Abstract
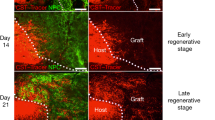
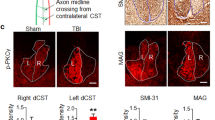
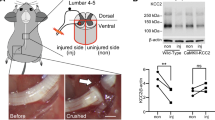
Despite the essential role of the corticospinal tract (CST) in controlling voluntary movements, successful regeneration of large numbers of injured CST axons beyond a spinal cord lesion has never been achieved. We found that PTEN/mTOR are critical for controlling the regenerative capacity of mouse corticospinal neurons. After development, the regrowth potential of CST axons was lost and this was accompanied by a downregulation of mTOR activity in corticospinal neurons. Axonal injury further diminished neuronal mTOR activity in these neurons. Forced upregulation of mTOR activity in corticospinal neurons by conditional deletion of Pten, a negative regulator of mTOR, enhanced compensatory sprouting of uninjured CST axons and enabled successful regeneration of a cohort of injured CST axons past a spinal cord lesion. Furthermore, these regenerating CST axons possessed the ability to reform synapses in spinal segments distal to the injury. Thus, modulating neuronal intrinsic PTEN/mTOR activity represents a potential therapeutic strategy for promoting axon regeneration and functional repair after adult spinal cord injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Raineteau, O. & Schwab, M.E. Plasticity of motor systems after incomplete spinal cord injury. Nat. Rev. Neurosci. 2, 263–273 (2001).
Blesch, A. & Tuszynski, M.H. Spinal cord injury: plasticity, regeneration and the challenge of translational drug development. Trends Neurosci. 32, 41–47 (2009).
Zheng, B., Lee, J.K. & Xie, F. Genetic mouse models for studying inhibitors of spinal axon regeneration. Trends Neurosci. 29, 640–646 (2006).
Deumens, R., Koopmans, G.C. & Joosten, E.A. Regeneration of descending axon tracts after spinal cord injury. Prog. Neurobiol. 77, 57–89 (2005).
Thallmair, M., Metz, G.A., Z'Graggen, W.J., Raineteau, O., Kartje, G.L. & Schwab, M.E. Neurite growth inhibitors restrict plasticity and functional recovery following corticospinal tract lesions. Nat. Neurosci. 1, 124–131 (1998).
Cafferty, W.B. & Strittmatter, S.M. The Nogo-Nogo receptor pathway limits a spectrum of adult CNS axonal growth. J. Neurosci. 26, 12242–12250 (2006).
Case, L.C. & Tessier-Lavigne, M. Regeneration of the adult central nervous system. Curr. Biol. 15, R749–R753 (2005).
Schnell, L. & Schwab, M.E. Axonal regeneration in the rat spinal cord produced by an antibody against myelin-associated neurite growth inhibitors. Nature 343, 269–272 (1990).
Savio, T. & Schwab, M.E. Lesioned corticospinal tract axons regenerate in myelin-free rat spinal cord. Proc. Natl. Acad. Sci. USA 87, 4130–4133 (1990).
García-Alías, G., Barkhuysen, S., Buckle, M. & Fawcett, J.W. Chondroitinase ABC treatment opens a window of opportunity for task-specific rehabilitation. Nat. Neurosci. 12, 1145–1151 (2009).
Leaver, S.G. et al. AAV-mediated expression of CNTF promotes long-term survival and regeneration of adult rat retinal ganglion cells. Gene Ther. 13, 1328–1341 (2006).
Schnell, L., Schneider, R., Kolbeck, R., Barde, Y.A. & Schwab, M.E. Neurotrophin-3 enhances sprouting of corticospinal tract during development and after adult spinal cord lesion. Nature 367, 170–173 (1994).
Hiebert, G.W., Khodarahmi, K., McGraw, J., Steeves, J.D. & Tetzlaff, W. Brain-derived neurotrophic factor applied to the motor cortex promotes sprouting of corticospinal fibers, but not regeneration into a peripheral nerve transplant. J. Neurosci. Res. 69, 160–168 (2002).
Hollis, E.R. II, Lu, P., Blesch, A. & Tuszynski, M.H. IGF-I gene delivery promotes corticospinal neuronal survival, but not regeneration, after adult CNS injury. Exp. Neurol. 215, 53–59 (2009).
David, S. & Aguayo, A.J. Axonal elongation into peripheral nervous system “bridges” after central nervous system injury in adult rats. Science 214, 931–933 (1981).
Richardson, P.M., Issa, V.M. & Aguayo, A.J. Regeneration of long spinal axons in the rat. J. Neurocytol. 13, 165–182 (1984).
Bregman, B.S., Kunkel-Bagden, E., McAtee, M. & O'Neill, A. Extension of the critical period for developmental plasticity of the corticospinal pathway. J. Comp. Neurol. 282, 355–370 (1989).
Park, K.K. et al. Promoting axon regeneration in the adult CNS by modulation of the PTEN/mTOR pathway. Science 322, 963–966 (2008).
Bareyre, F.M. et al. The injured spinal cord spontaneously forms a new intraspinal circuit in adult rats. Nat. Neurosci. 7, 269–277 (2004).
Weidner, N., Ner, A., Salimi, N. & Tuszynski, M.H. Spontaneous corticospinal axonal plasticity and functional recovery after adult central nervous system injury. Proc. Natl. Acad. Sci. USA 98, 3513–3518 (2001).
Ma, X.M. & Blenis, J. Molecular mechanisms of mTOR-mediated translational control. Nat. Rev. Mol. Cell Biol. 10, 307–318 (2009).
Groszer, M. et al. Negative regulation of neural stem/progenitor cell proliferation by the Pten tumor suppressor gene in vivo. Science 294, 2186–2189 (2001).
Hasegawa, H. & Wang, F. Visualizing mechanosensory endings of TrkC-expressing neurons in HS3ST-2-hPLAP mice. J. Comp. Neurol. 511, 543–556 (2008).
Bareyre, F.M., Kerschensteiner, M., Misgeld, T. & Sanes, J.R. Transgenic labeling of the corticospinal tract for monitoring axonal responses to spinal cord injury. Nat. Med. 11, 1355–1360 (2005).
Simonen, M. et al. Systemic deletion of the myelin-associated outgrowth inhibitor Nogo-A improves regenerative and plastic responses after spinal cord injury. Neuron 38, 201–211 (2003).
Kim, J.E., Li, S., GrandPre, T., Qiu, D. & Strittmatter, S.M. Axon regeneration in young adult mice lacking Nogo-A/B. Neuron 38, 187–199 (2003).
Zheng, B. et al. Lack of enhanced spinal regeneration in Nogo-deficient mice. Neuron 38, 213–224 (2003).
Steward, O. et al. Regenerative growth of corticospinal tract axons via the ventral column after spinal cord injury in mice. J. Neurosci. 28, 6836–6847 (2008).
Fujiki, M., Zhang, Z., Guth, L. & Steward, O. Genetic influences on cellular reactions to spinal cord injury: activation of macrophages/microglia and astrocytes is delayed in mice carrying a mutation (WldS) that causes delayed Wallerian degeneration. J. Comp. Neurol. 371, 469–484 (1996).
Inman, D.M. & Steward, O. Ascending sensory, but not other long-tract axons, regenerate into the connective tissue matrix that forms at the site of a spinal cord injury in mice. J. Comp. Neurol. 462, 431–449 (2003).
Tom, V.J., Steinmetz, M.P., Miller, J.H., Doller, C.M. & Silver, J. Studies on the development and behavior of the dystrophic growth cone, the hallmark of regeneration failure, in an in vitro model of the glial scar and after spinal cord injury. J. Neurosci. 24, 6531–6539 (2004).
Lee, J.K. et al. Reassessment of corticospinal tract regeneration in Nogo-deficient mice. J. Neurosci. 29, 8649–8654 (2009).
Bundesen, L.Q., Scheel, T.A., Bregman, B.S. & Kromer, L.F. Ephrin-B2 and EphB2 regulation of astrocyte-meningeal fibroblast interactions in response to spinal cord lesions in adult rats. J. Neurosci. 23, 7789–7800 (2003).
Maier, I.C. et al. Constraint-induced movement therapy in the adult rat after unilateral corticospinal tract injury. J. Neurosci. 28, 9386–9403 (2008).
Varoqui, H., Schäfer, M.K., Zhu, H., Weihe, E. & Erickson, J.D. Identification of the differentiation-associated Na+/PI transporter as a novel vesicular glutamate transporter expressed in a distinct set of glutamatergic synapses. J. Neurosci. 22, 142–155 (2002).
Persson, S. et al. Distribution of vesicular glutamate transporters 1 and 2 in the rat spinal cord, with a note on the spinocervical tract. J. Comp. Neurol. 497, 683–701 (2006).
Zhou, F.Q., Zhou, J., Dedhar, S., Wu, Y.H. & Snider, W.D. NGF-induced axon growth is mediated by localized inactivation of GSK-3beta and functions of the microtubule plus end binding protein APC. Neuron 42, 897–912 (2004).
Park, K.K., Liu, K., Hu, Y., Kanter, J.L. & He, Z. PTEN/mTOR and axon regeneration. Exp. Neurol. 223, 45–50 (2010).
Zhao, M. et al. Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-gamma and PTEN. Nature 442, 457–460 (2006).
Yin, H.S. & Selzer, M.E. Axonal regeneration in lamprey spinal cord. J. Neurosci. 3, 1135–1144 (1983).
Bernstein, J.J. & Gelderd, J.B. Synaptic reorganization following regeneration of goldfish spinal cord. Exp. Neurol. 41, 402–410 (1973).
García-Alías, G., Barkhuysen, S., Buckle, M. & Fawcett, J.W. Chondroitinase ABC treatment opens a window of opportunity for task-specific rehabilitation. Nat. Neurosci. 12, 1145–1151 (2009).
Houle, J.D. et al. Combining an autologous peripheral nervous system “bridge” and matrix modification by chondroitinase allows robust, functional regeneration beyond a hemisection lesion of the adult rat spinal cord. J. Neurosci. 26, 7405–7415 (2006).
Alto, L.T. et al. Chemotropic guidance facilitates axonal regeneration and synapse formation after spinal cord injury. Nat. Neurosci. 12, 1106–1113 (2009).
Pearse, D.D. et al. cAMP and Schwann cells promote axonal growth and functional recovery after spinal cord injury. Nat. Med. 10, 610–616 (2004).
Kennard, M.A. age and other factors in motor recovery from precentral lesions in monkeys. Am. J. Physiol. 115, 138–146 (1936).
Berger, W. Characteristics of locomotor control in children with cerebral palsy. Neurosci. Biobehav. Rev. 22, 579–582 (1998).
Courtine, G. et al. Recovery of supraspinal control of stepping via indirect propriospinal relay connections after spinal cord injury. Nat. Med. 14, 69–74 (2008).
Arlotta, P. et al. Neuronal subtype-specific genes that control corticospinal motor neuron development in vivo. Neuron 45, 207–221 (2005).
Acknowledgements
We thank F. Wang for providing Rosa-stop-PLAP mice and M. Greenberg, M. Tessier-Lavigne and C. Woolf for reading the manuscript. This work was supported by grants from Wings for Life (K.L. and Z.H.), the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation (Z.H.), the Craig H. Neilson Foundation (Y.L. and K.K.P.), the National Institute of Neurological Disorders and Stroke (Z.H., B.Z. and O.S.), the International Spinal Research Trust (Z.H.) and a private contribution to the Reeve-Irvine Research Center (O.S.). R.W. is the recipient of a predoctoral fellowship from the National Institute of Neurological Disorders and Stroke.
Author information
Authors and Affiliations
Contributions
K.L., Y.L., J.K.L., R.S., R.W., I.S.-K., A.T., K.K.P., D.J., B.C., B.X., L.C. and O.S. performed the experiments and analyzed the data. K.L., J.K.L., O.S., B.Z. and Z.H. drafted and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
O.S. and Z.H. are co-founders of Axonis.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13 (PDF 7816 kb)
Rights and permissions
About this article
Cite this article
Liu, K., Lu, Y., Lee, J. et al. PTEN deletion enhances the regenerative ability of adult corticospinal neurons. Nat Neurosci 13, 1075–1081 (2010). https://doi.org/10.1038/nn.2603
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2603
This article is cited by
-
The role of TSC1 and TSC2 proteins in neuronal axons
Molecular Psychiatry (2024)
-
M2 Microglia-derived Exosomes Promote Spinal Cord Injury Recovery in Mice by Alleviating A1 Astrocyte Activation
Molecular Neurobiology (2024)
-
Low-Dose LPS Modulates Microglia/Macrophages Phenotypic Transformation to Amplify Rehabilitation Effects in Chronic Spinal Cord Injured (CSCI) Mice
Molecular Neurobiology (2024)
-
Unleashing Axonal Regeneration Capacities: Neuronal and Non-neuronal Changes After Injuries to Dorsal Root Ganglion Neuron Central and Peripheral Axonal Branches
Molecular Neurobiology (2024)
-
TRPM7 Mediates BSCB Disruption After Spinal Cord Injury by Regulating the mTOR/JMJD3 Axis in Rats
Molecular Neurobiology (2024)