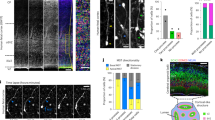
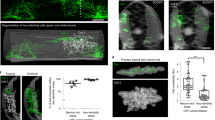
Abstract
Neural progenitors exhibit cell cycle–dependent interkinetic nuclear migration (INM) along the apicobasal axis. Despite recent advances in understanding its underlying molecular mechanisms, the processes to which INM contributes mechanically and the regulation of INM by the apicobasally elongated morphology of progenitors remain unclear. We found that knockdown of the cell-surface molecule TAG-1 resulted in retraction of neocortical progenitors' basal processes. Highly shortened stem-like progenitors failed to undergo basalward INM and became overcrowded in the periventricular (subapical) space. Surprisingly, the overcrowded progenitors left the apical surface and migrated into basal neuronal territories. These observations, together with the results of in toto imaging and physical tests, suggest that progenitors may sense and respond to excessive mechanical stress. Although, unexpectedly, the heterotopic progenitors remained stem-like and continued to sequentially produce neurons until the late embryonic period, histogenesis was severely disrupted. Thus, INM is essential for preventing overcrowding of nuclei and their somata, thereby ensuring normal brain histogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Schaper, A. The earliest differentiation in the central nervous system of vertebrates. Science 5, 430–431 (1897).
Sauer, F.C. Mitosis in the neural tube. J. Comp. Neurol. 62, 377–405 (1935).
Chenn, A. & McConnell, S.K. Cleavage orientation and the asymmetric inheritance of Notch1 immunoreactivity in mammalian neurogenesis. Cell 82, 631–641 (1995).
Taverna, E. & Huttner, W.B. Neural progenitor nuclei in motion. Neuron 67, 906–914 (2010).
Lui, J.H., Hansen, D.V. & Kriegstein, A.R. Development and evolution of the human neocortex. Cell 146, 18–36 (2011).
Reiner, O., Sapir, T. & Gerlitz, G. Interkinetic nuclear movement in the ventricular zone of the cortex. J. Mol. Neurosci. 46, 516–526 (2012).
Sanes, J.R. Analysing cell lineage with a recombinant virus. Trends Neurosci. 12, 21–28 (1989).
Miyata, T., Kawaguchi, A., Okano, H. & Ogawa, M. Asymmetric inheritance of radial glial fibers by cortical neurons. Neuron 31, 727–741 (2001).
Noctor, S.C., Flint, A.C., Weissman, T.A., Dammerman, R.S. & Kriegstein, A.R. Neurons derived from radial glial cells establish radial units in neocortex. Nature 409, 714–720 (2001).
Tsai, J.-W., Chen, Y., Kriegstein, A. & Vallee, R.B. RNA interference blocks neural stem cell division, morphogenesis, and motility at multiple steps. J. Cell Biol. 170, 935–945 (2005).
Xie, Z. et al. Cep120 and TACC control interkinetic nuclear migration and neural progenitor pool. Neuron 56, 79–93 (2007).
Norden, C., Young, S., Link, B.A. & Harris, W.A. Actomyosin is the main driver of interkinetic nuclear migration in the retina. Cell 138, 1195–1208 (2009).
Tsai, J.W., Lian, W.N., Kemal, S., Kriegstein, A.R. & Vallee, R.B. Kinesin 3 and cytoplasmic dynein mediate interkinetic nuclear migration in neural stem cells. Nat. Neurosci. 13, 1463–1471 (2010).
Schenk, J., Wilsch-Brauninger, M., Calegari, F. & Huttner, W.B. Myosin II is required for interkinetic nuclear migration of neural progenitors. Proc. Natl. Acad. Sci. USA 106, 16487–16492 (2009).
Kosodo, Y. et al. Regulation of interkinetic nuclear migration by cell cycle-coupled active and passive mechanisms in the developing brain. EMBO J. 30, 1690–1704 (2011).
Leung, L., Klopper, A.V., Grill, S.W., Harris, W.A. & Norden, C. Apical migration of nuclei during G2 is a prerequisite for all nuclear motion in zebrafish neuroepithelia. Development 138, 5003–5013 (2011).
Smart, I.H.M. The operation of ependymal 'choke' in neurogenesis. J. Anat. 99, 941–943 (1965).
Fietz, S.A. et al. OSVZ progenitors of human and ferret neocortex are epithelial- like and expand by integrin signaling. Nat. Neurosci. 13, 690–699 (2010).
Hansen, D.V., Lui, J.H., Parker, P.R. & Kriegstein, A.R. Neurogenic radial glia in the outer subventricular zone of human neocortex. Nature 464, 554–561 (2010).
Barkovich, A.J. et al. A developmental and genetic classification for malformations of cortical development: update 2012. Brain 135, 1348–1369 (2012).
Götz, M. & Huttner, W.B. The cell biology of neurogenesis. Nat. Rev. Mol. Cell Biol. 6, 777–788 (2005).
Konno, D. et al. Neuroepithelial progenitors undergo LGN-dependent planar divisions to maintain self-renewability during mammalian neurogenesis. Nat. Cell Biol. 10, 93–101 (2008).
Postiglione, M.P. et al. Mouse inscrutable induces apical-basal spindle orientation to facilitate intermediate progenitor generation in the developing neocortex. Neuron 72, 269–284 (2011).
Furley, A.J. et al. The axonal glycoprotein TAG-1 is an immunoglobulin superfamily member with neurite outgrowth-promoting activity. Cell 61, 157–170 (1990).
Abe, T. et al. Establishment of conditional reporter mouse lines at ROSA26 locus for live cell imaging. Genesis 49, 579–590 (2011).
Miyanaga, Y., Matsuoka, S. & Ueda, M. Single-molecule imaging techniques to visualize chemotactic signaling events on the membrane of living Dictyostelium cells. Methods Mol. Biol. 571, 417–435 (2009).
Qian, H., Sheetz, M.P. & Elson, E.L. Single particle tracking. Analysis of diffusion and flow in two-dimensional systems. Biophys. J. 60, 910–921 (1991).
Kusumi, A., Sako, Y. & Yamamoto, M. Confined lateral diffusion of membrane receptors as studied by single particle tracking (nanovid microscopy). Effects of calcium-induced differentiation in cultured epithelial cells. Biophys. J. 65, 2021–2040 (1993).
Nishizawa, Y. et al. Survey of the morphogenetic dynamics of the ventricular surface of the developing mouse neocortex. Dev. Dyn. 236, 3061–3070 (2007).
Shimogori, T. & Ogawa, M. Gene application with in utero electroporation in mouse embryonic brain. Dev. Growth Differ. 50, 499–506 (2008).
Miyata, T. & Ogawa, M. Twisting of neocortical progenitor cells underlies a spring-like mechanism for daughter cell migration. Curr. Biol. 17, 146–151 (2007).
Munji, R.N., Choe, Y., Li, G., Siegenthaler, J.A. & Pleasure, S.J. Wnt signaling regulates neuronal differentiation of cortical intermediate progenitors. J. Neurosci. 31, 1676–1687 (2011).
Molyneaux, B.J., Arlotta, P., Menezes, J.R. & Macklis, J.D. Neuronal subtype specification in the cerebral cortex. Nat. Rev. Neurosci. 8, 427–437 (2007).
Bizzoca, A. et al. F3/Contactin acts as a modulator of neurogenesis during cerebral cortex development. Dev. Biol. 365, 133–151 (2012).
Milev, P., Maurel, P., Haring, M., Margolis, R.K. & Margolis, R.U. TAG-1/Axonin-1 is a high-affinity ligand of neurocan, phosphacan/protein-tyrosine phosphatase-ζ/β, and N-CAM. J. Biol. Chem. 271, 15716–15723 (1996).
Sittaramane, V. et al. The cell adhesion molecule Tag1, transmembrane protein Stbm/Vangl2, and Lamininα1 exhibit genetic interactions during migration of facial branchiomotor neurons in zebrafish. Dev. Biol. 325, 363–373 (2009).
Fukamauchi, F. et al. TAG-1-deficient mice have marked elevation of adenosine A1 receptors in the hippocampus. Biochem. Biophys. Res. Commun. 281, 220–226 (2001).
Shitamukai, A., Konno, D. & Matsuzaki, F. Oblique radial glial divisions in the developing mouse neocortex induce self-renewing progenitors outside the germinal zone that resemble primate outer subventricular zone progenitors. J. Neurosci. 31, 3683–3695 (2011).
Marinari, E. et al. Live-cell delamination counterbalances epithelial growth to limit tissue overcrowding. Nature 484, 542–545 (2012).
Itoh, Y. et al. Scratch regulates neuronal migration onset via an epithelial-mesenchymal transition-like mechanism. Nat. Neurosci. 16, 416–425 (2013).
Fitzgerald, M.P., Covio, M. & Lee, K.S. Disturbances in the positioning, proliferation and apoptosis of neural progenitors contribute to subcortical band heterotopia formation. Neuroscience 176, 455–471 (2011).
Morin, X., Jaouen, F. & Durbec, P. Control of planar divisions by the G-protein regulator LGN maintains progenitors in the chick neuroepithelium. Nat. Neurosci. 10, 1440–1448 (2007).
Chenn, A. & Walsh, C.A. Regulation of cerebral cortical size by control of cell cycle exit in neural precursors. Science 297, 365–369 (2002).
Inglis-Broadgate, S.L. et al. FGFR3 regulates brain size by controlling progenitor cell proliferation and apoptosis during embryonic development. Dev. Biol. 279, 73–85 (2005).
Zedević, N. Cellular composition of the telencephalic wall in human embryos. Early Hum. Dev. 32, 131–149 (1993).
Bayer, S.A. & Altman, J. The Human Brain During the Late First Trimester (CRC Press Taylor & Francis Group, 2006).
Tsunekawa, Y. et al. Cyclin D2 in the basal process of neural progenitors is linked to non-equivalent cell fates. EMBO J. 31, 1879–1892 (2012).
Siegenthaler, J.A. et al. Retinoic acid from the meninges regulates cortical neuron generation. Cell 139, 597–609 (2009).
Rørth, P. Fellow travelers: emergent properties of collective cell migration. EMBO Rep. 13, 984–991 (2012).
Heisenberg, C.-P. & Bellaïche, Y. Forces in tissue morphogenesis and patterning. Cell 153, 948–962 (2013).
Kondo, T. & Hayashi, S. Mitotic cell rounding accelerates epithelial invagination. Nature 494, 125–129 (2013).
Kawabata, I. et al. Electroporation-mediated gene transfer system applied to cultured CNS neurons. Neuroreport 15, 971–975 (2004).
Sakakibara, A. et al. Dynamics of centrosome translocation and microtubule organization in neocortical neurons during distinct modes of polarization. Cereb. Cortex doi:10.1093/cercor/bhs411 (10 January 2013).10.1093/cercor/bhs411
Nakamuta, S. et al. Local application of neurotrophins specifies axons through inositol 1,4,5-trisphosphate, calcium, and Ca2+/calmodulin-dependent protein kinases. Sci. Signal. 4, ra76 (2011).
Sapir, T. et al. Accurate balance of the polarity kinase MARK2/Par-1 is required for proper cortical neuronal migration. J. Neurosci. 28, 5710–5720 (2008).
Hino, T. et al. Accelerated modification of the zona pellucida is the primary cause of decreased fertilizability of oocyte in the 129 inbred mouse strain. Zygote 19, 315–322 (2011).
McLean, I.W. & Nakane, P.K. Periodate-lysine-paraformaldehyde fixative. A new fixation for immunoelectron microscopy. J. Histochem. Cytochem. 22, 1077–1083 (1974).
Honda, H., Tanemura, M. & Nagai, T. A three-dimensional vertex dynamics cell model of space-filling polyhedra simulating cell behaviour in a cell aggregate. J. Theor. Biol. 226, 439–453 (2004).
Eiraku, M. et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature 472, 51–56 (2011).
Okuda, S., Inoue, Y., Eiraku, M., Sasai, Y. & Adachi, T. Modeling cell proliferation for simulating three-dimensional tissue morphogenesis based on a reversible network reconnection framework. Biomech. Model Mechanobiol. doi:10.1007/s10237-012-0458-8 (30 November 2012).10.1007/s10237-012-0458-8
Acknowledgements
We thank T. Shimogori for instruction in utero electroporation at E10; S. Nakagawa (RIKEN Advanced Science Institute), K. Ohta (Kumamoto University), S. Takada (Japan National Institute for Basic Biology), F. Matsuzaki (RIKEN Center for Developmental Biology), H. Kimura (Osaka University), S. Okabe (University of Tokyo) and T. Sapir (Weizmann Institute of Science) for plasmids; Y. Itoh and Y. Gotoh (both University of Tokyo) for antibodies; F. Matsuzaki and M. Ogawa for critical reading of an earlier version of the manuscript; S. Okuda, H. Takagi, K. Nagayama, T. Matsumoto for suggestions; members of the Matsuzaki, Nakajima and Ueda laboratories for discussions; and M. Masaoka, T. Kawaue, J. Yamada and N. Noguchi for excellent technical assistance. This work was supported by Japan Ministry of Education, Culture, Sports, Science and Technology Grant-in-Aid for Scientific Research on Innovative Areas “Cross-talk between moving cells and microenvironment as a basis of emerging order” 22111006 (T.M.), Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (A) 21240027 (T.M.), MEXT Global COE (Center of Excellence) program “Integrated Functional Molecular Medicine for Neuronal and Neoplastic Disorders” (T.M.), and JSPS Grant-in-Aid for Young Scientists (B) 23700378 (M.O.).
Author information
Authors and Affiliations
Contributions
M.O. and T.M. designed the experiments and performed data analysis. M.O. performed most of the experiments. T.N., K.N. and K.K. supported RNAi experiments. Y.E. supported mechanical experiments. K.T., K. Oda. and Y.W. provided TAG-1−/− mice. T.F. generated Rosa26-lyn-venus mice. T.S., K. Saito, K. Sagou, A.S., Y.E., K. Ota and T.F. supported live imaging and cell tracking. A.K. contributed to gene expression profiling and statistics. M.U. and K. Ota supported MSD analysis. T.K. and S.H. contributed to laser ablation and analysis. T.W., Y.I., and T.A. performed mechanical simulations. M.O. and T.M. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Cellular and tissue-level behaviors revealing the mechanical properties at or near the apical surface of developing cerebral walls
a) Tangential in toto monitoring of plasma membranes of all cells in the periventricular space. M-phase cells divide horizontally, laterally pushing their neighbors, mainly the apical processes of the surrounding cells. Newly generated daughter cells quickly reduce sectional area, indicating their migration away from the apical surface. b) Bending of apical processes. Case 1 (interval between the two observations was 2.7 hr) and Case 2 (1.1-hr interval) display processes of pairs of sibling daughter cells. Case 3 (2.8-hr interval) involves a single cell. Note that all of these apical processes were bent when a new M-phase cell (blue) came down to the apical surface. If the space contained other voluminous and less flexible structures (e.g., cell bodies) instead of these thin processes, it would be more difficult for M-phase cells to take a normal globular morphology than is observed here. c) MSD curves for nuclei moving basally (green) ranged from a non-linear pattern to a linear (fluctuating or “stationary”) pattern independently of the initial nuclear position (bins 1–5 from the apical most to basal; each is 20 μm wide). Previous studies on zebrafish NE revealed that non-linear MSD patterns are seen only transiently in basalward movements of nuclei very close to the apical surface (i.e., those of daughter cells immediately after their generation at the surface)12,16. By contrast, in our MSD analysis of basally migrating nuclei, the non-linear (directional) pattern was observed regardless of the initial distance of a tested nucleus from the apical surface. d,e) Schematic illustration of two patterns of sibling daughter cells' behaviors. d) A basally connected progenitor divides, and one daughter that has inherited the basal process moves its nucleus more quickly than its sister. This is analogous to “priority boarding” in air travel or “staggered commuting” in metropolitan railway systems, and may contribute to the normally prompt stratification of nuclei/somata in each column. e) A progenitor cell without a basal process divides, generating two sibling daughters. The sibling daughter's somata can compete with each other for space into which they might basally migrate, thereby laterally expanding a column to which they belong. Such expansion may cause mechanical conflicts between neighboring columns. This situation arises in TAG-1–KD neuroepithelia, and is abnormal.
Supplementary Figure 2 Results showing TAG-1 deficiency–induced shortening of progenitors, contractility of the apical surface and slight retraction of the basal process by laser ablation at the basal surface
a) Stage-dependent disappearance of the progenitor's basal process (number of GFP+ VZ cells examined = 238 cells for E10 1 E12, 102 cells for E11 1 E13, 134 cells for E12 1 E14, and 125 cells for E13 1 E15). TAG-1 shRNA electroporation at E11, E12, and E13 revealed that the phenotype of basal-process disappearance was reproduced frequently in embryos electoroporated at E11, but rarely in those electoroporated at E13; E12-electroporated embryos exhibited an intermediate phenotype. This difference may be explained partially by the observation that TAG-1 distribution shifts from the most basal region at E11–E12 (Fig. 3a) to deeper regions at E13 and beyond (data not shown). Alternatively, progenitors' requirement for TAG-1 and/or the mechanisms of basal process morphogenesis might be regulated in a stage-dependent manner. b) The shortening (basal-disconnection) phenotype was reproduced by shRNA against a diffent TAG-1 sequence. c) The apically accumulated GFP+ cells were positive for Nestin. The overall pattern of the course of Nestin+ fibers was abnormal, and did not show a typical radial arrangement. d) Comparison of cleavage orientation of apically dividing cells between control and TAG-1–KD VZs at E12. e) Comparison of progenitors' morphology between normal and TAG-1–KD VZs. In contrast to the control progenitor cells (left), which have thin flexible apical processes after successful basalward nucleokinesis, TAG-1–deficient progenitor cells that could not send the nucleus sufficiently basally (right) were voluminous at the apical side, with much thicker processes than controls. f) When each cerebral wall (E14) was divided into apical and basal parts, the apical part still bent apically, indicating that apicalward bending of each intact wall is not secondary to expansion of the basal part. Bending/curling of cerebral wall slices occurred not only when slices were made along the ventral-to-dorsal axis (Fig. 4h and this panel), but also when sliced along the anterior-to-posterior axis (data not shown). Slices prepared at E12, E13, or E14 showed similar bending/curling. g) Apicalward bulging (arrow) and tangential expansion of a TAG-1–KD cerebral wall. Magenta is Ki67 immunoreactivity. Length between the dorsal (D) and ventral (V) border of the pallial wall part increased in TAG-1–KD henmispheres. h) Schematic illustration showing morphology and periventricular nuclear density of control and TAG-1-KD (shortened) progenitor cells. i) Laser ablation experiments on the basal surface. Cross-sectional live inspection of a cerebral wall that received a laser pulse onto the basal surface approximately 60 min before slice preparation. Basal processes (arrowhead) were retracted only slightly and cell bodies (arrow) were at normal positions during observation for 12 h.
Supplementary Figure 3 Results showing progenitor delamination in TAG-1–KD cerebral walls and schematic illustration of overcrowding induced by Wnt3a overexression
a) Close observation of progenitor delamination behavior (Fig. 5a and Supplementary Movie 10). Six progenitor cells with shortened morphologies were clustered apically. Next, weakening of the fluorescence at the apical surface was detected (black arrows), followed about 40 min later by dislocation of four cells (white arrows). It is possible that somal dislocation after apical detachment involves a pushing or squeezing force from the highly congested periventricular space. b) Stage-dependent occurrence of delamination. Although electroporation of TAG-1 shRNA at E10 always resulted in massive delamination of progenitor cells by E13, which remained abundantly at E15 (left, outlined), RNAi electroporation at E12 did not induce such a clear delamination phenotype, showing a normal pattern of Pax6 immunoreacitivy at E15 (right). c) Schematic illustration showing progenitor morphology and VZ nuclear density in control and Wnt3a-overexpressed cerebral walls. Illustration is based on data shown in Fig. 6 and another result that the averaged apical area was greater in VZ cells of Wnt3a-induced over-proliferation experiments (4.31 ± 0.19 μm2, n=181) than in VZ cells of the control (3.60 ± 0.17 μm2, n=180) (p=0.00026).
Supplementary Figure 4 Behaviors of heterotopic progenitor cells in TAG-1–KD cerebral walls
a) Different classes of neocortical neurons are sequentially produced in TAG-1–KD cerebral walls displaying a mosaic-like abnormal histogenetic pattern. Note that a certain fraction of Cux1+ neurons were found more basally than in the control, probably reflecting heterotopic production of these neurons, as suggested by in vitro analysis (Fig. 8g,h). b,c) Expression of ZO1 (b) and N-cadherin (c) at the apical surface of a TAG-1–deficient E13 cerebral wall. Aberrant expression of these “apical markers” was seen in portions where heterotopic progenitor cells formed aggregates.
Supplementary Figure 5 Schematic illustration summarizing the present study
Schematic illustration summarizing the present study. Normal basalward INM facilitated by the basal process prevents periventricular overcrowding, thereby ensuring normal histogenesis.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 9610 kb)
Supplementary Video 1
Live imaging of nuclei in the ventricular zone of the cerebral wall at E13 (MOV 69293 kb)
Supplementary Video 2
In toto tangential live imaging of the periventricular space (MOV 7885 kb)
Supplementary Video 3
Time-lapse observation of pair-generated daughter cells (MOV 7432 kb)
Supplementary Video 4
Retraction and misrouting of basal processes live-monitored in a TAG-1–KD E11 cerebral wall slice (MOV 10528 kb)
Supplementary Video 5
Live observation of the shortened progenitor cells in a slice prepared at E12 from a TAG-1–KD cerebral wall (MOV 8514 kb)
Supplementary Video 6
Apicalward nucleokinesis of non–TAG-1–KD cells affected by the congested TAG-1–KD cells in the cerebral wall of an E12 H2B-mCherry transgenic mouse (MOV 33859 kb)
Supplementary Video 7
Tangential in toto monitoring in the periventricular space of the cerebral wall of an E12 Lyn-Venus transgenic mouse embryo that had been electroporated with TAG-1 shRNA at E10 (MOV 7947 kb)
Supplementary Video 8
Centripetal bending of freshly prepared normal and TAG-1–KD E12 cerebral wall slices (MOV 28563 kb)
Supplementary Video 9
Laser experiments demonstrating contractility of the apical surface (MOV 22786 kb)
Supplementary Video 10
Overcrowding-induced delamination of progenitor cells (MOV 48969 kb)
Supplementary Video 11
Animation simulating the wild-type situation (MP4 2297 kb)
Supplementary Video 12
Animation simulating basal disconnection to mimic TAG-1–KD (MP4 2573 kb)
Supplementary Video 13
Animation simulating overproliferation corresponding to Wnt3a-overexpression (MP4 2478 kb)
Supplementary Video 14
Time-lapse observation of EB3-EGFP in control and shortened TAG-1–KD cells (MOV 53768 kb)
Rights and permissions
About this article
Cite this article
Okamoto, M., Namba, T., Shinoda, T. et al. TAG-1–assisted progenitor elongation streamlines nuclear migration to optimize subapical crowding. Nat Neurosci 16, 1556–1566 (2013). https://doi.org/10.1038/nn.3525
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3525
This article is cited by
-
Temporal morphogen gradient-driven neural induction shapes single expanded neuroepithelium brain organoids with enhanced cortical identity
Nature Communications (2023)
-
Neuronal migration prevents spatial competition in retinal morphogenesis
Nature (2023)
-
Epithelial tissue folding pattern in confined geometry
Biomechanics and Modeling in Mechanobiology (2020)
-
Using the Allen gene expression atlas of the adult mouse brain to gain further insight into the physiological significance of TAG-1/Contactin-2
Brain Structure and Function (2020)
-
A multiscale mathematical model of cell dynamics during neurogenesis in the mouse cerebral cortex
BMC Bioinformatics (2019)