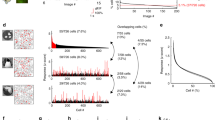
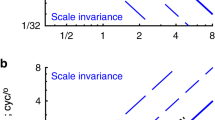
Abstract
Receptive fields in primary visual cortex (V1) are categorized as simple or complex, depending on their spatial selectivity to stimulus contrast polarity. We studied the dependence of this classification on visual context by comparing, in the same cell, the synaptic responses to three classical receptive field mapping protocols: sparse noise, ternary dense noise and flashed Gabor noise. Intracellular recordings revealed that the relative weights of simple-like and complex-like receptive field components were scaled so as to make the same receptive field more simple-like with dense noise stimulation and more complex-like with sparse or Gabor noise stimulations. However, once these context-dependent receptive fields were convolved with the corresponding stimulus, the balance between simple-like and complex-like contributions to the synaptic responses appeared to be invariant across input statistics. This normalization of the linear/nonlinear input ratio suggests a previously unknown form of homeostatic control of V1 functional properties, optimizing the network nonlinearities to the statistical structure of the visual input.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hubel, D.H. & Wiesel, T. Receptive fields, binocular interaction and functional architecture in the cat's visual cortex. J. Physiol. (Lond.) 160, 106–154 (1962).
Movshon, J.A., Thompson, I. & Tolhurst, D. Spatial summation in the receptive fields of simple cells in the cat's striate cortex. J. Physiol. (Lond.) 283, 53–77 (1978).
DeAngelis, G.C., Ohzawa, I. & Freeman, R.D. Spatiotemporal organization of simple-cell receptive fields in the cat's striate cortex. II. Linearity of temporal and spatial summation. J. Neurophysiol. 69, 1118–1135 (1993).
Carandini, M. et al. Do we know what the early visual system does? J. Neurosci. 25, 10577–10597 (2005).
Movshon, J.A., Thompson, I. & Tolhurst, D. Receptive field organization of complex cells in the cat's striate cortex. J. Physiol. (Lond.) 283, 79–99 (1978).
Dean, A.F. & Tolhurst, D.J. On the distinctness of simple and complex cells in the visual cortex of the cat. J. Physiol. (Lond.) 344, 305–325 (1983).
Emerson, R.C. et al. Nonlinear directionally selective subunits in complex cells of cat striate cortex. J. Neurophysiol. 58, 33–65 (1987).
Skottun, B.C. et al. Classifying simple and complex cells on the basis of response modulation. Vision Res. 31, 1079–1086 (1991).
Priebe, N.J. et al. The contribution of spike threshold to the dichotomy of cortical simple and complex cells. Nat. Neurosci. 7, 1113–1122 (2004).
Chance, F.S., Nelson, S.B. & Abbott, L.F. Complex cells as cortically amplified simple cells. Nat. Neurosci. 2, 277–282 (1999).
Tao, L. et al. An egalitarian network model for the emergence of simple and complex cells in visual cortex. Proc. Natl. Acad. Sci. USA 101, 366–371 (2004).
Rust, N.C. et al. Spatiotemporal elements of macaque v1 receptive fields. Neuron 46, 945–956 (2005).
Bardy, C. et al. “Simplification” of responses of complex cells in cat striate cortex: suppressive surrounds and “feedback” inactivation. J. Physiol. (Lond.) 574, 731–750 (2006).
Victor, J.D. et al. Responses of V1 neurons to two-dimensional hermite functions. J. Neurophysiol. 95, 379–400 (2006).
Albrecht, D.G., Farrar, S.B. & Hamilton, D.B. Spatial contrast adaptation characteristics of neurones recorded in the cat's visual cortex. J. Physiol. (Lond.) 347, 713–739 (1984).
Ohzawa, I., Sclar, G. & Freeman, R.D. Contrast gain control in the cat's visual system. J. Neurophysiol. 54, 651–667 (1985).
Carandini, M. & Ferster, D. A tonic hyperpolarization underlying contrast adaptation in cat visual cortex. Science 276, 949–952 (1997).
Sanchez-Vives, M.V., Nowak, L.G. & McCormick, D.A. Membrane mechanisms underlying contrast adaptation in cat area 17 in vivo. J. Neurosci. 20, 4267–4285 (2000).
Sharpee, T.O. et al. Adaptive filtering enhances information transmission in visual cortex. Nature 439, 936–942 (2006).
Felsen, G. et al. Cortical sensitivity to visual features in natural scenes. PLoS Biol. 3, e342 (2005).
David, S.V., Vinje, W.E. & Gallant, J.L. Natural stimulus statistics alter the receptive field structure of v1 neurons. J. Neurosci. 24, 6991–7006 (2004).
Yeh, C.I. et al. Stimulus ensemble and cortical layer determine V1 spatial receptive fields. Proc. Natl. Acad. Sci. USA 106, 14652–14657 (2009).
Victor, J.D. et al. Laminar and orientation-dependent characteristics of spatial nonlinearities: implications for the computational architecture of visual cortex. J. Neurophysiol. 102, 3414–3432 (2009).
Mata, M.L. & Ringach, D.L. Spatial overlap of ON and OFF subregions and its relation to response modulation ratio in macaque primary visual. J. Neurophysiol. 93, 919–928 (2005).
Stryker, M. & Blakemore, C. Saccadic and disjunctive eye movements in cats. Vision Res. 12, 2005–2013 (1972).
Carandini, M. & Heeger, D.J. Summation and division by neurons in primate visual cortex. Science 264, 1333–1336 (1994).
Crowder, N.A. et al. Complex cells increase their phase sensitivity at low contrasts and following adaptation. J. Neurophysiol. 98, 1155–1166 (2007).
Aertsen, A.M. et al. Dynamics of neuronal firing correlation: modulation of “effective connectivity”. J. Neurophysiol. 61, 900–917 (1989).
Martinez, L.M. & Alonso, J.M. Construction of complex receptive fields in cat primary visual cortex. Neuron 32, 515–525 (2001).
Martinez, L.M. et al. Receptive field structure varies with layer in the primary visual cortex. Nat. Neurosci. 8, 372–379 (2005).
LeVay, S. & Gilbert, C.D. Laminar patterns of geniculocortical projection in the cat. Brain Res. 113, 1–19 (1976).
Lesica, N.A. et al. Adaptation to stimulus contrast and correlations during natural visual stimulation. Neuron 55, 479–491 (2007).
Smirnakis, S.M. et al. Adaptation of retinal processing to image contrast and spatial scale. Nature 386, 69–73 (1997).
So, Y.T. & Shapley, R. Spatial tuning of cells in and around lateral geniculate nucleus of the cat: X and Y relay cells and perigeniculate interneurons. J. Neurophysiol. 45, 107–120 (1981).
Sillito, A.M. The contribution of inhibitory mechanisms to the receptive field properties of neurones in the striate cortex of the cat. J. Physiol. (Lond.) 250, 305–329 (1975).
Troyer, T.W. et al. Contrast-invariant orientation tuning in cat visual cortex: thalamocortical input tuning and correlation-based intracortical connectivity. J. Neurosci. 18, 5908–5927 (1998).
Lauritzen, T.Z. & Miller, K.D. Different roles for simple-cell and complex-cell inhibition in V1. J. Neurosci. 23, 10201–10213 (2003).
Hirsch, J.A. et al. Functionally distinct inhibitory neurons at the first stage of visual cortical processing. Nat. Neurosci. 6, 1300–1308 (2003).
Liu, B.H. et al. Visual receptive field structure of cortical inhibitory neurons revealed by two-photon imaging guided recording. J. Neurosci. 29, 10520–10532 (2009).
Debanne, D., Shulz, D.E. & Fregnac, Y. Activity-dependent regulation of “on” and “off” responses in cat visual cortical receptive fields. J. Physiol. (Lond.) 508, 523–548 (1998).
Nauhaus, I. et al. Stimulus contrast modulates functional connectivity in visual cortex. Nat. Neurosci. 12, 70–76 (2009).
Müller, J.R. et al. Rapid adaptation in visual cortex to the structure of images. Science 285, 1405–1408 (1999).
Felsen, G. et al. Dynamic modification of cortical orientation tuning mediated by recurrent connections. Neuron 36, 945–954 (2002).
Nagel, K.I. & Doupe, A.J. Temporal processing and adaptation in the songbird auditory forebrain. Neuron 51, 845–859 (2006).
Maravall, M. et al. Shifts in coding properties and maintenance of information transmission during adaptation in barrel cortex. PLoS Biol. 5, e19 (2007).
Higley, M.J. & Contreras, D. Frequency adaptation modulates spatial integration of sensory responses in the rat whisker system. J. Neurophysiol. 97, 3819–3824 (2007).
Brenner, N., Bialek, W. & de Ruyter van Steveninck, R. Adaptive rescaling maximizes information transmission. Neuron 26, 695–702 (2000).
Wainwright, M.J. Visual adaptation as optimal information transmission. Vision Res. 39, 3960–3974 (1999).
Westwick, D.T. & Kearney, R.E. Identification of Nonlinear Physiological Systems (Wiley-IEEE, 2003).
Acknowledgements
We are thankful to G. Sadoc for his invaluable technical assistance in developing the stimulation software and kernel analysis library tools. We thank A. Davison for comments and suggestions on the manuscript. We are thankful to Z. Kisvarday and K. Sari for their help in the biocytin labeling protocol. We acknowledge the financial support of CNRS, the Agence Nationale de la Recherche (Natstats and V1-Complex), European community contracts Facets (FP6-2004-IST-FETPI 15879) and Brain-i-nets (FP7-2009-ICT-FET 243914).
Author information
Authors and Affiliations
Contributions
The study was conceived by J.F., C.M. and Y.F. The experiments were performed by J.F., C.M. and M.P. J.F. performed the data analysis and model simulations. J.F., C.M. and Y.F. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 and Notes 1–3 (PDF 1809 kb)
Rights and permissions
About this article
Cite this article
Fournier, J., Monier, C., Pananceau, M. et al. Adaptation of the simple or complex nature of V1 receptive fields to visual statistics. Nat Neurosci 14, 1053–1060 (2011). https://doi.org/10.1038/nn.2861
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2861
This article is cited by
-
Efficient and adaptive sensory codes
Nature Neuroscience (2021)
-
A large-scale standardized physiological survey reveals functional organization of the mouse visual cortex
Nature Neuroscience (2020)
-
Practical realization of discrete-time Volterra series for high-order nonlinearities
Nonlinear Dynamics (2019)
-
The feature-specific propagation of orientation and direction adaptation from areas 17 to 21a in cats
Scientific Reports (2017)
-
Spatiotemporal receptive fields of barrel cortex revealed by reverse correlation of synaptic input
Nature Neuroscience (2014)