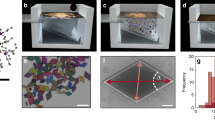
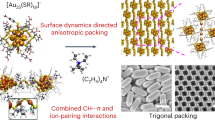
Abstract
Understanding how polyhedra pack into extended arrangements is integral to the design and discovery of crystalline materials at all length scales1,2,3. Much progress has been made in enumerating and characterizing the packing of polyhedral shapes4,5,6, and the self-assembly of polyhedral nanocrystals into ordered superstructures7,8,9. However, directing the self-assembly of polyhedral nanocrystals into densest packings requires precise control of particle shape10, polydispersity11, interactions and driving forces 12. Here we show with experiment and computer simulation that a range of nanoscale Ag polyhedra can self-assemble into their conjectured densest packings6. When passivated with adsorbing polymer, the polyhedra behave as quasi-hard particles and assemble into millimetre-sized three-dimensional supercrystals by sedimentation. We also show, by inducing depletion attraction through excess polymer in solution, that octahedra form an exotic superstructure with complex helical motifs rather than the densest Minkowski lattice13. Such large-scale Ag supercrystals may facilitate the design of scalable three-dimensional plasmonic metamaterials for sensing14,15, nanophotonics16 and photocatalysis17.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wells, A. F. Structural Inorganic Chemistry (Oxford Univ. Press, 1984).
O’Keeffe, M. & Hyde, B. G. Crystal Structures I. Patterns and Symmetry (Mineralogical Society of America, 1996).
Murray, C. B., Kagan, C. R. & Bawendi, M. G. Synthesis and characterization of monodisperse nanocrystals and close-packed nanocrystal assemblies. Annu. Rev. Mater. Sci. 30, 545–610 (2000).
Betke, U. Densest lattice packings of 3-polytopes. Comput. Geom. 16, 157–186 (2000).
Haji-Akbari, A. et al. Disordered, quasicrystalline and crystalline phases of densely packed tetrahedra. Nature 462, 773–777 (2009).
Torquato, S. & Jiao, Y. Dense packings of the Platonic and Archimedean solids. Nature 460, 876–879 (2009).
Tao, A. R., Ceperley, D. P., Sinsermsuksakul, P., Neureuther, A. R. & Yang, P. Self-organized silver nanoparticles for three-dimensional plasmonic crystals. Nano Lett. 8, 4033–4038 (2008).
Xie, S. et al. Supercrystals from crystallization of octahedral MnO nanocrystals. J. Phys. Chem. C 113, 19107–19111 (2009).
Quan, Z. & Fang, J. Superlattices with non-spherical building blocks. Nano Today 5, 390–411 (2010).
Glotzer, S. C. & Solomon, M. S. Anisotropy of building blocks and their assembly into complex structures. Nature Mater. 6, 557 (2007).
Auer, S. & Frenkel, D. Suppression of crystal nucleation in polydisperse colloids due to increase of the surface free energy. Nature 413, 711–713 (2001).
Bishop, K. J. M., Wilmer, C. E., Soh, S. & Grzybowski, B. A. Nanoscale forces and their uses in self-assembly. Small 5, 1600–1630 (2009).
Minkowski, H. Dichteste gitterförmige Lagerung kongruenter Körper. Nachr. Akad. Wiss. Göttingen Math.-Phys. Kl. II, 311–355 (1904).
Kabashin, A. V. et al. Plasmonic nanorod metamaterials for biosensing. Nature Mater. 8, 867–871 (2009).
Novo, C., Funston, A. M. & Mulvaney, P. Direct observation of chemical reactions on single gold nanocrystals using surface plasmon spectroscopy. Nature Nanotech. 3, 598–602 (2008).
Pendry, J. B., Schurig, D. & Smith, D. R. Controlling electromagnetic fields. Science 312, 1780–1782 (2006).
Hung, W. H., Aykol, M., Valley, D., Hou, W. & Cronin, S. B. Plasmon resonant enhancement of carbon monoxide catalysis. Nano Lett. 10, 1314–8 (2010).
Tao, A., Sinsermsuksakul, P. & Yang, P. Polyhedral silver nanocrystals with distinct scattering signatures. Angew. Chem. Int. Ed. 45, 4597–4601 (2006).
Sun, Y. & Xia, Y. Shape-controlled synthesis of gold and silver nanoparticles. Science 298, 2176–2179 (2002).
Chen, E. R., Engel, M. & Glotzer, S. C. Dense crystalline dimer packings of regular tetrahedra. Discrete Comput. Geom. 44, 253–280 (2010).
Kallus, Y., Elser, V. & Gravel, S. Dense periodic packings of tetrahedra with small repeating units. Discrete Comput. Geom. 44, 245–252 (2010).
Torquato, S. & Jiao, Y. Exact constructions of a family of dense periodic packings of tetrahedra. Phys. Rev. E 81, 041310 (2010).
Weeks, J. D., Chandler, D. & Andersen, H. C. Role of repulsive forces in determining the equilibrium structure of simple liquids. J. Chem. Phys. 54, 5237–5247 (1971).
Ruths, M., Yoshizawa, H., Fetters, L. J. & Israelachvili, J. N. Depletion attraction versus steric repulsion in a system of weakly adsorbing polymers—effects of concentration and adsorption conditions. Macromolecules 29, 7193–7203 (1996).
Asakura, S. & Oosawa, F. Interaction between particles suspended in solutions of macromolecules. J. Polym. Sci. 33, 183–192 (1958).
Baranov, D. et al. Assembly of colloidal semiconductor nanorods in solution by depletion attraction. Nano Lett. 10, 743–749 (2010).
Dogic, Z. & Fraden, S. Development of model colloidal liquid crystals and the kinetics of the isotropic–smectic transition. Phil. Tran. R. Soc. A 359, 997–1015 (2001).
Hanfland, M., Syassen, K., Christensen, N. E. & Novikov, D. L. New high-pressure phases of lithium. Nature 408, 174–178 (2000).
Rabani, E., Reichman, D. R., Geissler, P. L. & Brus, L. E. Drying-mediated self-assembly of nanoparticles. Nature 426, 271–274 (2003).
Ozin, G. A., Arsenault, A. C. & Cardemartiri, L. in Nanochemistry: A Chemical Approach to Nanomaterials 2nd edn 335–428 (RSC Publishing, 2008).
Talapin, D. V., Lee, J-S., Kovalenko, M. V. & Shevchenko, E. V. Prospects of colloidal nanocrystals for electronic and optoelectronic applications. Chem. Rev. 110, 389–458 (2010).
Stebe, K. J., Lewandowski, E. & Ghosh, M. Materials science. Oriented assembly of metamaterials. Science 325, 159–160 (2009).
Louis, A. A., Bolhuis, P. G., Hansen, J. P. & Meijer, E. J. Can polymer coils be modeled as ‘soft colloids’? Phys. Rev. Lett. 85, 2522–2525 (2000).
Acknowledgements
This work was partially supported by the Defense Advanced Research Projects Agency. The shape-selective synthesis part of the work is supported by the Director, Office of Basic Energy Sciences, Materials Sciences and Engineering Division, of the US Department of Energy under contract no DE-AC02-05CH11231. M.G. was supported by the Austrian Science Fund (FWF) under grant no J 3106-N16. M.G. and P.L.G. were supported by the National Science Foundation under grant no CHE-0910981. A.W-C. was supported by the US Department of Energy under contract no DE-AC02-05CH11231. Work at the Molecular Foundry was supported by the Office of Science, Office of Basic Energy Sciences, of the US Department of Energy under contract no DE-AC02-05CH11231. P.Y. thanks the National Science Foundation for the Waterman Award.
Author information
Authors and Affiliations
Contributions
J.H. and M.G. both contributed extensively to this work. J.H. initiated the study, conceived and conducted the experiments, analysed results and cowrote the paper. M.G. identified all crystal structures, conceived and implemented the simulations and theoretical models, analysed results and cowrote the paper. A.W-C. suggested simulations and experiments, analysed results, helped prepare figures and cowrote the paper. P.L.G. suggested simulations and experiments, analysed results and cowrote the paper. P.Y. initiated the study, suggested experiments, analysed results and cowrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1379 kb)
Supplementary Information
Supplementary Information (PDF 12 kb)
Supplementary Information
Supplementary Movie (MPG 2786 kb)
Supplementary Information
Supplementary Movie (MPG 4372 kb)
Supplementary Information
Supplementary Movie (MPG 6196 kb)
Supplementary Information
Supplementary Movie (MPG 4507 kb)
Rights and permissions
About this article
Cite this article
Henzie, J., Grünwald, M., Widmer-Cooper, A. et al. Self-assembly of uniform polyhedral silver nanocrystals into densest packings and exotic superlattices. Nature Mater 11, 131–137 (2012). https://doi.org/10.1038/nmat3178
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3178
This article is cited by
-
Bioinspired magnetic cilia: from materials to applications
Microsystems & Nanoengineering (2023)
-
Metal-organic framework template-guided electrochemical lithography on substrates for SERS sensing applications
Nature Communications (2023)
-
Mesoporous colloidal silica cubes with catalytically active cores
Journal of Porous Materials (2023)
-
Low-dimensional assemblies of metal-organic framework particles and mutually coordinated anisotropy
Nature Communications (2022)
-
Assembly of planar chiral superlattices from achiral building blocks
Nature Communications (2022)