Abstract
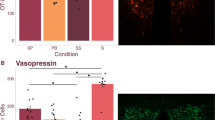
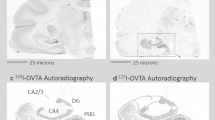
In the socially monogamous prairie vole (Microtus ochrogaster), mating induces enduring pair-bonds that are initiated by partner preference formation and regulated by a variety of neurotransmitters, including oxytocin, vasopressin and dopamine. We examined potential epigenetic mechanisms mediating pair-bond regulation and found that the histone deacetylase inhibitors sodium butyrate and trichostatin A (TSA) facilitated partner preference formation in female prairie voles in the absence of mating. This was associated with a specific upregulation of oxytocin receptor (OTR, oxtr) and vasopressin V1a receptor (V1aR, avpr1a) in the nucleus accumbens (NAcc), through an increase in histone acetylation at their respective promoters. Furthermore, TSA-facilitated partner preference was prevented by OTR or V1aR blockade in the NAcc. Notably, mating-induced partner preference triggered the same epigenetic regulation of oxtr and avpr1a gene promoters as TSA. These observations indicate that TSA and mating facilitate partner preference through epigenetic events, providing, to the best of our knowledge, the first direct evidence for epigenetic regulation of pair-bonding.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bora, E., Yucel, M. & Allen, N.B. Neurobiology of human affiliative behavior: implications for psychiatric disorders. Curr. Opin. Psychiatry 22, 320–325 (2009).
Thomas, J.A. & Birney, E.C. Parental care and mating system of the prairie vole (Microtus ochrogaster). Behav. Ecol. Sociobiol. 5, 171–186 (1979).
Getz, L.L. & Hofmann, J.E. Social organization in free-living prairie voles (Microtus ochrogaster). Behav. Ecol. Sociobiol. 18, 275–282 (1986).
Williams, J.R., Catania, K.C. & Carter, C.S. Development of partner preferences in female prairie voles (Microtus ochrogaster): the role of social and sexual experience. Horm. Behav. 26, 339–349 (1992).
Young, K.A., Gobrogge, K.L., Liu, Y. & Wang, Z. The neurobiology of pair bonding: insights from a socially monogamous rodent. Front. Neuroendocrinol. 32, 53–69 (2011).
Liu, Y., Curtis, J.T. & Wang, Z. Vasopressin in the lateral septum regulates pair bond formation in male prairie voles (Microtus ochrogaster). Behav. Neurosci. 115, 910–919 (2001).
Lim, M.M. & Young, L.J. Vasopressin-dependent neural circuits underlying pair bond formation in the monogamous prairie vole. Neuroscience 125, 35–45 (2004).
Liu, Y. & Wang, Z.X. Nucleus accumbens oxytocin and dopamine interact to regulate pair bond formation in female prairie voles. Neuroscience 121, 537–544 (2003).
Gingrich, B., Liu, Y., Cascio, C., Wang, Z. & Insel, T.R. Dopamine D2 receptors in the nucleus accumbens are important for social attachment in female prairie voles (Microtus ochrogaster). Behav. Neurosci. 114, 173–183 (2000).
Aragona, B.J. et al. Nucleus accumbens dopamine differentially mediates the formation and maintenance of monogamous pair bonds. Nat. Neurosci. 9, 133–139 (2006).
Liu, Y. et al. Nucleus accumbens dopamine mediates amphetamine-induced impairment of social bonding in a monogamous rodent species. Proc. Natl. Acad. Sci. USA 107, 1217–1222 (2010).
Keebaugh, A.C. & Young, L.J. Increasing oxytocin receptor expression in the nucleus accumbens of pre-pubertal female prairie voles enhances alloparental responsiveness and partner preference formation as adults. Horm. Behav. 60, 498–504 (2011).
Ross, H.E. et al. Variation in oxytocin receptor density in the nucleus accumbens has differential effects on affiliative behaviors in monogamous and polygamous voles. J. Neurosci. 29, 1312–1318 (2009).
Pitkow, L.J. et al. Facilitation of affiliation and pair-bond formation by vasopressin receptor gene transfer into the ventral forebrain of a monogamous vole. J. Neurosci. 21, 7392–7396 (2001).
Veenema, A.H. Toward understanding how early-life social experiences alter oxytocin- and vasopressin-regulated social behaviors. Horm. Behav. 61, 304–312 (2012).
Francis, D.D., Young, L.J., Meaney, M.J. & Insel, T.R. Naturally occurring differences in maternal care are associated with the expression of pxytocin and vasopressin (V1a) receptors: gender differences. J. Neuroendocrinol. 14, 349–353 (2002).
Champagne, F.A. et al. Maternal care associated with methylation of the estrogen receptor-α1b promoter and estrogen receptor-α expression in the medial preoptic area of female offspring. Endocrinology 147, 2909–2915 (2006).
Murgatroyd, C. et al. Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat. Neurosci. 12, 1559–1566 (2009).
Weaver, I.C.G. et al. Epigenetic programming by maternal behavior. Nat. Neurosci. 7, 847–854 (2004).
Weaver, I.C.G., Meaney, M.J. & Szyf, M. Maternal care effects on the hippocampal transcriptome and anxiety-mediated behaviors in the offspring that are reversible in adulthood. Proc. Natl. Acad. Sci. USA 103, 3480–3485 (2006).
Bonthuis, P.J., Patteson, J.K. & Rissman, E.F. Acquisition of sexual receptivity: roles of chromatin acetylation, estrogen receptor-α, and ovarian hormones. Endocrinology 152, 3172–3181 (2011).
Zhong, S., Fields, C.R., Su, N., Pan, Y.X. & Robertson, K.D. Pharmacologic inhibition of epigenetic modifications, coupled with gene expression profiling, reveals novel targets of aberrant DNA methylation and histone deacetylation in lung cancer. Oncogene 26, 2621–2634 (2007).
Yoshida, M., Horinouchi, S. & Beppu, T. Trichostatin A and trapoxin: novel chemical probes for the role of histone acetylation in chromatin structure and function. Bioessays 17, 423–430 (1995).
Monneret, C. Histone deacetylase inhibitors. Eur. J. Med. Chem. 40, 1–13 (2005).
Ross, H.E. et al. Characterization of the oxytocin system regulating affiliative behavior in female prairie voles. Neuroscience 162, 892–903 (2009).
Young, L.J., Huot, B., Nilsen, R., Wang, Z. & Insel, T.R. Species differences in central oxytocin receptor gene expression: comparative analysis of promoter sequences. J. Neuroendocrinol. 8, 777–783 (1996).
Witt, O., Deubzer, H.E., Milde, T. & Oehme, I. HDAC family: What are the cancer relevant targets? Cancer Lett. 277, 8–21 (2009).
Vecsey, C.G. et al. Histone deacetylase inhibitors enhance memory and synaptic plasticity via CREB: CBP-dependent transcriptional activation. J. Neurosci. 27, 6128–6140 (2007).
Monsey, M.S., Ota, K.T., Akingbade, I.F., Hong, E.S. & Schafe, G.E. Epigenetic alterations are critical for fear memory consolidation and synaptic plasticity in the lateral amygdala. PLoS ONE 6, e19958 (2011).
Van Lint, C., Emiliani, S. & Verdin, E. The expression of a small fraction of cellular genes is changed in response to histone hyperacetylation. Gene Expr. 5, 245–253 (1996).
Mariadason, J.M., Corner, G.A. & Augenlicht, L.H. Genetic reprogramming in pathways of colonic cell maturation induced by short chain fatty acids: comparison with Trichostatin A, sulindac, and curcumin and implications for chemoprevention of colon cancer. Cancer Res. 60, 4561–4572 (2000).
Halsall, J., Gupta, V., O'Neill, L.P., Turner, B.M. & Nightingale, K.P. Genes are often sheltered from the global histone hyperacetylation induced by HDAC inhibitors. PLoS ONE 7, e33453 (2012).
Ito, K. & Adcock, I. Histone acetylation and histone deacetylation. Mol. Biotechnol. 20, 99–106 (2002).
Molfese, D.L. Advancing neuroscience through epigenetics: molecular mechanisms of learning and memory. Dev. Neuropsychol. 36, 810–827 (2011).
Young, L.J., Lim, M.M., Gingrich, B. & Insel, T.R. Cellular mechanisms of social attachment. Horm. Behav. 40, 133–138 (2001).
Hawk, J.D., Florian, C. & Abel, T. Post-training intrahippocampal inhibition of class I histone deacetylases enhances long-term object-location memory. Learn. Mem. 18, 367–370 (2011).
Stefanko, D.P., Barrett, R.M., Ly, A.R., Reolon, G.K. & Wood, M.A. Modulation of long-term memory for object recognition via HDAC inhibition. Proc. Natl. Acad. Sci. USA 106, 9447–9452 (2009).
Yang, X.-J. & Seto, E. Lysine acetylation: codified crosstalk with other posttranslational modifications. Mol. Cell 31, 449–461 (2008).
Cho, M.M., DeVries, A.C., Williams, J.R. & Carter, C.S. The effects of oxytocin and vasopressin on partner preferences in male and female prairie voles (Microtus ochrogaster). Behav. Neurosci. 113, 1071–1079 (1999).
Insel, T.R. & Hulihan, T.J. A gender-specific mechanism for pair bonding: oxytocin and partner preference formation in monogamous voles. Behav. Neurosci. 109, 782–789 (1995).
Stimson, L. & La Thangue, N.B. Biomarkers for predicting clinical responses to HDAC inhibitors. Cancer Lett. 280, 177–183 (2009).
Iannitti, T. & Palmieri, B. Clinical and experimental applications of sodium phenylbutyrate. Drugs R D. 11, 227–249 (2011).
Kumar, A. et al. Chromatin remodeling is a key mechanism underlying cocaine-induced plasticity in striatum. Neuron 48, 303–314 (2005).
Schroeder, F.A., Lin, C.L., Crusio, W.E. & Akbarian, S. Antidepressant-like effects of the histone deacetylase inhibitor, sodium butyrate, in the mouse. Biol. Psychiatry 62, 55–64 (2007).
Manning, M. et al. Peptide and non-peptide agonists and antagonists for the vasopressin and oxytocin V1a, V1b, V2 and OT receptors: research tools and potential therapeutic agents. in Progress in Brain Research (eds. D.N. Inga & L. Rainer) 473–512 (Elsevier, 2008).
Witt, D.M. & Insel, T.R. A selective oxytocin antagonist attenuates progesterone facilitation of female sexual behavior. Endocrinology 128, 3269–3276 (1991).
Winslow, J.T., Hastings, N., Carter, C.S., Harbaugh, C.R. & Insel, T.R. A role for central vasopressin in pair bonding in monogamous prairie voles. Nature 365, 545–548 (1993).
Insel, T.R. Oxytocin–a neuropeptide for affiliation: evidence from behavioral, receptor autoradiographic, and comparative studies. Psychoneuroendocrinology 17, 3–35 (1992).
Insel, T.R., Wang, Z. & Ferris, C. Patterns of brain vasopressin receptor distribution associated with social organization in microtine rodents. J. Neurosci. 14, 5381–5392 (1994).
Hollis, F., Duclot, F., Gunjan, A. & Kabbaj, M. Individual differences in the effect of social defeat on anhedonia and histone acetylation in the rat hippocampus. Horm. Behav. 59, 331–337 (2011).
Acknowledgements
We also thank M. Manning for the generous gift of the OTR antagonist OTA(T) (University of Toledo). This work was supported by grants from the National Institute of Mental Health (MHR21-083128 to M.K. and Z.W., and MHR01-058616 to Z.W.).
Author information
Authors and Affiliations
Contributions
H.W., F.D. and Y.L. performed the experiments. H.W. and F.D. analyzed the data. H.W., F.D., Z.W. and M.K. designed the study. F.D., Z.W. and M.K. wrote the paper. All of the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4 (PDF 1011 kb)
Rights and permissions
About this article
Cite this article
Wang, H., Duclot, F., Liu, Y. et al. Histone deacetylase inhibitors facilitate partner preference formation in female prairie voles. Nat Neurosci 16, 919–924 (2013). https://doi.org/10.1038/nn.3420
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3420
This article is cited by
-
Histone H4 acetylation regulates behavioral inter-individual variability in zebrafish
Genome Biology (2018)
-
Genes Related to Oxytocin and Arginine-Vasopressin Pathways: Associations with Autism Spectrum Disorders
Neuroscience Bulletin (2017)
-
Hippocampal Acetylation may Improve Prenatal-Stress-Induced Depression-Like Behavior of Male Offspring Rats Through Regulating AMPARs Expression
Neurochemical Research (2017)