Abstract
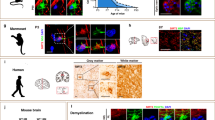
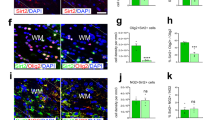
Permanent damage to white matter tracts, comprising axons and myelinating oligodendrocytes, is an important component of brain injuries of the newborn that cause cerebral palsy and cognitive disabilities, as well as multiple sclerosis in adults. However, regulatory factors relevant in human developmental myelin disorders and in myelin regeneration are unclear. We found that AXIN2 was expressed in immature oligodendrocyte progenitor cells (OLPs) in white matter lesions of human newborns with neonatal hypoxic-ischemic and gliotic brain damage, as well as in active multiple sclerosis lesions in adults. Axin2 is a target of Wnt transcriptional activation that negatively feeds back on the pathway, promoting β-catenin degradation. We found that Axin2 function was essential for normal kinetics of remyelination. The small molecule inhibitor XAV939, which targets the enzymatic activity of tankyrase, acted to stabilize Axin2 levels in OLPs from brain and spinal cord and accelerated their differentiation and myelination after hypoxic and demyelinating injury. Together, these findings indicate that Axin2 is an essential regulator of remyelination and that it might serve as a pharmacological checkpoint in this process.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Back, S.A. et al. Selective vulnerability of late oligodendrocyte progenitors to hypoxia-ischemia. J. Neurosci. 22, 455–463 (2002).
Khwaja, O. & Volpe, J.J. Pathogenesis of cerebral white matter injury of prematurity. Arch. Dis. Child. Fetal Neonatal Ed. 93, F153–F161 (2008).
Woodward, L.J., Anderson, P.J., Austin, N.C., Howard, K. & Inder, T.E. Neonatal MRI to predict neurodevelopmental outcomes in preterm infants. N. Engl. J. Med. 355, 685–694 (2006).
Compston, A. & Coles, A. Multiple sclerosis. Lancet 372, 1502–1517 (2008).
Chang, A., Tourtellotte, W.W., Rudick, R. & Trapp, B.D. Premyelinating oligodendrocytes in chronic lesions of multiple sclerosis. N. Engl. J. Med. 346, 165–173 (2002).
Kuhlmann, T. et al. Differentiation block of oligodendroglial progenitor cells as a cause for remyelination failure in chronic multiple sclerosis. Brain 131, 1749–1758 (2008).
Káradóttir, R., Cavelier, P., Bergersen, L.H. & Attwell, D. NMDA receptors are expressed in oligodendrocytes and activated in ischaemia. Nature 438, 1162–1166 (2005).
Billiards, S.S. et al. Myelin abnormalities without oligodendrocyte loss in periventricular leukomalacia. Brain Pathol. 18, 153–163 (2008).
Segovia, K.N. et al. Arrested oligodendrocyte lineage maturation in chronic perinatal white matter injury. Ann. Neurol. 63, 520–530 (2008).
Fancy, S.P.J. et al. Dysregulation of the Wnt pathway inhibits timely myelination and remyelination in the mammalian CNS. Genes Dev. 23, 1571–1585 (2009).
Ye, F. et al. HDAC1 and HDAC2 regulate oligodendrocyte differentiation by disrupting the beta-catenin–TCF interaction. Nat. Neurosci. 12, 829–838 (2009).
Feigenson, K., Reid, M., See, J., Crenshaw, E.B. III & Grinspan, J.B. Wnt signaling is sufficient to perturb oligodendrocyte maturation. Mol. Cell. Neurosci. 42, 255–265 (2009).
Ikeda, S. et al. Axin, a negative regulator of the Wnt signaling pathway, forms a complex with GSK-3beta and beta-catenin and promotes GSK-3beta–dependent phosphorylation of beta-catenin. EMBO J. 17, 1371–1384 (1998).
Behrens, J. et al. Functional interaction of an axin homolog, conductin, with beta-catenin, APC, and GSK3beta. Science 280, 596–599 (1998).
Molenaar, M. et al. XTcf-3 transcription factor mediates beta-catenin–induced axis formation in Xenopus embryos. Cell 86, 391–399 (1996).
Behrens, J. et al. Functional interaction of beta-catenin with the transcription factor LEF-1. Nature 382, 638–642 (1996).
Jho, E.H. et al. Wnt/beta-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol. Cell. Biol. 22, 1172–1183 (2002).
Lustig, B. et al. Negative feedback loop of Wnt signaling through upregulation of conductin/axin2 in colorectal and liver tumors. Mol. Cell. Biol. 22, 1184–1193 (2002).
Zeng, Y.A. & Nusse, R. Wnt proteins are self-renewal factors for mammary stem cells and promote their long-term expansion in culture. Cell Stem Cell 6, 568–577 (2010).
Dessaud, E., McMahon, A.P. & Briscoe, J. Pattern formation in the vertebrate neural tube: a sonic hedgehog morphogen-regulated transcriptional network. Development 135, 2489–2503 (2008).
Dao, D.Y. et al. Axin2 regulates chondrocyte maturation and axial skeletal development. J. Orthop. Res. 28, 89–95 (2010).
Emery, B. et al. Myelin gene regulatory factor is a critical transcriptional regulator required for CNS myelination. Cell 138, 172–185 (2009).
Franklin, R.J.M. & ffrench-Constant, C. Remyelination in the CNS: from biology to therapy. Nat. Rev. Neurosci. 9, 839–855 (2008).
Arnett, H.A. et al. bHLH transcription factor Olig1 is required to repair demyelinated lesions in the CNS. Science 306, 2111–2115 (2004).
Huang, S.M. et al. Tankyrase inhibition stabilizes axin and antagonizes Wnt signaling. Nature 461, 614–620 (2009).
Huang, J.K. et al. Retinoid X receptor gamma signaling accelerates CNS remyelination. Nat. Neurosci. 14, 45–53 (2011).
Duncan, I.D., Brower, A., Kondo, Y., Curlee, J.F. Jr. & Schultz, R.D. Extensive remyelination of the CNS leads to functional recovery. Proc. Natl. Acad. Sci. USA 106, 6832–6836 (2009).
Kotter, M.R., Li, W.W., Zhao, C. & Franklin, R.J.M. Myelin impairs CNS remyelination by inhibiting oligodendrocyte precursor cell differentiation. J. Neurosci. 26, 328–332 (2006).
Mi, S. et al. LINGO-1 negatively regulates myelination by oligodendrocytes. Nat. Neurosci. 8, 745–751 (2005).
Zhang, Y. et al. Notch1 signaling plays a role in regulating precursor differentiation during CNS remyelination. Proc. Natl. Acad. Sci. USA 106, 19162–19167 (2009).
Watkins, T.A., Emery, B., Mulinyawe, S. & Barres, B.A. Distinct stages of myelination regulated by gamma-secretase and astrocytes in a rapidly myelinating CNS coculture system. Neuron 60, 555–569 (2008).
Silbereis, J.C., Huang, E.J., Back, S.A. & Rowitch, D.H. Towards improved animal models of neonatal white matter injury associated with cerebral palsy. Dis. Model Mech. 3, 678–688 (2010).
Lock, C. et al. Gene-microarray analysis of multiple sclerosis lesions yields new targets validated in autoimmune encephalomyelitis. Nat. Med. 8, 500–508 (2002).
Schüller, U. et al. Acquisition of granule neuron precursor identity is a critical determinant of progenitor cell competence to form Shh-induced medulloblastoma. Cancer Cell 14, 123–134 (2008).
Harada, N. et al. Intestinal polyposis in mice with a dominant-stable mutation of the beta-catenin gene. EMBO J. 18, 5931–5942 (1999).
Harrington, E. et al. Oligodendrocyte PTEN is required for myelin and axonal integrity, not remyelination. Ann. Neurol. 68, 703–716 (2010).
Birgbauer, E., Rao, T.S. & Webb, M. Lysolecithin induces demyelination in vitro in a cerebellar slice culture system. J. Neurosci. Rev. 78, 157–166 (2004).
Acknowledgements
We thank W. Birchmeier for Axin2-lacZ mice. This work was supported by a Promise 2010 grant from the National Multiple Sclerosis Society (to R.J.M.F. and D.H.R.), the United Kingdom Multiple Sclerosis Society (to R.J.M.F.), the US National Institutes of Health (NS040511 and NS047572 to D.H.R.) and the Medical Scientist Training Program at the University of California, San Francisco (to E.P.H.). S.E.B. is a Harry Weaver Neuroscience Scholar of the National Multiple Sclerosis Society. R.N. and D.H.R. are Howard Hughes Medical Institute Investigators.
Author information
Authors and Affiliations
Contributions
S.P.J.F. helped conceive the project and performed all experiments and analysis, with the exception of the following. E.P.H. performed and analyzed all experiments related to in vitro OLP cultures. T.J.Y. performed and analyzed the ex vivo cerebellar slice cultures. J.C.S. helped analyze Wnt pathway activation in murine hypoxic injury. C.Z. performed the electron microscopy and C.C.B. performed the G ratio analysis. S.E.B. performed bioinformatics. J.J.O. and E.J.H. procured human brain developmental tissue. R.J.M.F. and D.H.R. conceived the experiments and oversaw all aspects of the analysis. The paper was written by S.P.J.F., R.N., R.J.M.F. and D.H.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13 and Supplementary Table 1 (PDF 7637 kb)
Rights and permissions
About this article
Cite this article
Fancy, S., Harrington, E., Yuen, T. et al. Axin2 as regulatory and therapeutic target in newborn brain injury and remyelination. Nat Neurosci 14, 1009–1016 (2011). https://doi.org/10.1038/nn.2855
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2855
This article is cited by
-
Tankyrase inhibition interferes with junction remodeling, induces leakiness, and disturbs YAP1/TAZ signaling in the endothelium
Naunyn-Schmiedeberg's Archives of Pharmacology (2024)
-
Localized microglia dysregulation impairs central nervous system myelination in development
Acta Neuropathologica Communications (2023)
-
Higher throughput workflow with sensitive, reliable and automatic quantification of myelination in vitro suitable for drug screening
Scientific Reports (2023)
-
Neuroglial components of brain lesions may provide new therapeutic strategies for multiple sclerosis
Neurological Sciences (2023)
-
Signaling mechanisms involved in the regulation of remyelination in multiple sclerosis: a mini review
Journal of Molecular Medicine (2023)