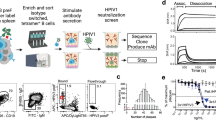
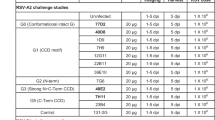
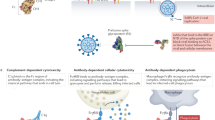
Abstract
Respiratory syncytial virus (RSV) and human metapneumovirus (HMPV) are two closely related viruses that cause bronchiolitis and pneumonia in infants and the elderly1, with a significant health burden2–6. There are no licensed vaccines or small-molecule antiviral treatments specific to these two viruses at present. A humanized murine monoclonal antibody (palivizumab) is approved to treat high-risk infants for RSV infection7,8, but other treatments, as well as vaccines, for both viruses are still in development. Recent epidemiological modelling suggests that cross-immunity between RSV, HMPV and human parainfluenzaviruses may contribute to their periodic outbreaks9, suggesting that a deeper understanding of host immunity to these viruses may lead to enhanced strategies for their control. Cross-reactive neutralizing antibodies to the RSV and HMPV fusion (F) proteins have been identified10,11. Here, we examine the structural basis for cross-reactive antibody binding to RSV and HMPV F protein by two related, independently isolated antibodies, MPE8 and 25P13. We solved the structure of the MPE8 antibody bound to RSV F protein and identified the 25P13 antibody from an independent blood donor. Our results indicate that both antibodies use germline residues to interact with a conserved surface on F protein that could guide the emergence of cross-reactivity. The induction of similar cross-reactive neutralizing antibodies using structural vaccinology approaches could enhance intrinsic cross-immunity to these paramyxoviruses and approaches to controlling recurring outbreaks.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Collins, P. L. & Karron, R. A. in Fields Virology 6th edn, Vol. 1 (eds Knipe, D. M. & Howley, P. M. ) Ch. 38 (Wolters Kluwer Health Adis (ESP), 2013).
Hall, C. B., Simoes, E. A. & Anderson, L. J. Clinical and epidemiologic features of respiratory syncytial virus. Curr. Top. Microbiol. Immunol. 372, 39–57 (2013).
Falsey, A. R., Hennessey, P. A., Formica, M. A., Cox, C. & Walsh, E. E. Respiratory syncytial virus infection in elderly and high-risk adults. N. Engl. J. Med. 352, 1749–1759 (2005).
Hall, C. B. The burgeoning burden of respiratory syncytial virus among children. Infect. Disord. Drug Targets 12, 92–97 (2012).
Edwards, K. M. et al. Burden of human metapneumovirus infection in young children. N. Engl. J. Med. 368, 633–643 (2013).
Williams, J. V. et al. Human metapneumovirus and lower respiratory tract disease in otherwise healthy infants and children. N. Engl. J. Med. 350, 443–450 (2004).
Fenton, C., Scott, L. J. & Plosker, G. L. Palivizumab: a review of its use as prophylaxis for serious respiratory syncytial virus infection. Paediatr. Drugs 6, 177–197 (2004).
The IMpact-RSV Study Group. Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from respiratory syncytial virus infection in high-risk infants. Pediatrics 102, 531–537 (1998).
Bhattacharyya, S., Gesteland, P. H., Korgenski, K., Bjornstad, O. N. & Adler, F. R. Cross-immunity between strains explains the dynamical pattern of paramyxoviruses. Proc. Natl Acad. Sci. USA 112, 13396–13400 (2015).
Corti, D. et al. Cross-neutralization of four paramyxoviruses by a human monoclonal antibody. Nature 501, 439–443 (2013).
Schuster, J. E. et al. A broadly neutralizing human monoclonal antibody exhibits in vivo efficacy against both human metapneumovirus and respiratory syncytial virus. J. Infect. Dis. 211, 216–225 (2015).
Jardetzky, T. S. & Lamb, R. A. Activation of paramyxovirus membrane fusion and virus entry. Curr. Opin. Virol. 5, 24–33 (2014).
Wen, X. et al. Structure of the human metapneumovirus fusion protein with neutralizing antibody identifies a pneumovirus antigenic site. Nat. Struct. Mol. Biol. 19, 461–463 (2012).
McLellan, J. S. et al. Structure of RSV fusion glycoprotein trimer bound to a prefusion-specific neutralizing antibody. Science 340, 1113–1117 (2013).
McLellan, J. S., Yang, Y., Graham, B. S. & Kwong, P. D. Structure of the respiratory syncytial virus fusion glycoprotein in the post-fusion conformation reveals preservation of neutralizing epitopes. J. Virol. 85, 7788–7796 (2011).
Swanson, K. A. et al. Structural basis for immunization with postfusion respiratory syncytial virus fusion F glycoprotein (RSV F) to elicit high neutralizing antibody titers. Proc. Natl Acad. Sci. USA 108, 9619–9624 (2011).
Yin, H. S., Wen, X., Paterson, R. G., Lamb, R. A. & Jardetzky, T. S. Structure of the parainfluenza virus 5 F protein in its metastable, prefusion conformation. Nature 439, 38–44 (2006).
Mas, V. et al. Engineering, structure and immunogenicity of the human metapneumovirus F protein in the postfusion conformation. PLoS Pathogens 12, e1005859 (2016).
Joyce, M. G. et al. Iterative structure-based improvement of a fusion-glycoprotein vaccine against RSV. Nat. Struct. Mol. Biol. 23, 811–820 (2016).
Boyington, J. C. et al. Structure-based design of head-only fusion glycoprotein immunogens for respiratory syncytial virus. PLoS ONE 11, e0159709 (2016).
Stewart-Jones, G. B. et al. A cysteine zipper stabilizes a pre-fusion F glycoprotein vaccine for respiratory syncytial virus. PLoS ONE 10, e0128779 (2015).
McLellan, J. S. et al. Structure-based design of a fusion glycoprotein vaccine for respiratory syncytial virus. Science 342, 592–598 (2013).
Wen, X. et al. A chimeric pneumovirus fusion protein carrying neutralizing epitopes of both MPV and RSV. PLoS ONE 11, e0155917 (2016).
Gilman, M. S. et al. Characterization of a prefusion-specific antibody that recognizes a quaternary, cleavage-dependent epitope on the RSV fusion glycoprotein. PLoS Pathog. 11, e1005035 (2015).
Lee, P. S. & Wilson, I. A. Structural characterization of viral epitopes recognized by broadly cross-reactive antibodies. Curr. Top. Microbiol. Immunol. 386, 323–341 (2015).
Doria-Rose, N. A. & Joyce, M. G. Strategies to guide the antibody affinity maturation process. Curr. Opin. Virol. 11, 137–147 (2015).
Schmidt, A. G. et al. Viral receptor-binding site antibodies with diverse germline origins. Cell 161, 1026–1034 (2015).
Zhou, T. et al. Structural repertoire of HIV-1-neutralizing antibodies targeting the CD4 supersite in 14 donors. Cell 161, 1280–1292 (2015).
Storoni, L. C., McCoy, A. J. & Read, R. J. Likelihood-enhanced fast rotation functions. Acta Crystallogr. D 60, 432–438 (2004).
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Zwart, P. H. et al. Automated structure solution with the PHENIX suite. Methods Mol. Biol. 426, 419–435 (2008).
Adams, P. D. et al. PHENIX: building new software for automated crystallographic structure determination. Acta Crystallogr. D 58, 1948–1954 (2002).
Emsley, P. & Cowtan, K. COOT: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Smith, S. A. et al. Persistence of circulating memory B cell clones with potential for dengue virus disease enhancement for decades following infection. J. Virol. 86, 2665–2675 (2012).
Acknowledgements
The authors acknowledge support from the members of the Jardetzky, Lamb and Crowe laboratories. This research was supported in part by National Institutes of Health research grants AI-23173 (to R.A.L.) and GM-61050 (to T.S.J.). R.A.L. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
X.W., J.J.M., J.T.B., R.A.L., J.E.C. and T.S.J. conceived and designed the experiments. X.W., J.J.M. and J.T.B. performed the experiments. X.W., J.J.M., J.T.B., R.A.L., J.E.C. and T.S.J. analysed the data. X.W., J.J.M. and J.T.B. contributed reagents, materials and analysis tools. X.W., J.J.M., J.T.B., R.A.L., J.E.C. and T.S.J. wrote and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
One of the authors (J.E.C.) declares a competing interest. J.E.C. is a consultant to Compuvax and a member of the Scientific Advisory Board of Meissa Vaccines. The remaining authors declare no competing interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–7; Supplementary Tables 1 and 2; Supplementary References. (PDF 2668 kb)
Rights and permissions
About this article
Cite this article
Wen, X., Mousa, J., Bates, J. et al. Structural basis for antibody cross-neutralization of respiratory syncytial virus and human metapneumovirus. Nat Microbiol 2, 16272 (2017). https://doi.org/10.1038/nmicrobiol.2016.272
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.272
This article is cited by
-
Cross-protective antibodies against common endemic respiratory viruses
Nature Communications (2023)
-
A human antibody potently neutralizes RSV by targeting the conserved hydrophobic region of prefusion F
Science China Life Sciences (2023)
-
Aerosol delivery of SARS-CoV-2 human monoclonal antibodies in macaques limits viral replication and lung pathology
Nature Communications (2023)
-
Structure-based design of prefusion-stabilized human metapneumovirus fusion proteins
Nature Communications (2022)
-
Profiling of hMPV F-specific antibodies isolated from human memory B cells
Nature Communications (2022)