions in the developing mouse neocortex induce self-renewing progenitors outside the germinal zone that resemble primate outer subventricular zone progenitors. J. Neurosci. 31, 3683–3695 (2011).
Houtman, S.H., Rutteman, M., De Zeeuw, C.I. & French, P.J. Echinoderm microtubule-associated protein like protein 4, a member of the echinoderm microtubule-associated protein family, stabilizes microtubules. Neuroscience 144, 1373–1382 (2007).
Kosodo, Y. et al. Asymmetric distribution of the apical plasma membrane during neurogenic divisions of mammalian neuroepithelial cells. EMBO J. 23, 2314–2324 (2004).
Dubreuil, V., Marzesco, A.M., Corbeil, D., Huttner, W.B. & Wilsch-Bräuninger, M. Midbody and primary cilium of neural progenitors release extracellular membrane particles enriched in the stem cell marker prominin-1. J. Cell Biol. 176, 483–495 (2007).
Weimer, J.M. et al. MARCKS modulates radial progenitor placement, proliferation and organization in the developing cerebral cortex. Development 136, 2965–2975 (2009).
Feng, Y. & Walsh, C.A. Mitotic spindle regulation by Nde1 controls cerebral cortical size. Neuron 44, 279–293 (2004).
Postiglione, M.P. et al. Mouse inscuteable induces apical-basal spindle orientation to facilitate intermediate progenitor generation in the developing neocortex. Neuron 72, 269–284 (2011).
Lancaster, M.A. & Knoblich, J.A. Spindle orientation in mammalian cerebral cortical development. Curr. Opin. Neurobiol. 22, 737–746 (2012).
Fitzgerald, M.P., Covio, M. & Lee, K.S. Disturbances in the positioning, proliferation and apoptosis of neural progenitors contribute to subcortical band heterotopia formation. Neuroscience 176, 455–471 (2011).
Niquille, M. et al. Transient neuronal populations are required to guide callosal axons: a role for semaphorin 3C. PLoS Biol. 7, e1000230 (2009).
Martynoga, B., Morrison, H., Price, D.J. & Mason, J.O. Foxg1 is required for specification of ventral telencephalon and region-specific regulation of dorsal telencephalic precursor proliferation and apoptosis. Dev. Biol. 283, 113–127 (2005).
Bustin, S.A. et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 55, 611–622 (2009).
Vandesompele, J. et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol. 3, research0034.1–research0034.11 (2002).
Gaspard, N. et al. Generation of cortical neurons from mouse embryonic stem cells. Nat. Protoc. 4, 1454–1463 (2009).
Acknowledgements
We thank the Inserm Avenir program, the French Agence National de la Recherche (ANR-08-MNP-013 (F.F.), ANR-13-BSV4-0008-01 (F.F.), ANR Blanc 1103-01 (J.C.), projet R11039KK, ANR E-Rare Program, convention 2011-RaARE-012-01) (J.C.), the Fondation Bettencourt Schueller and the Federation pour la recherche sur le cerveau (FRC) for grants awarded to F.F., the FRC Rotary for an equipment grant awarded to the Institut du Fer à Moulin, the Swiss National Science Foundation (SNSF 31003A-135574, SPUM-33CM30-124089 and 33CM30-140332) and the Fondation Gianni Biaggi de Blasys for grants awarded to A.C., and the SNSF (31003A-125379) for grants awarded to E.W. We are particularly grateful to families and clinicians for access to their DNA samples. We also thank B. Barry, C. Walsh and collaborating clinicians for access to certain of their severe heterotopia patients. We thank W. van Ijcken for performing exome sequencing and R. Schot for excellent analysis of exome data of family 3489. We also thank W.B. Dobyns (Seattle Children's Research Institute) for critical review and classification of the MRI of the affected member of family 3489. We are grateful to D. Turner (University of Michigan) and N. Heintz (Rockefeller University) for reagents. We thank the imaging and animal house platforms at the Institut du Fer à Moulin and the Région Ile de France for support, as well as the animal facility and the Cellular Imaging Facility at the Département des Neurosiences Fondamentales. We thank J. Lemarchand, Y. Saillour, D. Derbala, M. Foglio, A. Boland, D. Zelenika, I. Gut, J.P. Hornung, D. Valloton, M. Niquille, C. Devenoges, N. Narboux-Neme, A. Cabrera, E. Bruel-Jungerman, R. Khalaf-Nazzal, X. Jaglin, T.D.H. Iuliano, M. Karababa, N. Magalhaes, M. Nosten-Bertrand, G. Grannec, A. Houllier and I. Moutkine for their contributions to this work. We are grateful to M. Groszer, J.-A. Girault, A. Houdusse, C. Moores, R. Bayliss, A. Fry, S. Cappello and M. Götz for discussions. The group of F.F. is affiliated with the Ecole des Neurosciences de Paris (ENP) and the Bio-Psy laboratory of excellence. The research leading to a part of these results received funding from the European Union Seventh Framework Programme FP7/2007–2013 under the project DESIRE (grant agreement no. 602531), la Fondation pour la Recherche Médicale (FRM, J Chelly–Equipe FRM 2013: DEQ2000326477) la Fondation Jacques Espinasse & Danièle (JED)-Belgique and the Spanish Ministry of Economy and Competitiveness grant (BFU2012-33473) awarded to V.B.
Author information
Authors and Affiliations
Contributions
M.K. and F.P.D.T. carried out HeCo phenotyping and genotyping projects. F.P.D.T. characterized Eml1 at the genomic, cDNA and protein levels. F.P.D.T. and S.B. performed Vero, N2A, progenitor and neuronal cultures and analysis of EML1 and Eml1 (wild type and mutant) in vitro and mouse in situ hybridization studies, aided by K.B. S.B. performed rescue and shRNA in utero electroporations and analyses with help from R.B. M.K. performed the time lapse movies, pictures and reconstructed the movies. C.L. corrected the movie reconstruction and analyzed the films. C.D.J.R. and V.B. performed ferret in situ hybridizations and in utero transplantation analyses. K.P., R. Oegema, G.M.M., N.B.-B., A.G.L.M., P.B. and J.C. screened and characterized patients. K.P. and N.B.-B. followed and screened a patient DNA panel and controls. R. Oegema and G.M.M. performed exome sequencing and mutation confirmation. R. Olaso and J.-F.D. performed transcriptome experiments, genotyping data analysis and database submissions. D.B. aided with microtubule experiments. W.C. performed the second-round mouse genotyping experiment. E.W., A.C. and F.F. directed the study, analyzed the data and wrote the manuscript, greatly helped by M.K., F.P.D.T. and S.B.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Supplementary Tables 1 and 2 (PDF 2773 kb)
Supplementary Methods Checklist
(PDF 474 kb)
Rights and permissions
About this article
Cite this article
Kielar, M., Tuy, F., Bizzotto, S. et al. Mutations in Eml1 lead to ectopic progenitors and neuronal heterotopia in mouse and human. Nat Neurosci 17, 923–933 (2014). https://doi.org/10.1038/nn.3729
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3729
This article is cited by
-
Novel role of the synaptic scaffold protein Dlgap4 in ventricular surface integrity and neuronal migration during cortical development
Nature Communications (2022)
-
EML1 is essential for retinal photoreceptor migration and survival
Scientific Reports (2022)
-
Androgens increase excitatory neurogenic potential in human brain organoids
Nature (2022)
-
Variant-specific effects define the phenotypic spectrum of HNRNPH2-associated neurodevelopmental disorders in males
Human Genetics (2022)
-
A novel missense variant in the EML1 gene associated with bilateral ribbon-like subcortical heterotopia leads to ciliary defects
Journal of Human Genetics (2021)