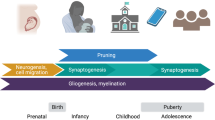
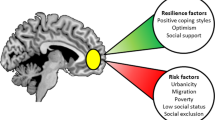
Abstract
Experiential factors shape the neural circuits underlying social and emotional behavior from the prenatal period to the end of life. These factors include both incidental influences, such as early adversity, and intentional influences that can be produced in humans through specific interventions designed to promote prosocial behavior and well-being. Here we review important extant evidence in animal models and humans. Although the precise mechanisms of plasticity are still not fully understood, moderate to severe stress appears to increase the growth of several sectors of the amygdala, whereas the effects in the hippocampus and prefrontal cortex tend to be opposite. Structural and functional changes in the brain have been observed with cognitive therapy and certain forms of meditation and lead to the suggestion that well-being and other prosocial characteristics might be enhanced through training.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Adolphs, R. Conceptual challenges and directions for social neuroscience. Neuron 65, 752–767 (2010).
Erickson, K.I. et al. Exercise training increases size of hippocampus and improves memory. Proc. Natl. Acad. Sci. USA 108, 3017–3022 (2011).
Disner, S.G., Beevers, C.G. & Haigh, E.A.P. & Beck, A.T. Neural mechanisms of the cognitive model of depression. Nat. Rev. Neurosci. 12, 467–477 (2011).
Clark, D.A. & Beck, A.T. Cognitive theory and therapy of anxiety and depression: convergence with neurobiological findings. Trends Cogn. Sci. 14, 418–424 (2010).
Lutz, A., Slagter, H., Dunne, J.D. & Davidson, R.J. Attention regulation and monitoring in meditation. Trends Cogn. Sci. 12, 163–169 (2008).
Kuhl, P.K. Brain mechanisms in early language acquisition. Neuron 67, 713–727 (2010).
Nelson, C.A. et al. Cognitive recovery in socially deprived young children: the Bucharest Early Intervention Project. Science 318, 1937–1940 (2007).
Sullivan, R.M. & Holman, P.J. Transitions in sensitive period attachment learning in infancy: the role of corticosterone. Neurosci. Biobehav. Rev. 34, 835–844 (2010).
Bavelier, D., Levi, D.M., Li, R.W., Dan, Y. & Hensch, T.K. Removing brakes on adult brain plasticity: from molecular to behavioral interventions. J. Neurosci. 30, 14964–14971 (2010).
Carlson, M.C. et al. Evidence for neurocognitive plasticity in at-risk older adults: the experience corps program. J. Gerontol. A Biol. Sci. Med. Sci. 64, 1275–1282 (2009).
Hölzel, B.K. et al. Stress reduction correlates with structural changes in the amygdala. Soc. Cogn. Affect. Neurosci. 5, 11–17 (2010).
Durlak, J.A., Weissberg, R.P., Dymnicki, A.B., Taylor, R.D. & Schellinger, K.B. The impact of enhancing students' social and emotional learning: a meta-analysis of school-based universal interventions. Child Dev. 82, 405–432 (2011).
Diamond, A. & Lee, K. Interventions shown to aid executive function development in children 4 to 12 years old. Science 333, 959–964 (2011).
Bennett, E.L., Diamond, M.C., Krech, D. & Rosenzweig, M.R. Chemical and anatomical plasticity of the brain. Science 146, 610–619 (1964).
Hebb, D.O. The Organization of Behavior: a Neuropsychological Theory (Wiley, New York, 1949).
Diamond, M.C. The aging brain: some enlightening and optimistic results. Am. Sci. 66, 66–71 (1978).
Markham, J.A. & Greenough, W.T. Experience-driven brain plasticity: beyond the synapse. Neuron Glia Biol. 1, 351–363 (2004).
McEwen, B.S. Physiology and neurobiology of stress and adaptation: central role of the brain. Physiol. Rev. 87, 873–904 (2007).
Bloss, E.B., Janssen, W.G., McEwen, B.S. & Morrison, J.H. Interactive effects of stress and aging on structural plasticity in the prefrontal cortex. J. Neurosci. 30, 6726–6731 (2010).
Liston, C. & Gan, W.-B. Glucocorticoids are critical regulators of dendritic spine development and plasticity in vivo. Proc. Natl. Acad. Sci. USA 108, 16074–16079 (2011).
Popoli, M., Yan, Z., McEwen, B.S. & Sanacora, G. The stressed synapse: the impact of stress and glucocorticoids on glutamate transmission. Nat. Rev. Neurosci. 13, 22–37 (2012).
McEwen, B.S. & Milner, T.A. Hippocampal formation: shedding light on the influence of sex and stress on the brain. Brain Res. Rev. 55, 343–355 (2007).
McEwen, B.S. & Alves, S.E. Estrogen actions in the central nervous system. Endocr. Rev. 20, 279–307 (1999).
Dumitriu, D., Rapp, P.R., McEwen, B.S. & Morrison, J.H. Estrogen and the aging brain: an elixir for the weary cortical network. Ann. NY Acad. Sci. 1204, 104–112 (2010).
Chen, J.-R. et al. Gonadal hormones modulate the dendritic spine densities of primary cortical pyramidal neurons in adult female rat. Cereb. Cortex 19, 2719–2727 (2009).
Cameron, H.A. & Gould, E. The control of neuronal birth and survival. in Receptor Dynamics in Neural Development (ed. C.A. Shaw) 141–157 (CRC Press, 1996).
Kaplan, M.S. Environment complexity stimulates visual cortex neurogenesis: death of a dogma and a research career. Trends Neurosci. 24, 617–620 (2001).
Altman, J. & Bayer, S.A. Mosaic organization of the hippocampal neuroepithelium and the multiple germinal sources of dentate granule cells. J. Comp. Neurol. 301, 325–342 (1990).
Nottebohm, F. From bird song to neurogenesis. Sci. Am. 260, 74–79 (1989).
Brown, J. et al. Enriched environment and physical activity stimulate hippocampal but not olfactory bulb neurogenesis. Eur. J. Neurosci. 17, 2042–2046 (2003).
Kozorovitskiy, Y. & Gould, E. Dominance hierarchy influences adult neurogenesis in the dentate gyrus. J. Neurosci. 24, 6755–6759 (2004).
Gould, E., McEwen, B.S., Tanapat, P., Galea, L.A. & Fuchs, E. Neurogenesis in the dentate gyrus of the adult tree shrew is regulated by psychosocial stress and NMDA receptor activation. J. Neurosci. 17, 2492–2498 (1997).
Magariños, A.M., McEwen, B.S., Flügge, G. & Fuchs, E. Chronic psychosocial stress causes apical dendritic atrophy of hippocampal CA3 pyramidal neurons in subordinate tree shrews. J. Neurosci. 16, 3534–3540 (1996).
Vyas, A., Mitra, R., Shankaranarayana Rao, B.S. & Chattarji, S. Chronic stress induces contrasting patterns of dendritic remodeling in hippocampal and amygdaloid neurons. J. Neurosci. 22, 6810–6818 (2002).
Liston, C. et al. Stress-induced alterations in prefrontal cortical dendritic morphology predict selective impairments in perceptual attentional set-shifting. J. Neurosci. 26, 7870–7874 (2006).
McEwen, B.S. & Gianaros, P.J. Stress- and allostasis-induced brain plasticity. Annu. Rev. Med. 62, 431–445 (2011).
Shonkoff, J.P., Boyce, W.T. & McEwen, B.S. Neuroscience, molecular biology and the childhood roots of health disparities: building a new framework for health promotion and disease prevention. J. Am. Med. Assoc. 301, 2252–2259 (2009).
Anda, R.F., Butchart, A., Felitti, V.J. & Brown, D.W. Building a framework for global surveillance of the public health implications of adverse childhood experiences. Am. J. Prev. Med. 39, 93–98 (2010).
Isgor, C., Kabbaj, M., Akil, H. & Watson, S.J. Delayed effects of chronic variable stress during peripubertal-juvenile period on hippocampal morphology and on cognitive and stress axis functions in rats. Hippocampus 14, 636–648 (2004).
Moriceau, S. & Sullivan, R.M. Maternal presence serves as a switch between learning fear and attraction in infancy. Nat. Neurosci. 9, 1004–1006 (2006).
Kaufman, D. et al. Early appearance of the metabolic syndrome in socially reared bonnet macaques. J. Clin. Endocrinol. Metab. 90, 404–408 (2005).
Coplan, J.D. et al. Variable foraging demand rearing: Sustained elevations in cisternal cerebrospinal fluid corticotropin-releasing factor concentrations in adult primates. Biol. Psychiatry 50, 200–204 (2001).
Christoffel, D.J. et al. IkB kinase regulates social defeat stress-induced synaptic and behavioral plasticity. J.Neurosci. 31, 314–321 (2011).
Miczek, K.A., Yap, J.J. & Covington, H.E. Social stress, therapeutics and drug abuse: preclinical models of escalated and depressed intake. Pharmacol. Ther. 120, 102–128 (2008).
Robinson, T.E. & Kolb, B. Structural plasticity associated with exposure to drugs of abuse. Neuropharmacology 47 Suppl 1, 33–46 (2004).
Russo, S.J. et al. The addicted synapse: mechanisms of synaptic and structural plasticity in nucleus accumbens. Trends Neurosci. 33, 267–276 (2010).
Roitman, M.F., Na, E., Anderson, G., Jones, T.A. & Bernstein, I.L. Induction of a salt appetite alters dendritic morphology in nucleus accumbens and sensitizes rats to amphetamine. J. Neurosci. 22, RC225 (2002).
Levine, S., Haltmeyer, G.C., Karas, G.G. & Denenberg, V.H. Physiological and behavioral effects of infantile stimulation. Physiol. Behav. 2, 55–59 (1967).
Meaney, M.J. & Szyf, M. Environmental programming of stress responses through DNA methylation: life at the interface between a dynamic environment and a fixed genome. Dialogues Clin. Neurosci. 7, 103–123 (2005).
Akers, K.G. et al. Social competitiveness and plasticity of neuroendocrine function in old age: influence of neonatal novelty exposure and maternal care reliability. PLoS ONE 3, e2840 (2008).
Tang, A.C., Akers, K.G., Reeb, B.C., Romeo, R.D. & McEwen, B.S. Programming social, cognitive and neuroendocrine development by early exposure to novelty. Proc. Natl. Acad. Sci. USA 103, 15716–15721 (2006).
Parker, K.J. & Maestripieri, D. Identifying key features of early stressful experiences that produce stress vulnerability and resilience in primates. Neurosci. Biobehav. Rev. 35, 1466–1483 (2011).
van Hasselt, F.N. et al. Adult hippocampal glucocorticoid receptor expression and dentate synaptic plasticity correlate with maternal care received by individuals early in life. Hippocampus 22, 255–266 (2011).
Parker, K.J., Buckmaster, C.L., Schatzberg, A.F. & Lyons, D.M. Prospective investigation of stress inoculation in young monkeys. Arch. Gen. Psychiatry 61, 933–941 (2004).
Parker, K.J., Buckmaster, C.L., Justus, K.R., Schatzberg, A.F. & Lyons, D.M. Mild early life stress enhances prefrontal-dependent response inhibition in monkeys. Biol. Psychiatry 57, 848–855 (2005).
Katz, M. et al. Prefrontal plasticity and stress inoculation-induced resilience. Dev. Neurosci. 31, 293–299 (2009).
Maya Vetencourt, J.F. et al. The antidepressant fluoxetine restores plasticity in the adult visual cortex. Science 320, 385–388 (2008).
Spolidoro, M. et al. Food restriction enhances visual cortex plasticity in adulthood. Nature Commun. 2, 320 (2011).
Southwell, D.G., Froemke, R.C., Alvarez-Buylla, A., Stryker, M.P. & Gandhi, S.P. Cortical plasticity induced by inhibitory neuron transplantation. Science 327, 1145–1148 (2010).
Duman, R.S. & Monteggia, L.M. A neurotrophic model for stress-related mood disorders. Biol. Psychiatry 59, 1116–1127 (2006).
Chollet, F. et al. Fluoxetine for motor recovery after acute ischaemic stroke (FLAME): a randomized placebo-controlled trial. Lancet Neurol. 10, 123–130 (2011).
Castrén, E. & Rantamäki, T. The role of BDNF and its receptors in depression and antidepressant drug action: reactivation of developmental plasticity. Dev. Neurobiol. 70, 289–297 (2010).
Shonkoff, J.P. & Garner, A.S. The lifelong effects of early childhood adversity and toxic stress. Pediatrics 129, 232–246 (2012).
Shonkoff, J.P. Protecting brains, not simply stimulating minds. Science 333, 982–983 (2011).
Heim, C., Shugart, M., Craighead, W.E. & Nemeroff, C.B. Neurobiological and psychiatric consequences of child abuse and neglect. Dev. Psychobiol. 52, 671–690 (2010).
Gould, F. et al. The effects of child abuse and neglect on cognitive functioning in adulthood. J. Psychiatr. Res. 46, 500–506 (2012).
Choi, J., Jeong, B., Rohan, M.L., Polcari, A.M. & Teicher, M.H. Preliminary evidence for white matter tract abnormalities in young adults exposed to parental verbal abuse. Biol. Psychiatry 65, 227–234 (2009).
Hanson, J.L. et al. Early stress is associated with alterations in the orbitofrontal cortex: a tensor-based morphometry investigation of brain structure and behavioral risk. J. Neurosci. 30, 7466–7472 (2010).
Loman, M.M. & Gunnar, M.R. Early experience and the development of stress reactivity and regulation in children. Neurosci. Biobehav. Rev. 34, 867–876 (2010).
McGowan, P.O. et al. Epigenetic regulation of the glucocorticoid receptor in human brain associates with childhood abuse. Nat. Neurosci. 12, 342–348 (2009).
Tottenham, N. et al. Prolonged institutional rearing is associated with atypically large amygdala volume and difficulties in emotion regulation. Dev. Sci. 13, 46–61 (2010).
Lupien, S.J. et al. Larger amygdala but no change in hippocampal volume in 10-year-old children exposed to maternal depressive symptomatology since birth. Proc. Natl. Acad. Sci. USA 108, 14324–14329 (2011).
Morey, R.A. et al. A comparison of automated segmentation and manual tracing for quantifying hippocampal and amygdala volumes. Neuroimage 45, 855–866 (2009).
Tottenham, N. & Sheridan, M.A. A review of adversity, the amygdala and the hippocampus: a consideration of developmental timing. Front. Hum. Neurosci. 3, 68 (2009).
Nacewicz, B.M. et al. Amygdala volume and nonverbal social impairment in adolescent and adult males with autism. Arch. Gen. Psychiatry 63, 1417–1428 (2006).
Mosconi, M.W. et al. Longitudinal study of amygdala volume and joint attention in 2- to 4-year-old children with autism. Arch. Gen. Psychiatry 66, 509–516 (2009).
Ochsner, K.N. & Gross, J.J. The cognitive control of emotion. Trends Cogn. Sci. 9, 242–249 (2005).
Wager, T.D., Davidson, M.L., Hughes, B.L. & Lindquist, M.A. & Ochsner, K.N. Prefrontal-subcortical pathways mediating successful emotion regulation. Neuron 59, 1037–1050 (2008).
Urry, H.L. et al. Amygdala and ventromedial prefrontal cortex are inversely coupled during regulation of negative affect and predict the diurnal pattern of cortisol secretion among older adults. J. Neurosci. 26, 4415–4425 (2006).
Davidson, R.J. Well-being and affective style: neural substrates and biobehavioural correlates. Philos. Trans. R. Soc. Lond. B Biol. Sci. 359, 1395–1411 (2004).
Davidson, R.J., Putnam, K.M. & Larson, C.L. Dysfunction in the neural circuitry of emotion regulation: a possible prelude to violence. Science 289, 591–594 (2000).
Johnstone, T., van Reekum, C.M., Urry, H.L., Kalin, N.H. & Davidson, R.J. Failure to regulate: counterproductive recruitment of top-down prefrontal-subcortical circuitry in major depression. J. Neurosci. 27, 8877–8884 (2007).
Kim, M.J. & Whalen, P.J. The structural integrity of an amygdala-prefrontal pathway predicts trait anxiety. J. Neurosci. 29, 11614–8 (2009).
Coan, J.A., Schaefer, H.S. & Davidson, R.J. Lending a hand: social regulation of the neural response to threat. Psychol. Sci. 17, 1032–1039 (2006).
Uchino, B.N., Cacioppo, J.T. & Kiecolt-Glaser, J.K. The relationship between social support and physiological processes: a review with emphasis on underlying mechanisms and implications for health. Psychol. Bull. 119, 488–531 (1996).
Buss, C. et al. Maternal care modulates the relationship between prenatal risk and hippocampal volume in women but not in men. J. Neurosci. 27, 2592–5 (2007).
Fava, G.A., Rafanelli, C., Cazzaro, M., Conti, S. & Grandi, S. Well-being therapy. A novel psychotherapeutic approach for residual symptoms of affective disorders. Psychol. Med. 28, 475–480 (1998).
DeRubeis, R.J., Siegle, G.J. & Hollon, S.D. Cognitive therapy versus medication for depression: treatment outcomes and neural mechanisms. Nat. Rev. Neurosci. 9, 788–796 (2008).
de Lange, F.P. et al. Increase in prefrontal cortical volume following cognitive behavioural therapy in patients with chronic fatigue syndrome. Brain 131, 2172–2180 (2008).
Hofmann, S.G., Grossman, P. & Hinton, D.E. Loving-kindness and compassion meditation: potential for psychological interventions. Clin. Psychol. Rev. 31, 1126–1132 (2011).
Lutz, A., Greischar, L.L., Rawlings, N.B., Ricard, M. & Davidson, R.J. Long-term meditators self-induce high-amplitude gamma synchrony during mental practice. Proc. Natl. Acad. Sci. USA 101, 16369 (2004).
Lutz, A., Brefczynski-Lewis, J., Johnstone, T. & Davidson, R.J. Regulation of the neural circuitry of emotion by compassion meditation: effects of meditative expertise. PLoS One 3, e1897 (2008).
Uhlhaas, P.J., Roux, F., Rodriguez, E., Rotarska-Jagiela, A. & Singer, W. Neural synchrony and the development of cortical networks. Trends Cogn. Sci. 14, 72–80 (2010).
Kemeny, M.E. et al. Contemplative/emotion training reduces negative emotional behavior and promotes prosocial responses. Emotion published online (12 December 2011).
Farb, N.A. et al. Attending to the present: mindfulness meditation reveals distinct neural modes of self-reference. Soc. Cogn. Affect. Neurosci. 2, 313–322 (2007).
Brewer, J.A. et al. Meditation experience is associated with differences in default mode network activity and connectivity. Proc. Natl. Acad. Sci. USA 108, 1–6 (2011).
Christoff, K., Gordon, A.M., Smallwood, J., Smith, R. & Schooler, J.W. Experience sampling during fMRI reveals default network and executive system contributions to mind wandering. Proc. Natl. Acad. Sci. USA 106, 8719–8724 (2009).
Killingsworth, M.A. & Gilbert, D.T. A wandering mind is an unhappy mind. Science 330, 932 (2010).
Chambers, R., Gullone, E. & Allen, N.B. Mindful emotion regulation: an integrative review. Clin. Psychol. Rev. 29, 560–572 (2009).
Moffitt, T.E. et al. A gradient of childhood self-control predicts health, wealth, and public safety. Proc. Natl. Acad. Sci. USA 108, 2693–2698 (2011).
Acknowledgements
R.J.D. is supported by grants from the National Institute of Mental Health (R01-MH43454 and P50-MH084051), the National Center for Complementary and Alternative Medicine (P01-AT004952), the Fetzer Institute, the John Templeton Foundation, and gifts from Bryant Wangard and Ralph Robinson, Ann Down, Keith and Arlene Bronstein, and the John W. Kluge Foundation. B.S.M. is supported by US National Institutes of Health grants R01 MH41256 and 5P01 MH58911.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Davidson, R., McEwen, B. Social influences on neuroplasticity: stress and interventions to promote well-being. Nat Neurosci 15, 689–695 (2012). https://doi.org/10.1038/nn.3093
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3093
This article is cited by
-
Environmental and sociocultural factors are associated with pain-related brain structure among diverse individuals with chronic musculoskeletal pain: intersectional considerations
Scientific Reports (2024)
-
The role of attitudes towards contradiction in psychological resilience: the cortical mechanism of conflicting resolution networks
Scientific Reports (2024)
-
The neuropsychopharmacology of acetyl-L-carnitine (LAC): basic, translational and therapeutic implications
Discover Mental Health (2024)
-
Assessing the Effect of a 2-Year Mindfulness-Based Training Programme on Personal and Professional Functioning: A Mixed-Methods Study
Mindfulness (2024)
-
Chronic stress and turnover intention of resident physicians after experiencing COVID-19
BMC Medical Education (2023)