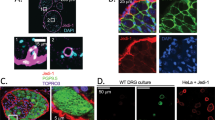
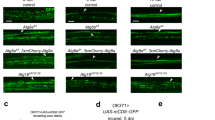
Abstract
During the development of peripheral ganglia, 50% of the neurons that are generated undergo apoptosis. How the massive numbers of corpses are removed is unknown. We found that satellite glial cell precursors are the primary phagocytic cells for apoptotic corpse removal in developing mouse dorsal root ganglia (DRG). Confocal and electron microscopic analysis revealed that glial precursors, rather than macrophages, were responsible for clearing most of the dead DRG neurons. Moreover, we identified Jedi-1, an engulfment receptor, and MEGF10, a purported engulfment receptor, as homologs of the invertebrate engulfment receptors Draper and CED-1 expressed in the glial precursor cells. Expression of Jedi-1 or MEGF10 in fibroblasts facilitated binding to dead neurons, and knocking down either protein in glial cells or overexpressing truncated forms lacking the intracellular domain inhibited engulfment of apoptotic neurons. Together, these results suggest a cellular and molecular mechanism by which neuronal corpses are culled during DRG development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Bennet, M.R., Gibson, W.G. & Lemon, G. Neuronal cell death, nerve growth factor and neurotrophic models: 50 years on. Auton. Neurosci. 95, 1–23 (2002).
Hamburger, V. & Levi-Montalcini, R. Proliferation, differentiation and degeneration in the spinal ganglia of the chick embryo under normal and experimental conditions. J. Exp. Zool. 111, 457–501 (1949).
Savill, J., Dransfield, I., Gregory, C. & Haslett, C. A blast from the past: clearance of apoptotic cells regulates immune responses. Nat. Rev. Immunol. 2, 965–975 (2002).
Yuan, J., Lipinski, M. & Degterev, A. Diversity in the mechanisms of neuronal cell death. Neuron 40, 401–413 (2003).
Hume, D.A., Perry, V.H. & Gordon, S. Immunohistochemical localization of a macrophage-specific antigen in developing mouse retina: phagocytosis of dying neurons and differentiation of microglial cells to form a regular array in the plexiform layers. J. Cell Biol. 97, 253–257 (1983).
Perry, V.H., Hume, D.A. & Gordon, S. Immunohistochemical localization of macrophages and microglia in the adult and developing mouse brain. Neuroscience 15, 313–326 (1985).
O'Connor, T.M. & Wyttenbach, C.R. Cell death in the embryonic chick spinal cord. J. Cell Biol. 60, 448–459 (1974).
Pannese, E. The response of the satellite and other non-neuronal cells to the degeneration of neuroblasts in chick embryo spinal ganglia. Cell Tissue Res. 190, 1–14 (1978).
Bratton, D.L. & Henson, P.M. Apoptotic cell recognition: will the real phosphatidylserine receptor(s) please stand up? Curr. Biol. 18, R76–R79 (2008).
Gregory, C.D. & Brown, S.B. Apoptosis: eating sensibly. Nat. Cell Biol. 7, 1161–1163 (2005).10.1038/ncb1205-1161
Grimsley, C. & Ravichandran, K.S. Cues for apoptotic cell engulfment: eat-me, don't eat-me and come-get-me signals. Trends Cell Biol. 13, 648–656 (2003).
Henson, P.M. & Hume, D.A. Apoptotic cell removal in development and tissue homeostasis. Trends Immunol. 27, 244–250 (2006).
Freeman, M.R., Delrow, J., Kim, J., Johnson, E. & Doe, C.Q. Unwrapping glial biology: Gcm target genes regulating glial development, diversification, and function. Neuron 38, 567–580 (2003).
Zhou, Z., Hartwieg, E. & Horvitz, H.R. CED-1 is a transmembrane receptor that mediates cell corpse engulfment in C. elegans. Cell 104, 43–56 (2001).
Awasaki, T. et al. Essential role of the apoptotic cell engulfment genes draper and ced-6 in programmed axon pruning during Drosophila metamorphosis. Neuron 50, 855–867 (2006).
MacDonald, J.M. et al. The Drosophila cell corpse engulfment receptor Draper mediates glial clearance of severed axons. Neuron 50, 869–881 (2006).
Manaka, J. et al. Draper-mediated and phosphatidylserine-independent phagocytosis of apoptotic cells by Drosophila hemocytes/macrophages. J. Biol. Chem. 279, 48466–48476 (2004).
Savill, J. & Fadok, V. Corpse clearance defines the meaning of cell death. Nature 407, 784–788 (2000).
Hamon, Y. et al. Cooperation between engulfment receptors: the case of ABCA1 and MEGF10. PLoS One 1, e120 (2006).
Fariñas, I., Yoshida, C.K., Backus, C. & Reichardt, L.F. Lack of neurotrophin-3 results in death of spinal sensory neurons and premature differentiation of their precursors. Neuron 17, 1065–1078 (1996).
White, F.A. et al. Synchronous onset of NGF and TrkA survival dependence in developing dorsal root ganglia. J. Neurosci. 16, 4662–4672 (1996).
Kurtz, A. et al. The expression pattern of a novel gene encoding brain-fatty acid binding protein correlates with neuronal and glial cell development. Development 120, 2637–2649 (1994).
Schreiner, S. et al. Hypomorphic Sox10 alleles reveal novel protein functions and unravel developmental differences in glial lineages. Development 134, 3271–3281 (2007).
Taylor, M.K., Yeager, K. & Morrison, S.J. Physiological Notch signaling promotes gliogenesis in the developing peripheral and central nervous systems. Development 134, 2435–2447 (2007).
Woodhoo, A., Dean, C.H., Droggiti, A., Mirsky, R. & Jessen, K.R. The trunk neural crest and its early glial derivatives: a study of survival responses, developmental schedules and autocrine mechanisms. Mol. Cell. Neurosci. 25, 30–41 (2004).
Britsch, S. et al. The transcription factor Sox10 is a key regulator of peripheral glial development. Genes Dev. 15, 66–78 (2001).
Fariñas, I., Cano-Jaimez, M., Bellmunt, E. & Soriano, M. Regulation of neurogenesis by neurotrophins in developing spinal sensory ganglia. Brain Res. Bull. 57, 809–816 (2002).
Maro, G.S. et al. Neural crest boundary cap cells constitute a source of neuronal and glial cells of the PNS. Nat. Neurosci. 7, 930–938 (2004).
Ernfors, P., Lee, K.F., Kucera, J. & Jaenisch, R. Lack of neurotrophin-3 leads to deficiencies in the peripheral nervous system and loss of limb proprioceptive afferents. Cell 77, 503–512 (1994).
Tessarollo, L., Vogel, K.S., Palko, M.E., Reid, S.W. & Parada, L.F. Targeted mutation in the neurotrophin-3 gene results in loss of muscle sensory neurons. Proc. Natl. Acad. Sci. USA 91, 11844–11848 (1994).
Murphy, P. et al. The regulation of Krox-20 expression reveals important steps in the control of peripheral glial cell development. Development 122, 2847–2857 (1996).
Okada, A., Lansford, R., Weimann, J.M., Fraser, S.E. & McConnell, S.K. Imaging cells in the developing nervous system with retrovirus expressing modified green fluorescent protein. Exp. Neurol. 156, 394–406 (1999).
Hoopfer, E.D. et al. Wlds protection distinguishes axon degeneration following injury from naturally occurring developmental pruning. Neuron 50, 883–895 (2006).
Suzuki, E. & Nakayama, M. MEGF10 is a mammalian ortholog of CED-1 that interacts with clathrin assembly protein complex 2 medium chain and induces large vacuole formation. Exp. Cell Res. 313, 3729–3742 (2007).
Griffin, J.W., George, R. & Ho, T. Macrophage systems in peripheral nerves. A review. J. Neuropathol. Exp. Neurol. 52, 553–560 (1993).
Hirata, K. & Kawabuchi, M. Myelin phagocytosis by macrophages and nonmacrophages during Wallerian degeneration. Microsc. Res. Tech. 57, 541–547 (2002).
Bishop, D.L., Misgeld, T., Walsh, M.K., Gan, W.B. & Lichtman, J.W. Axon branch removal at developing synapses by axosome shedding. Neuron 44, 651–661 (2004).
Aldskogius, H. & Arvidsson, J. Nerve cell degeneration and death in the trigeminal ganglion of the adult rat following peripheral nerve transection. J. Neurocytol. 7, 229–250 (1978).
Hanani, M. Satellite glial cells in sensory ganglia: from form to function. Brain Res. Brain Res. Rev. 48, 457–476 (2005).
Fenzi, F., Benedetti, M.D., Moretto, G. & Rizzuto, N. Glial cell and macrophage reactions in rat spinal ganglion after peripheral nerve lesions: an immunocytochemical and morphometric study. Arch. Ital. Biol. 139, 357–365 (2001).
Yu, X., Lu, N. & Zhou, Z. Phagocytic receptor CED-1 initiates a signaling pathway for degrading engulfed apoptotic cells. PLoS Biol. 6, e61 (2008).
Ravichandran, K.S. & Lorenz, U. Engulfment of apoptotic cells: signals for a good meal. Nat. Rev. Immunol. 7, 964–974 (2007).
Reddien, P.W., Cameron, S. & Horvitz, H.R. Phagocytosis promotes programmed cell death in C. elegans. Nature 412, 198–202 (2001).
Kurant, E., Axelrod, S., Leaman, D. & Gaul, U. Six-microns-under acts upstream of Draper in the glial phagocytosis of apoptotic neurons. Cell 133, 498–509 (2008).
Ziegenfuss, J.S. et al. Draper-dependent glial phagocytic activity is mediated by Src and Syk family kinase signaling. Nature 453, 935–939 (2008).
Nagata, S. Autoimmune diseases caused by defects in clearing dead cells and nuclei expelled from erythroid precursors. Immunol. Rev. 220, 237–250 (2007).
Silva, M.T., do Vale, A. & Dos Santos, N.M. Secondary necrosis in multicellular animals: an outcome of apoptosis with pathogenic implications. Apoptosis 13, 463–482 (2008).
Wu, H.H. et al. Autoregulation of neurogenesis by GDF11. Neuron 37, 197–207 (2003).
Jin, Y., Jorgensen, E., Hartwieg, E. & Horvitz, H.R. The Caenorhabditis elegans gene unc-25 encodes glutamic acid decarboxylase and is required for synaptic transmission, but not synaptic development. J. Neurosci. 19, 539–548 (1999).
Acknowledgements
The authors thank the Statistics and Methodology Services at the Vanderbilt Kennedy Center for assistance on statistical analysis and C. Yoon, C. Jones and other members of the Carter laboratory for technical assistance and helpful suggestions. This work was supported by grants from the US National Institutes of Health (NS048249 and NS064278 to B.D.C., GM067848 to Z.Z.), a Muscular Dystrophy Association Development grant (MDA4023) to H.-H.W., a US National Institutes of Health Minority Access to Research Careers Predoctoral Fellowship (GM079911) to V.V., and the Ministerio de Ciencia e Innovación (SAF), Ministerio de Sanidad (TerCel and Ciberned), Fundación la Caixa, and Generalitat Valenciana (Prometeo) to I.F.
Author information
Authors and Affiliations
Contributions
H.-H.W. and B.D.C. initiated and developed the overall concept and design of the project. H.-H.W. also performed, analyzed and interpreted most of the experiments and prepared the initial version of the manuscript. E.B. performed the quantitative histological analysis of neuronal corpse engulfment in Ntf3+/+ and Ntf3−/− mice. J.L.S. generated some of the Jedi-1 and MEGF10 constructs, performed the binding experiment and some of the immunostaining analyses. V.V. performed all of the experiments with C. elegans. C.B. assisted with the immunostaining on sections and generated the shRNA construct for MEGF10. L.F.R. provided technical expertise for electron microscopy analysis and critical intellectual input for this study. I.F. performed the electron microscopy analysis, supervised the quantitative histological analysis and provided intellectual input. Z.Z. designed and supervised the C. elegans study and provided intellectual input. B.D.C. directed the overall project and prepared the final version of the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 1836 kb)
Rights and permissions
About this article
Cite this article
Wu, HH., Bellmunt, E., Scheib, J. et al. Glial precursors clear sensory neuron corpses during development via Jedi-1, an engulfment receptor. Nat Neurosci 12, 1534–1541 (2009). https://doi.org/10.1038/nn.2446
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2446
This article is cited by
-
Glial Draper signaling triggers cross-neuron plasticity in bystander neurons after neuronal cell death in Drosophila
Nature Communications (2023)
-
SVEP1 is an endogenous ligand for the orphan receptor PEAR1
Nature Communications (2023)
-
Astrocytic phagocytosis contributes to demyelination after focal cortical ischemia in mice
Nature Communications (2022)
-
AXL kinase-mediated astrocytic phagocytosis modulates outcomes of traumatic brain injury
Journal of Neuroinflammation (2021)
-
The sympathetic nervous system in development and disease
Nature Reviews Neuroscience (2021)