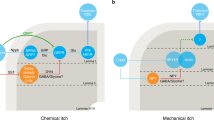
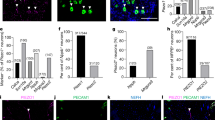
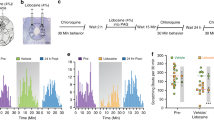
Abstract
Itch is described as an irritating sensation that triggers a desire to scratch. However, this definition hardly seems fitting for the millions of people who suffer from intractable itch. Indeed, the Buddhist philosopher Nā´áäāgārjuna more aptly stated, “There is pleasure when an itch is scratched. But to be without an itch is more pleasurable still.” Chronic itch is widespread and very difficult to treat. In this review we focus on the molecules, cells and circuits in the peripheral and central nervous systems that drive acute and chronic itch transmission. Understanding the itch circuitry is critical to developing new therapies for this intractable disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ikoma, A., Steinhoff, M., Stander, S., Yosipovitch, G. & Schmelz, M. The neurobiology of itch. Nat. Rev. Neurosci. 7, 535–547 (2006).
Patel, T. & Yosipovitch, G. Therapy of pruritus. Expert Opin. Pharmacother. 11, 1673–1682 (2010).
Elmariah, S.B. & Lerner, E.A. Topical therapies for pruritus. Semin. Cutan. Med. Surg. 30, 118–126 (2011).
Davidson, S. & Giesler, G.J. The multiple pathways for itch and their interactions with pain. Trends Neurosci. 33, 550–558 (2010).
Ross, S.E. Pain and itch: insights into the neural circuits of aversive somatosensation in health and disease. Curr. Opin. Neurobiol. 21, 880–887 (2011).
McNeil, B. & Dong, X. Peripheral mechanisms of itch. Neurosci. Bull. 28, 100–110 (2012).
Akiyama, T. & Carstens, E. Neural processing of itch. Neuroscience 250, 697–714 (2013).
Garibyan, L., Rheingold, C.G. & Lerner, E.A. Understanding the pathophysiology of itch. Dermatol. Ther. 26, 84–91 (2013).
Shim, W.S. et al. TRPV1 mediates histamine-induced itching via the activation of phospholipase A2 and 12-lipoxygenase. J. Neurosci. 27, 2331–2337 (2007).
Imamachi, N. et al. TRPV1-expressing primary afferents generate behavioral responses to pruritogens via multiple mechanisms. Proc. Natl. Acad. Sci. USA 106, 11330–11335 (2009).
Akiyama, T., Carstens, M.I. & Carstens, E. Enhanced scratching evoked by PAR-2 agonist and 5-HT but not histamine in a mouse model of chronic dry skin itch. Pain 151, 378–383 (2010).
Wilson, S.R. et al. The epithelial cell-derived atopic dermatitis cytokine TSLP activates neurons to induce itch. Cell 155, 285–295 (2013).
Kim, S.J. et al. Analysis of cellular and behavioral responses to imiquimod reveals a unique itch pathway in transient receptor potential vanilloid 1 (TRPV1)-expressing neurons. Proc. Natl. Acad. Sci. USA 108, 3371–3376 (2011).
Wilson, S.R. et al. TRPA1 is required for histamine-independent, Mas-related G protein–coupled receptor-mediated itch. Nat. Neurosci. 14, 595–602 (2011).
Liu, Q. et al. Sensory neuron-specific GPCR Mrgprs are itch receptors mediating chloroquine-induced pruritus. Cell 139, 1353–1365 (2009).
Liu, Q. et al. Mechanisms of itch evoked by β-alanine. J. Neurosci. 32, 14532–14537 (2012).
Zylka, M.J., Rice, F.L. & Anderson, D.J. Topographically distinct epidermal nociceptive circuits revealed by axonal tracers targeted to Mrgprd. Neuron 45, 17–25 (2005).
Cavanaugh, D.J. et al. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc. Natl. Acad. Sci. USA 106, 9075–9080 (2009).
Rau, K.K. et al. Mrgprd enhances excitability in specific populations of cutaneous murine polymodal nociceptors. J. Neurosci. 29, 8612–8619 (2009).
Shields, S.D., Cavanaugh, D.J., Lee, H., Anderson, D.J. & Basbaum, A.I. Pain behavior in the formalin test persists after ablation of the great majority of C-fiber nociceptors. Pain 151, 422–429 (2010).
Mishra, S.K., Tisel, S.M., Orestes, P., Bhangoo, S.K. & Hoon, M.A. TRPV1-lineage neurons are required for thermal sensation. EMBO J. 30, 582–593 (2011).
Steinhoff, M. et al. Proteinase-activated receptor-2 in human skin: tissue distribution and activation of keratinocytes by mast cell tryptase. Exp. Dermatol. 8, 282–294 (1999).
Steinhoff, M. et al. Proteinase-activated receptor-2 mediates itch: a novel pathway for pruritus in human skin. J. Neurosci. 23, 6176–6180 (2003).
Shimada, S.G., Shimada, K.A. & Collins, J.G. Scratching behavior in mice induced by the proteinase-activated receptor-2 agonist, SLIGRL-NH2. Eur. J. Pharmacol. 530, 281–283 (2006).
Liu, Q. et al. The distinct roles of two GPCRs, MrgprC11 and PAR2, in itch and hyperalgesia. Sci. Signal. 4, ra45 (2011).
Reddy, V.B., Shimada, S.G., Sikand, P., Lamotte, R.H. & Lerner, E.A. Cathepsin S elicits itch and signals via protease-activated receptors. J. Invest. Dermatol. 130, 1468–1470 (2010).
Kim, N. et al. Overexpression of cathepsin S induces chronic atopic dermatitis in mice. J. Invest. Dermatol. 132, 1169–1176 (2012).
Liu, T., Xu, Z.Z., Park, C.K., Berta, T. & Ji, R.R. Toll-like receptor 7 mediates pruritus. Nat. Neurosci. 13, 1460–1462 (2010).
Liu, T. et al. TLR3 deficiency impairs spinal cord synaptic transmission, central sensitization, and pruritus in mice. J. Clin. Invest. 122, 2195–2207 (2012).
Liu, T., Gao, Y.J. & Ji, R.R. Emerging role of Toll-like receptors in the control of pain and itch. Neurosci. Bull. 28, 131–144 (2012).
Barton, G.M. & Kagan, J.C. A cell biological view of Toll-like receptor function: regulation through compartmentalization. Nat. Rev. Immunol. 9, 535–542 (2009).
Abreu, M.T., Fukata, M. & Arditi, M. TLR signaling in the gut in health and disease. J. Immunol. 174, 4453–4460 (2005).
Sabroe, I., Parker, L.C., Wilson, A.G., Whyte, M.K. & Dower, S.K. Toll-like receptors: their role in allergy and nonallergic inflammatory disease. Clin. Exp. Allergy 32, 984–989 (2002).
Wilson, S.R. et al. The ion channel TRPA1 is required for chronic itch. J. Neurosci. 33, 9283–9294 (2013).
Chiu, I.M. et al. Bacteria activate sensory neurons that modulate pain and inflammation. Nature 501, 52–57 (2013).
Salpietro, C. et al. TLR2 and TLR4 gene polymorphisms and atopic dermatitis in Italian children: a multicenter study. Int. J. Immunopathol. Pharmacol. 24, 33–40 (2011).
Mrabet-Dahbi, S. et al. The Toll-like receptor 2 R753Q mutation modifies cytokine production and Toll-like receptor expression in atopic dermatitis. J. Allergy Clin. Immunol. 121, 1013–1019 (2008).
Han, L. et al. A subpopulation of nociceptors specifically linked to itch. Nat. Neurosci. 16, 174–182 (2013).
McCoy, E.S. et al. Peptidergic CGRPα primary sensory neurons encode heat and itch and tonically suppress sensitivity to cold. Neuron 78, 138–151 (2013).
Roberson, D.P. et al. Activity-dependent silencing reveals functionally distinct itch-generating sensory neurons. Nat. Neurosci. 16, 910–918 (2013).
Namer, B. et al. Separate peripheral pathways for pruritus in man. J. Neurophysiol. 100, 2062–2069 (2008).
Johanek, L.M. et al. A role for polymodal C-fiber afferents in nonhistaminergic itch. J. Neurosci. 28, 7659–7669 (2008).
Davidson, S. et al. Pruriceptive spinothalamic tract neurons: physiological properties and projection targets in the primate. J. Neurophysiol. 108, 1711–1723 (2012).
Lagerström, M.C. et al. VGLUT2-dependent sensory neurons in the TRPV1 population regulate pain and itch. Neuron 68, 529–542 (2010).
Liu, Y. et al. VGLUT2-dependent glutamate release from nociceptors is required to sense pain and suppress itch. Neuron 68, 543–556 (2010).
Sun, Y.G. & Chen, Z.F. A gastrin-releasing peptide receptor mediates the itch sensation in the spinal cord. Nature 448, 700–703 (2007).
Sun, Y.G. et al. Cellular basis of itch sensation. Science 325, 1531–1534 (2009).
Mishra, S.K. & Hoon, M.A. The cells and circuitry for itch responses in mice. Science 340, 968–971 (2013).
Mishra, S.K., Holzman, S. & Hoon, M.A. A nociceptive signaling role for neuromedin B. J. Neurosci. 32, 8686–8695 (2012).
Sukhtankar, D.D. & Ko, M.C. Physiological function of gastrin-releasing peptide and neuromedin B receptors in regulating itch scratching behavior in the spinal cord of mice. PLoS ONE 8, e67422 (2013).
Fleming, M.S. et al. The majority of dorsal spinal cord gastrin releasing peptide is synthesized locally whereas neuromedin B is highly expressed in pain- and itch-sensing somatosensory neurons. Mol. Pain 8, 52 (2012).
Zhao, Z.Q. et al. Chronic itch development in sensory neurons requires BRAF signaling pathways. J. Clin. Invest. 123, 4769–4780 (2013).
Wang, X. et al. Excitatory superficial dorsal horn interneurons are functionally heterogeneous and required for the full behavioral expression of pain and itch. Neuron 78, 312–324 (2013).
Li, M.Z. et al. Molecular mapping of developing dorsal horn-enriched genes by microarray and dorsal/ventral subtractive screening. Dev. Biol. 292, 555–564 (2006).
Forssmann, W.G. et al. The heart is the center of a new endocrine, paracrine, and neuroendocrine system. Arch. Histol. Cytol. 52 (suppl.), 293–315 (1989).
Zhang, F.X. et al. Inhibition of inflammatory pain by activating B-type natriuretic peptide signal pathway in nociceptive sensory neurons. J. Neurosci. 30, 10927–10938 (2010).
Liu, X.Y. et al. Unidirectional cross-activation of GRPR by MOR1D uncouples itch and analgesia induced by opioids. Cell 147, 447–458 (2011).
Moser, H.R. & Giesler, G.J. Jr. Itch and analgesia resulting from intrathecal application of morphine: contrasting effects on different populations of trigeminothalamic tract neurons. J. Neurosci. 33, 6093–6101 (2013).
Xu, Y. et al. Ontogeny of excitatory spinal neurons processing distinct somatic sensory modalities. J. Neurosci. 33, 14738–14748 (2013).
Akiyama, T., Iodi Carstens, M. & Carstens, E. Transmitters and pathways mediating inhibition of spinal itch-signaling neurons by scratching and other counterstimuli. PLoS ONE 6, e22665 (2011).
Inan, S. & Cowan, A. Nalfurafine, a kappa opioid receptor agonist, inhibits scratching behavior secondary to cholestasis induced by chronic ethynylestradiol injections in rats. Pharmacol. Biochem. Behav. 85, 39–43 (2006).
Davidson, S., Zhang, X., Khasabov, S.G., Simone, D.A. & Giesler, G.J. Jr. Relief of itch by scratching: state-dependent inhibition of primate spinothalamic tract neurons. Nat. Neurosci. 12, 544–546 (2009).
Papoiu, A.D., Coghill, R.C., Kraft, R.A., Wang, H. & Yosipovitch, G. A tale of two itches. Common features and notable differences in brain activation evoked by cowhage and histamine induced itch. Neuroimage 59, 3611–3623 (2012).
Liu, B. et al. TRPA1 controls inflammation and pruritogen responses in allergic contact dermatitis. FASEB J. 27, 3549–3563 (2013).
Maurer, M. et al. Omalizumab for the treatment of chronic idiopathic or spontaneous urticaria. N. Engl. J. Med. 368, 924–935 (2013).
Grimstad, O. et al. Anti-interleukin-31-antibodies ameliorate scratching behaviour in NC/Nga mice: a model of atopic dermatitis. Exp. Dermatol. 18, 35–43 (2009).
Basbaum, A.I., Bautista, D.M., Scherrer, G. & Julius, D. Cellular and molecular mechanisms of pain. Cell 139, 267–284 (2009).
Tani, E. & Ishikawa, T. Histamine acts directly on calcitonin gene-related peptide- and substance P-containing trigeminal ganglion neurons as assessed by calcium influx and immunocytochemistry. Auris Nasus Larynx 17, 267–274 (1990).
Zhou, Z., Davar, G. & Strichartz, G. Endothelin-1 (ET-1) selectively enhances the activation gating of slowly inactivating tetrodotoxin-resistant sodium currents in rat sensory neurons: a mechanism for the pain-inducing actions of ET-1. J. Neurosci. 22, 6325–6330 (2002).
Gomes, L.O., Hara, D.B. & Rae, G.A. Endothelin-1 induces itch and pain in the mouse cheek model. Life Sci. 91, 628–633 (2012).
Liang, J., Ji, Q. & Ji, W. Role of transient receptor potential ankyrin subfamily member 1 in pruritus induced by endothelin-1. Neurosci. Lett. 492, 175–178 (2011).
Kim, D.K. et al. Involvement of serotonin receptors 5-HT1 and 5-HT2 in 12(S)-HPETE-induced scratching in mice. Eur. J. Pharmacol. 579, 390–394 (2008).
Stefansson, K. et al. Activation of proteinase-activated receptor-2 by human kallikrein-related peptidases. J. Invest. Dermatol. 128, 18–25 (2008).
Tsujii, K., Andoh, T., Lee, J.B. & Kuraishi, Y. Activation of proteinase-activated receptors induces itch-associated response through histamine-dependent and -independent pathways in mice. J. Pharmacol. Sci. 108, 385–388 (2008).
Zhu, Y. et al. Participation of proteinase-activated receptor-2 in passive cutaneous anaphylaxis-induced scratching behavior and the inhibitory effect of tacrolimus. Biol. Pharm. Bull. 32, 1173–1176 (2009).
Bautista, D.M., Pellegrino, M. & Tsunozaki, M. TRPA1: a gatekeeper for inflammation. Annu. Rev. Physiol. 75, 181–200 (2013).
Tominaga, M. & Caterina, M.J. Thermosensation and pain. J. Neurobiol. 61, 3–12 (2004).
Miyamoto, T., Nojima, H., Shinkado, T., Nakahashi, T. & Kuraishi, Y. Itch-associated response induced by experimental dry skin in mice. Jpn. J. Pharmacol. 88, 285–292 (2002).
Akiyama, T., Carstens, M.I. & Carstens, E. Spontaneous itch in the absence of hyperalgesia in a mouse hindpaw dry skin model. Neurosci. Lett. 484, 62–65 (2010).
Jin, H., He, R., Oyoshi, M. & Geha, R.S. Animal models of atopic dermatitis. J. Invest. Dermatol. 129, 31–40 (2009).
Seike, M., Ikeda, M., Kodama, H., Terui, T. & Ohtsu, H. Inhibition of scratching behaviour caused by contact dermatitis in histidine decarboxylase gene knockout mice. Exp. Dermatol. 14, 169–175 (2005).
Dey, D.D., Landrum, O. & Oaklander, A.L. Central neuropathic itch from spinal-cord cavernous hemangioma: a human case, a possible animal model, and hypotheses about pathogenesis. Pain 113, 233–237 (2005).
D'Almeida, J.A. et al. Behavioral changes of Wistar rats with experimentally-induced painful diabetic neuropathy. Arq. Neuropsiquiatr. 57, 746–752 (1999).
Stebbins, K.J. et al. Therapeutic efficacy of AM156, a novel prostanoid DP2 receptor antagonist, in murine models of allergic rhinitis and house dust mite-induced pulmonary inflammation. Eur. J. Pharmacol. 638, 142–149 (2010).
Zheng, T. et al. Transgenic expression of interleukin-13 in the skin induces a pruritic dermatitis and skin remodeling. J. Invest. Dermatol. 129, 742–751 (2009).
Hansson, L. et al. Epidermal overexpression of stratum corneum chymotryptic enzyme in mice: a model for chronic itchy dermatitis. J. Invest. Dermatol. 118, 444–449 (2002).
Konishi, H. et al. IL-18 contributes to the spontaneous development of atopic dermatitis-like inflammatory skin lesion independently of IgE/stat6 under specific pathogen-free conditions. Proc. Natl. Acad. Sci. USA 99, 11340–11345 (2002).
Briot, A. et al. Par2 inactivation inhibits early production of TSLP, but not cutaneous inflammation, in Netherton syndrome adult mouse model. J. Invest. Dermatol. 130, 2736–2742 (2010).
Brandt, E.B., Gibson, A.M., Bass, S., Rydyznski, C. & Khurana Hershey, G.K. Exacerbation of allergen-induced eczema in TLR4- and TRIF-deficient mice. J. Immunol. 191, 3519–3525 (2013).
Turner, M.J. et al. Topical application of a vitamin D analogue exacerbates atopic dermatitis and induces the atopic dermatitis-like phenotype in Stat6VT mice. Pediatr. Dermatol. 30, 574–578 (2013).
Nakajima, K. et al. Barrier abnormality due to ceramide deficiency leads to psoriasiform inflammation in a mouse model. J. Invest. Dermatol. 133, 2555–2565 (2013).
Yockey, L.J. et al. The absence of a microbiota enhances TSLP expression in mice with defective skin barrier but does not affect the severity of their allergic inflammation. J. Invest. Dermatol. 133, 2714–2721 (2013).
Yoo, J. et al. Spontaneous atopic dermatitis in mice expressing an inducible thymic stromal lymphopoietin transgene specifically in the skin. J. Exp. Med. 202, 541–549 (2005).
Oh, M.H. et al. TRPA1-dependent pruritus in IL-13-induced chronic atopic dermatitis. J. Immunol. 191, 5371–5382 (2013).
Wallace, K. et al. The PXR is a drug target for chronic inflammatory liver disease. J. Steroid Biochem. Mol. Biol. 120, 137–148 (2010).
Belghiti, M. et al. Potentiation of the transient receptor potential vanilloid 1 channel contributes to pruritogenesis in a rat model of liver disease. J. Biol. Chem. 288, 9675–9685 (2013).
Yilinuer, H., Yamaoka, J. & Kawana, S. Effect of epinastine hydrochloride on murine self-scratching behavior after skin-scratching stimulation. Arch. Dermatol. Res. 302, 19–26 (2010).
Honma, Y. et al. Cyclooxygenase-1 inhibition delays recovery of the cutaneous barrier disruption caused by mechanical scratching in mice. Br. J. Dermatol. 156, 1178–1187 (2007).
Acknowledgements
We are grateful to M. Pellegrino for critical comments on the manuscript. This work was supported by US National Institutes of Health (NIH) grants AR059385 and DOD007123A (D.M.B.), the intramural research program of the NIH NIDCR (M.A.H.) and a US National Science Foundation Fellowship (S.R.W.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All authors are in the process of filing a patent application related to some of the results described in this manuscript.
Rights and permissions
About this article
Cite this article
Bautista, D., Wilson, S. & Hoon, M. Why we scratch an itch: the molecules, cells and circuits of itch. Nat Neurosci 17, 175–182 (2014). https://doi.org/10.1038/nn.3619
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3619
This article is cited by
-
Exposure limits for indoor volatile substances concerning the general population: The role of population-based differences in sensory irritation of the eyes and airways for assessment factors
Archives of Toxicology (2024)
-
An Anterior Cingulate Cortex-to-Midbrain Projection Controls Chronic Itch in Mice
Neuroscience Bulletin (2023)
-
The Anterior Insula and its Projection to the Prelimbic Cortex are Involved in the Regulation of 5-HT-Induced Itch
Neuroscience Bulletin (2023)
-
Exploring neuronal mechanisms involved in the scratching behavior of a mouse model of allergic contact dermatitis by transcriptomics
Cellular & Molecular Biology Letters (2022)
-
No indications for altered EEG oscillatory activity in patients with chronic post-burn itch compared to healthy controls
Scientific Reports (2022)