Abstract
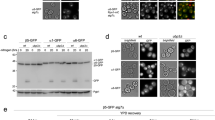
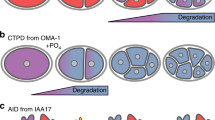
The ubiquitin-proteasome system (UPS) orchestrates many cellular and tissue-specific processes by degrading damaged and key regulatory proteins. To enable investigation of UPS activity in different cell types in a living animal, we developed a photoconvertible fluorescent UPS reporter system for live imaging and quantification of protein degradation in Caenorhabditis elegans. Our reporter consists of the photoconvertible fluorescent protein Dendra2 targeted for proteasomal degradation by fusion to the UbG76V mutant form of ubiquitin. In contrast to previous reporters, this system permits quantification of UPS activity independently of protein synthesis. Our reporter revealed that UPS-mediated protein degradation varies in a cell type–specific and age-dependent manner in C. elegans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
17 June 2010
In the version of this paper originally published, a reference to previous work on the use of Dendra2 as a reporter for protein stability in cultured cells should have been included (ref. 35). The error has been corrected in the PDF and HTML versions of the article.
References
Hanna, J. & Finley, D. A proteasome for all occasions. FEBS Lett. 581, 2854–2861 (2007).
Paul, S. Dysfunction of the ubiquitin-proteasome system in multiple disease conditions: therapeutic approaches. Bioessays 30, 1172–1184 (2008).
Vernace, V.A., Schmidt-Glenewinkel, T. & Figueiredo-Pereira, M.E. Aging and regulated protein degradation: who has the UPPer hand? Aging Cell 6, 599–606 (2007).
Dantuma, N.P., Lindsten, K., Glas, R., Jellne, M. & Masucci, M.G. Short-lived green fluorescent proteins for quantifying ubiquitin/proteasome-dependent proteolysis in living cells. Nat. Biotechnol. 18, 538–543 (2000).
Bence, N.F., Sampat, R.M. & Kopito, R.R. Impairment of the ubiquitin-proteasome system by protein aggregation. Science 292, 1552–1555 (2001).
Holmberg, C.I., Staniszewski, K.E., Mensah, K.N., Matouschek, A. & Morimoto, R.I. Inefficient degradation of truncated polyglutamine proteins by the proteasome. EMBO J. 23, 4307–4318 (2004).
Lindsten, K., Menéndez-Benito, V., Masucci, M.G. & Dantuma, N.P. A transgenic mouse model of the ubiquitin/proteasome system. Nat. Biotechnol. 21, 897–902 (2003).
Kumarapeli, A.R. et al. A novel transgenic mouse model reveals deregulation of the ubiquitin-proteasome system in the heart by doxorubicin. FASEB J. 19, 2051–2053 (2005).
Bove, J. et al. Proteasome inhibition and Parkinson′s disease modeling. Ann. Neurol. 60, 260–264 (2006).
Cook, C. et al. Aging is not associated with proteasome impairment in UPS reporter mice. PLoS One 4, e5888 (2009).
Alvarez-Castelao, B., Martin-Guerrero, I., Garcia-Orad, A. & Castano, J.G. CMV promoter up-regulation is the major cause of increased protein levels of unstable reporter proteins after treatment of living cells with proteasome inhibitors. J. Biol. Chem. 41, 28253–28262 (2009).
Johnson, E.S., Bartel, B., Seufert, W. & Varshavsky, A. Ubiquitin as a degradation signal. EMBO J. 11, 497–505 (1992).
Chudakov, D.M., Lukyanov, S. & Lukyanov, K.A. Tracking intracellular protein movements using photoswitchable fluorescent proteins PS-CFP2 and Dendra2. Nat. Protocols 2, 2024–2032 (2007).
Jantsch-Plunger, V. & Fire, A. Combinatorial structure of a body muscle-specific transcriptional enhancer in Caenorhabditis elegans. J. Biol. Chem. 269, 27021–27028 (1994).
Shimada, M., Kanematsu, K., Tanaka, K., Yokosawa, H. & Kawahara, H. Proteasomal ubiquitin receptor RPN-10 controls sex determination in Caenorhabditis elegans. Mol. Biol. Cell 17, 5356–5371 (2006).
van Nocker, S. et al. The multiubiquitin-chain-binding protein Mcb1 is a component of the 26S proteasome in Saccharomyces cerevisiae and plays a nonessential, substrate-specific role in protein turnover. Mol. Cell. Biol. 16, 6020–6028 (1996).
Stack, J.H., Whitney, M., Rodems, S.M. & Pollok, B.A. A ubiquitin-based tagging system for controlled modulation of protein stability. Nat. Biotechnol. 18, 1298–1302 (2000).
Verhoef, L.G. et al. Minimal length requirement for proteasomal degradation of ubiquitin-dependent substrates. FASEB J. 1, 123–133 (2008).
Kim, H.T., Kim, K.P., Uchiki, T., Gygi, S.P. & Goldberg, A.L. S5a promotes protein degradation by blocking synthesis of nondegradable forked ubiquitin chains. EMBO J. 13, 1867–1877 (2009).
Breusing, N. & Grune, T. Regulation of proteasome-mediated protein degradation during oxidative stress and aging. Biol. Chem. 389, 203–209 (2008).
Yun, C. et al. Proteasomal adaptation to environmental stress links resistance to proteotoxicity with longevity in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 105, 7094–7099 (2008).
Ghazi, A., Henis-Korenblit, S. & Kenyon, C. Regulation of Caenorhabditis elegans lifespan by a proteasomal E3 ligase complex. Proc. Natl. Acad. Sci. USA 104, 5947–5952 (2007).
Walker, G.A. & Lithgow, G.J. Lifespan extension in C. elegans by a molecular chaperone dependent upon insulin-like signals. Aging Cell 2, 131–139 (2003).
Yokoyama, K. et al. Extended longevity of Caenorhabditis elegans by knocking in extra copies of hsp70F, a homolog of mot-2 (mortalin)/mthsp70/Grp75. FEBS Lett. 516, 53–57 (2002).
Hsu, A.L., Murphy, C.T. & Kenyon, C. Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300, 1142–1145 (2003).
Mehta, R. et al. Proteasomal regulation of the hypoxic response modulates aging in C. elegans. Science 324, 1196–1198 (2009).
Li, W., Gao, B., Lee, S.M., Bennett, K. & Fang, D. RLE-1, an E3 ubiquitin ligase, regulates C. elegans aging by catalyzing DAF-16 polyubiquitination. Dev. Cell 12, 235–246 (2007).
Morley, J.F. & Morimoto, R.I. Regulation of longevity in Caenorhabditis elegans by heat shock factor and molecular chaperones. Mol. Biol. Cell 15, 657–664 (2004).
Herndon, L.A. et al. Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans. Nature 419, 808–814 (2002).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Brignull, H.R., Moore, F.E., Tang, S.J. & Morimoto, R.I. Polyglutamine proteins at the pathogenic threshold display neuron-specific aggregation in a pan-neuronal Caenorhabditis elegans model. J. Neurosci. 26, 7597–7606 (2006).
Mello, C.C., Kramer, J.M., Stinchcomb, D. & Ambros, V. Efficient gene transfer in C. elegans: extrachromosomal maintenance and integration of transforming sequences. EMBO J. 10, 3959–3970 (1991).
Timmons, L., Court, D.L. & Fire, A. Ingestion of bacterially expressed dsRNAs can produce specific and potent genetic interference in Caenorhabditis elegans. Gene 263, 103–112 (2001).
Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Zhang, L. et al. Method for real-time monitoring of protein degradation at the single cell level. Biotechniques 42, 446–450 (2007).
Acknowledgements
This study was supported by grants to C.I.H. from the Academy of Finland (113485 and 118450), the International Human Frontier Science Program Organization, Biocentrum Helsinki, the Sigrid Jusélius Foundation and the Magnus Ehrnrooth Foundation. G.H. was additionally supported by the University of Helsinki and O.M. by the Helsinki Biomedical Graduate School. We thank members of the Caenorhabditis Genetics Center for providing the N2 wild-type worms, S. Mitani (National Bioresource Project for the Nematode, Japan) for the rpn-10 mutant worms, R.I. Morimoto (Northwestern University) for the PF25B3.3::yfp expression vector and members of the Biomedicum Helsinki Molecular Imaging Unit for their help with confocal microscopy and imaging.
Author information
Authors and Affiliations
Contributions
G.H. and O.M. developed the method, performed the experiments, analyzed the data, made the figures and wrote the paper. C.I.H. developed the method, analyzed the data, wrote the paper and supported the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 and Supplementary Tables 1–2 (PDF 2945 kb)
Rights and permissions
About this article
Cite this article
Hamer, G., Matilainen, O. & Holmberg, C. A photoconvertible reporter of the ubiquitin-proteasome system in vivo. Nat Methods 7, 473–478 (2010). https://doi.org/10.1038/nmeth.1460
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1460
This article is cited by
-
USP10 regulates B cell response to SARS-CoV-2 or HIV-1 nanoparticle vaccines through deubiquitinating AID
Signal Transduction and Targeted Therapy (2022)
-
Tissue-specific effects of temperature on proteasome function
Cell Stress and Chaperones (2020)
-
L3MBTL1 regulates ALS/FTD-associated proteotoxicity and quality control
Nature Neuroscience (2019)
-
The role of protein clearance mechanisms in organismal ageing and age-related diseases
Nature Communications (2014)
-
Sensitive detection of proteasomal activation using the Deg-On mammalian synthetic gene circuit
Nature Communications (2014)