Abstract
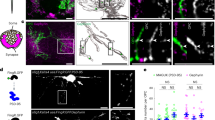
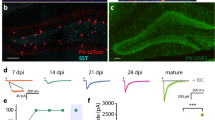
Adult neurogenesis occurs in the hippocampus and the olfactory bulb of the mammalian CNS. Recent studies have demonstrated that newborn granule cells of the adult hippocampus are postsynaptic targets of excitatory and inhibitory neurons, but evidence of synapse formation by the axons of these cells is still lacking. By combining retroviral expression of green fluorescent protein in adult-born neurons of the mouse dentate gyrus with immuno-electron microscopy, we found output synapses that were formed by labeled terminals on appropriate target cells in the CA3 area and the hilus. Furthermore, retroviral expression of channelrhodopsin-2 allowed us to light-stimulate newborn granule cells and identify postsynaptic target neurons by whole-cell recordings in acute slices. Our structural and functional evidence indicates that axons of adult-born granule cells establish synapses with hilar interneurons, mossy cells and CA3 pyramidal cells and release glutamate as their main neurotransmitter.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
20 July 2008
In the version of this article initially published online, the reference to earlier figures in the legend of Figure 5 is incorrect. For the legend of Figure 5c, the correct sentence should read "...(acquisition was interrupted from 20-25 min; same cell as in Fig. 4a-e)". The error has been corrected for all versions of this article.
References
Schinder, A.F. & Gage, F.H. A hypothesis about the role of adult neurogenesis in hippocampal function. Physiology (Bethesda) 19, 253–261 (2004).
Ming, G.L. & Song, H. Adult neurogenesis in the mammalian central nervous system. Annu. Rev. Neurosci. 28, 223–250 (2005).
Aimone, J.B., Wiles, J. & Gage, F.H. Potential role for adult neurogenesis in the encoding of time in new memories. Nat. Neurosci. 9, 723–727 (2006).
Lledo, P.M., Alonso, M. & Grubb, M.S. Adult neurogenesis and functional plasticity in neuronal circuits. Nat. Rev. Neurosci. 7, 179–193 (2006).
Zhao, C., Deng, W. & Gage, F.H. Mechanisms and functional implications of adult neurogenesis. Cell 132, 645–660 (2008).
van Praag, H. et al. Functional neurogenesis in the adult hippocampus. Nature 415, 1030–1034 (2002).
Schmidt-Hieber, C., Jonas, P. & Bischofberger, J. Enhanced synaptic plasticity in newly generated granule cells of the adult hippocampus. Nature 429, 184–187 (2004).
Espósito, M.S. et al. Neuronal differentiation in the adult hippocampus recapitulates embryonic development. J. Neurosci. 25, 10074–10086 (2005).
Ge, S. et al. GABA regulates synaptic integration of newly generated neurons in the adult brain. Nature 439, 589–593 (2006).
Laplagne, D.A. et al. Functional convergence of neurons generated in the developing and adult hippocampus. PLoS Biol. 4, e409 (2006).
Ge, S., Yang, C.H., Hsu, K.S., Ming, G.L. & Song, H. A critical period for enhanced synaptic plasticity in newly generated neurons of the adult brain. Neuron 54, 559–566 (2007).
Toni, N. et al. Synapse formation on neurons born in the adult hippocampus. Nat. Neurosci. 10, 727–734 (2007).
Shapiro, L.A., Upadhyaya, P. & Ribak, C.E. Spatiotemporal profile of dendritic outgrowth from newly born granule cells in the adult rat dentate gyrus. Brain Res. 1149, 30–37 (2007).
Henze, D.A., Urban, N.N. & Barrionuevo, G. The multifarious hippocampal mossy fiber pathway: a review. Neuroscience 98, 407–427 (2000).
Zhao, C., Teng, E.M., Summers, R.G., Jr, Ming, G.L. & Gage, F.H. Distinct morphological stages of dentate granule neuron maturation in the adult mouse hippocampus. J. Neurosci. 26, 3–11 (2006).
Amaral, D.G. Synaptic extensions from the mossy fibers of the fascia dentata. Anat. Embryol. (Berl.) 155, 241–251 (1979).
Frotscher, M. Mossy fiber synapses on glutamate decarboxylase–immunoreactive neurons: evidence for feed-forward inhibition in the CA3 region of the hippocampus. Exp. Brain Res. 75, 441–445 (1989).
Soriano, E. & Frotscher, M. Spiny nonpyramidal neurons in the CA3 region of the rat hippocampus are glutamate-like immunoreactive and receive convergent mossy fiber input. J. Comp. Neurol. 333, 435–448 (1993).
Chicurel, M.E. & Harris, K.M. Three-dimensional analysis of the structure and composition of CA3 branched dendritic spines and their synaptic relationships with mossy fiber boutons in the rat hippocampus. J. Comp. Neurol. 325, 169–182 (1992).
Amaral, D.G., Ishizuka, N. & Claiborne, B. Neurons, numbers and the hippocampal network. Prog. Brain Res. 83, 1–11 (1990).
Acsady, L., Kamondi, A., Sik, A., Freund, T. & Buzsaki, G. GABAergic cells are the major postsynaptic targets of mossy fibers in the rat hippocampus. J. Neurosci. 18, 3386–3403 (1998).
Boyden, E.S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268 (2005).
Li, X. et al. Fast noninvasive activation and inhibition of neural and network activity by vertebrate rhodopsin and green algae channelrhodopsin. Proc. Natl. Acad. Sci. USA 102, 17816–17821 (2005).
Walker, M.C., Ruiz, A. & Kullmann, D.M. Monosynaptic GABAergic signaling from dentate to CA3 with a pharmacological and physiological profile typical of mossy fiber synapses. Neuron 29, 703–715 (2001).
Gutierrez, R. The dual glutamatergic-GABAergic phenotype of hippocampal granule cells. Trends Neurosci. 28, 297–303 (2005).
Scharfman, H.E., Kunkel, D.D. & Schwartzkroin, P.A. Synaptic connections of dentate granule cells and hilar neurons: results of paired intracellular recordings and intracellular horseradish peroxidase injections. Neuroscience 37, 693–707 (1990).
Scharfman, H.E. Evidence from simultaneous intracellular recordings in rat hippocampal slices that area CA3 pyramidal cells innervate dentate hilar mossy cells. J. Neurophysiol. 72, 2167–2180 (1994).
Scharfman, H.E. Electrophysiological evidence that dentate hilar mossy cells are excitatory and innervate both granule cells and interneurons. J. Neurophysiol. 74, 179–194 (1995).
Kamiya, H., Shinozaki, H. & Yamamoto, C. Activation of metabotropic glutamate receptor type 2/3 suppresses transmission at rat hippocampal mossy fiber synapses. J. Physiol. (Lond.) 493, 447–455 (1996).
Amaral, D.G. & Dent, J.A. Development of the mossy fibers of the dentate gyrus. I. A light and electron microscopic study of the mossy fibers and their expansions. J. Comp. Neurol. 195, 51–86 (1981).
Seress, L. & Ribak, C.E. Postnatal development of CA3 pyramidal neurons and their afferents in the Ammon's horn of rhesus monkeys. Hippocampus 5, 217–231 (1995).
Laplagne, D.A. et al. Similar GABAergic inputs in dentate granule cells born during embryonic and adult neurogenesis. Eur. J. Neurosci. 25, 2973–2981 (2007).
Snyder, J.S., Kee, N. & Wojtowicz, J.M. Effects of adult neurogenesis on synaptic plasticity in the rat dentate gyrus. J. Neurophysiol. 85, 2423–2431 (2001).
Campbell, R.E. et al. A monomeric red fluorescent protein. Proc. Natl. Acad. Sci. USA 99, 7877–7882 (2002).
Standen, N.B. & Stanfield, P.R. A potential- and time-dependent blockade of inward rectification in frog skeletal muscle fibres by barium and strontium ions. J. Physiol. (Lond.) 280, 169–191 (1978).
Jonas, P., Major, G. & Sakmann, B. Quantal components of unitary EPSCs at the mossy fiber synapse on CA3 pyramidal cells of rat hippocampus. J. Physiol. (Lond.) 472, 615–663 (1993).
Kneisler, T.B. & Dingledine, R. Spontaneous and synaptic input from granule cells and the perforant path to dentate basket cells in the rat hippocampus. Hippocampus 5, 151–164 (1995).
Acknowledgements
We would like to thank J. Jepsen and N. Nashi for technical assistance, S. Herlitze for providing the ChR2 construct, L. Petreanu for helpful discussions, J. Simon for artwork, J. Fiala for providing software and M.L. Gage for helpful suggestions to improve this manuscript. We also acknowledge the support of the Picower Foundation, Lookout Foundation, McDonnell Foundation, US National Institutes of Health grants NS050217 (to F.H.G.) and NS038331 (to C.E.R.), Agencia Nacional de Promoción Científica, Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), and the Howard Hughes Medical Institute (to A.F.S.). D.A.L. and G.L. were supported by fellowships from CONICET.
Author information
Authors and Affiliations
Contributions
N.T. contributed to the concept, designed and carried out the structural experiments, analyzed the data, and wrote the manuscript. D.A.L. contributed to the concept, designed and performed the functional experiments, analyzed the data, and wrote the manuscript. C.Z. contributed to the experimental design, provided samples for the structural experiments, carried out and analyzed confocal images of presynaptic terminals, and revised the manuscript. G.L. prepared retroviral stocks, performed immunofluorescence and obtained images of ChR2-positive neurons. C.E.R. contributed to setting up the technique for immuno-electron microscopy of GFP, the analysis of electron micrographs, and the writing and revision of the manuscript. F.H.G. and A.F.S. contributed to the concept, designed the experiments, analyzed the data, wrote the manuscript and provided financial support.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Table 1 and Supplementary Methods (PDF 1896 kb)
Rights and permissions
About this article
Cite this article
Toni, N., Laplagne, D., Zhao, C. et al. Neurons born in the adult dentate gyrus form functional synapses with target cells. Nat Neurosci 11, 901–907 (2008). https://doi.org/10.1038/nn.2156
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2156
This article is cited by
-
Dorsal hippocampus to nucleus accumbens projections drive reinforcement via activation of accumbal dynorphin neurons
Nature Communications (2024)
-
Overexpress miR-132 in the Brain Parenchyma by a Non-invasive Way Improves Tissue Repairment and Releases Memory Impairment After Traumatic Brain Injury
Cellular and Molecular Neurobiology (2024)
-
Enhanced excitability but mature action potential waveforms at mossy fiber terminals of young, adult-born hippocampal neurons in mice
Communications Biology (2023)
-
Adult-born neurons in critical period maintain hippocampal seizures via local aberrant excitatory circuits
Signal Transduction and Targeted Therapy (2023)
-
Engram neurons: Encoding, consolidation, retrieval, and forgetting of memory
Molecular Psychiatry (2023)