Abstract
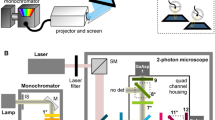
Drosophila melanogaster is a model organism rich in genetic tools to manipulate and identify neural circuits involved in specific behaviors. Here we present a technique for two-photon calcium imaging in the central brain of head-fixed Drosophila walking on an air-supported ball. The ball's motion is tracked at high resolution and can be treated as a proxy for the fly's own movements. We used the genetically encoded calcium sensor, GCaMP3.0, to record from important elements of the motion-processing pathway, the horizontal-system lobula plate tangential cells (LPTCs) in the fly optic lobe. We presented motion stimuli to the tethered fly and found that calcium transients in horizontal-system neurons correlated with robust optomotor behavior during walking. Our technique allows both behavior and physiology in identified neurons to be monitored in a genetic model organism with an extensive repertoire of walking behaviors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
10 January 2011
In the version of this article initially published, the units for angular position (degrees) in Figure 3a,b are incorrect. The correct unit should be mm. The error has been corrected in the HTML and PDF versions of the article.
References
Vosshall, L.B. Into the mind of a fly. Nature 450, 193–197 (2007).
Olsen, S.R. & Wilson, R.I. Cracking neural circuits in a tiny brain: new approaches for understanding the neural circuitry of Drosophila. Trends Neurosci. 31, 512–520 (2008).
Borst, A. Drosophila's view on insect vision. Curr. Biol. 19, R36–R47 (2009).
Simpson, J.H. Mapping and manipulating neural circuits in the fly brain. Adv. Genet. 65, 79–143 (2009).
Gouwens, N.W. & Wilson, R.I. Signal propagation in Drosophila central neurons. J. Neurosci. 29, 6239–6249 (2009).
Peron, S.P., Krapp, H.G. & Gabbiani, F. Influence of electrotonic structure and synaptic mapping on the receptive field properties of a collision-detecting neuron. J. Neurophysiol. 97, 159–177 (2007).
Haag, J. & Borst, A. Neural mechanism underlying complex receptive field properties of motion-sensitive interneurons. Nat. Neurosci. 7, 628–634 (2004).
Denk, W., Strickler, J.H. & Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990).
Wang, J.W., Wong, A.M., Flores, J., Vosshall, L.B. & Axel, R. Two-photon calcium imaging reveals an odor-evoked map of activity in the fly brain. Cell 112, 271–282 (2003).
Jayaraman, V. & Laurent, G. Evaluating a genetically encoded optical sensor of neural activity using electrophysiology in intact adult fruit flies. Front. Neural Circuits 1, 3 (2007).
Ng, M. et al. Transmission of olfactory information between three populations of neurons in the antennal lobe of the fly. Neuron 36, 463–474 (2002).
Churchland, M.M., Afshar, A. & Shenoy, K.V. A central source of movement variability. Neuron 52, 1085–1096 (2006).
Bohm, H., Schildberger, K. & Huber, F. Visual and acoustic course control in the cricket Gryllus-Bimaculatus. J. Exp. Biol. 159, 235–248 (1991).
Mason, A.C., Oshinsky, M.L. & Hoy, R.R. Hyperacute directional hearing in a microscale auditory system. Nature 410, 686–690 (2001).
Maimon, G., Straw, A.D. & Dickinson, M.H. Active flight increases the gain of visual motion processing in Drosophila. Nat. Neurosci. 13, 393–399 (2010).
Evarts, E.V. Relation of pyramidal tract activity to force exerted during voluntary movement. J. Neurophysiol. 31, 14–27 (1968).
Wurtz, R.H. Visual cortex neurons: response to stimuli during rapid eye movements. Science 162, 1148–1150 (1968).
Ono, T., Nakamura, K., Nishijo, H. & Fukuda, M. Hypothalamic neuron involvement in integration of reward, aversion, and cue signals. J. Neurophysiol. 56, 63–79 (1986).
Dombeck, D.A., Khabbaz, A.N., Collman, F., Adelman, T.L. & Tank, D.W. Imaging large-scale neural activity with cellular resolution in awake, mobile mice. Neuron 56, 43–57 (2007).
Egelhaaf, M. et al. Neural encoding of behaviourally relevant visual-motion information in the fly. Trends Neurosci. 25, 96–102 (2002).
Borst, A. & Haag, J. Neural networks in the cockpit of the fly. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 188, 419–437 (2002).
Joesch, M., Plett, J., Borst, A. & Reiff, D.F. Response properties of motion-sensitive visual interneurons in the lobula plate of Drosophila melanogaster. Curr. Biol. 18, 368–374 (2008).
Schnell, B. et al. Processing of horizontal optic flow in three visual interneurons of the Drosophila brain. J Neurophysiol. 103, 1646–1657 (2010).
Tian, L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 6, 875–881 (2009).
Hausen, K. & Wehrhahn, C. Neural circuits mediating visual flight control in flies. I. Quantitative comparison of neural and behavioral response characteristics. J. Neurosci. 9, 3828–3836 (1989).
Hausen, K. Motion sensitive interneurons in the optomotor system of the fly. 1. The horizontal cells—structure and signals. Biol. Cybern. 45, 143–156 (1982).
Hausen, K. Motion sensitive interneurons in the optomotor system of the fly. 2. The horizontal cells—receptive-field organization and response characteristics. Biol. Cybern. 46, 67–79 (1982).
Lehmann, F.O. & Dickinson, M.H. The changes in power requirements and muscle efficiency during elevated force production in the fruit fly Drosophila melanogaster. J. Exp. Biol. 200, 1133–1143 (1997).
Strausfeld, N.J. The head-neck system of the blowfly Calliphora. in The Head-Neck Sensory Motor System. (eds., Berthoz, A., Graf, W. and Vidal, P.P.) 56–63 (Oxford University Press, 1992).
Gotz, K.G. & Wenking, H. Visual control of locomotion in the walking fruitfly. Drosophila. J. Comp. Physiol. A Neuroethol. Sens. Neural Behav. Physiol. 85, 235–266 (1973).
Buchner, E. Elementary movement detectors in an insect visual-system. Biol. Cybern. 24, 85–101 (1976).
Lott, G.K., Rosen, M.J. & Hoy, R.R. An inexpensive sub-millisecond system for walking measurements of small animals based on optical computer mouse technology. J. Neurosci. Methods 161, 55–61 (2007).
Reiser, M.B. & Dickinson, M.H. A modular display system for insect behavioral neuroscience. J. Neurosci. Methods 167, 127–139 (2008).
Strauss, R., Schuster, S. & Gotz, K.G. Processing of artificial visual feedback in the walking fruit fly Drosophila melanogaster. J. Exp. Biol. 200, 1281–1296 (1997).
Scott, E.K., Raabe, T. & Luo, L. Structure of the vertical and horizontal system neurons of the lobula plate in Drosophila. J. Comp. Neurol. 454, 470–481 (2002).
Hausen, K. & Wehrhahn, C. Microsurgical lesion of horizontal cells changes optomotor yaw responses in the blowfly Calliphora Erythrocephala. Proc. Biol. Sci. 219, 211–216 (1983).
Geiger, G. & Nassel, D.R. Visual orientation behaviour of flies after selective laser beam ablation of interneurones. Nature 293, 398–399 (1981).
Blondeau, J. Electrically evoked course control in the fly Calliphora Erythrocephala. J. Exp. Biol. 92, 143–153 (1981).
Heisenberg, M., Wonneberger, R. & Wolf, R. Optomotor-blind—Drosophila mutant of lobula plate giant neurons. J. Comp. Physiol. 124, 287–296 (1978).
Durr, V. & Egelhaaf, M. In vivo calcium accumulation in presynaptic and postsynaptic dendrites of visual interneurons. J. Neurophysiol. 82, 3327–3338 (1999).
Haag, J. & Borst, A. Spatial distribution and characteristics of voltage-gated calcium signals within visual interneurons. J. Neurophysiol. 83, 1039–1051 (2000).
Haag, J. & Borst, A. Active membrane properties and signal encoding in graded potential neurons. J. Neurosci. 18, 7972–7986 (1998).
Pfeiffer, B.D. et al. Tools for neuroanatomy and neurogenetics in Drosophila. Proc. Natl. Acad. Sci. USA 105, 9715–9720 (2008).
Guizar-Sicairos, M., Thurman, S.T. & Fienup, J.R. Efficient subpixel image registration algorithms. Opt. Lett. 33, 156–158 (2008).
Reiff, D.F. et al. In vivo performance of genetically encoded indicators of neural activity in flies. J. Neurosci. 25, 4766–4778 (2005).
Olsen, S.R., Bhandawat, V. & Wilson, R.I. Excitatory interactions between olfactory processing channels in the Drosophila antennal lobe. Neuron 54, 89–103 (2007).
Bausenwein, B., Muller, N.R. & Heisenberg, M. Behavior-dependent activity labeling in the central complex, of Drosophila during controlled visual-stimulation. J. Comp. Neurol. 340, 255–268 (1994).
Branson, K., Robie, A.A., Bender, J., Perona, P. & Dickinson, M.H. High-throughput ethomics in large groups of Drosophila. Nat. Methods 6, 451–457 (2009).
Pologruto, T.A., Sabatini, B.L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 (2003).
Hopt, A. & Neher, E. Highly nonlinear photodamage in two-photon fluorescence microscopy. Biophys. J. 80, 2029–2036 (2001).
Acknowledgements
We thank J. Simpson and S. Hampel for cloning GCaMPs into pMUH; D. Hall and K. Hibbard for fly crossing; members of Janelia's Fly Core and particularly G. Zhang for stock maintenance; K. Svoboda for donations of equipment and for advice in setting up our two-photon microscopes; D. Flickinger, S. Bassin, T. Tabachnik and C. Werner for contributions to the optical and mechanical design; V. Iyer for developing new ScanImage features and for support; T. Ofstad for assistance with free-walking experiments; N. Kladt for assistance with calibration experiments; M. Ahrens for software contributions and P. Coen for carrying out pilot experiments with the ball tracker. B. Pfeiffer and G. Rubin (Janelia Farm) provided pMUH. G. Rubin gifted us R27B03-Gal4. For pilot experiments, D. Reiff (Max Planck Institute, Martinsried) provided DB331-Gal4 and L. Luo (Stanford University) provided line 3A-Gal4. We received generous advice and support from others at Janelia Farm, including members of the Svoboda lab. This work was supported by the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
J.D.S., M.E.C., G.K.L., M.B.R. and V.J. designed the project. J.D.S. designed the fly-physiology-with-behavior preparation with input from J.E.O., M.E.C. and V.J.; J.D.S., M.E.C., A.D., J.E.O. and V.J. designed the mechanical setup; M.B.R., J.D.S. and M.E.C. designed the LED arena; G.K.L. designed the ball tracker with input from M.B.R. and V.J.; M.E.C., J.D.S., M.B.R. and V.J. calibrated the tracker; V.J. performed high-speed video experiments; J.D.S. performed free-walking behavior experiments; J.D.S. and M.E.C. performed all other behavior and physiology experiments; J.D.S., M.E.C. and V.J. performed data analysis; M.E.C. performed fly crosses; and V.J., J.D.S., M.E.C., G.K.L. and M.B.R. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13 (PDF 1677 kb)
Supplementary Movie 1
Real-time movie showing multiple views of the fly walking in response to visual stimulation. Note: the xvid codec is required to play all movies. (AVI 5276 kb)
Supplementary Movie 2
Real-time movie showing fly-on-the-ball virtual two-dimensional trajectory during two-photon imaging trial in response to counterclockwise (blue) and then clockwise (red) global horizontal motion of vertical stripes (1-Hz spatial frequency). The frame rate was chosen to show fly's movements in real time. Frame size is 30 mm in the x dimension and 45 mm in the y dimension. (AVI 1580 kb)
Supplementary Movie 3
Real-time movie showing a second example of fly-on-the-ball virtual two-dimensional trajectory during two-photon imaging trial in response to counterclockwise (blue) and then clockwise (red) global horizontal motion of vertical stripes (1-Hz spatial frequency). The frame rate was chosen to show fly's movements in real time. Frame size is 30 mm in the x dimension and 45 mm in the y dimension. (AVI 1636 kb)
Supplementary Movie 4
Real-time movie showing (clockwise from top left): GCaMP signal from R27B03-Gal4 HS-neuron soma (shown in false color, linear intensity scale, window size: x = 62 μm, y = 67 μm); visual pattern presented to the fly (motion is first in the null direction for the HS neuron and then in the preferred direction); view of fly walking on ball from camera 3 (behind fly); traces of change in %ΔF/F (in green) and accumulated rotation (in black) as the motion stimuli are presented. Two-photon images were unfiltered (aside from xvid compression applied to entire movie) and not motion-corrected. (AVI 4623 kb)
Supplementary Movie 5
Real-time movie shows (clockwise from top left): GCaMP signal from R27B03-Gal4 HS-neuron dendrites (false color, linear intensity scale, window size: 43 μm); visual pattern presented to the fly (motion is first in the null direction for the HS neuron, then in the preferred direction and this protocol is repeated); view of fly walking on ball from camera 3 (behind fly); traces of change in %ΔF/F (in green) and accumulated rotation (in black) as the motion stimuli are presented. Two-photon images are unfiltered (aside from xvid compression of the movie) and not motion-corrected. (AVI 5414 kb)
Rights and permissions
About this article
Cite this article
Seelig, J., Chiappe, M., Lott, G. et al. Two-photon calcium imaging from head-fixed Drosophila during optomotor walking behavior. Nat Methods 7, 535–540 (2010). https://doi.org/10.1038/nmeth.1468
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1468
This article is cited by
-
Homeostatic synaptic plasticity rescues neural coding reliability
Nature Communications (2023)
-
The spatial and temporal structure of neural activity across the fly brain
Nature Communications (2023)
-
A rapid and bidirectional reporter of neural activity reveals neural correlates of social behaviors in Drosophila
Nature Neuroscience (2023)
-
A single photoreceptor splits perception and entrainment by cotransmission
Nature (2023)
-
Real-time denoising enables high-sensitivity fluorescence time-lapse imaging beyond the shot-noise limit
Nature Biotechnology (2023)