Abstract
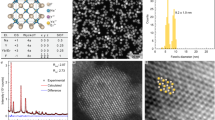
Nanocrystals with advanced magnetic or optical properties have been actively pursued for potential biological applications, including integrated imaging, diagnosis and therapy1,2,3,4,5,6,7,8,9. Among various magnetic nanocrystals10,11,12,13,14,15, FeCo has superior magnetic properties, but it has yet to be explored owing to the problems of easy oxidation and potential toxicity10,16,17. Previously, FeCo nanocrystals with multilayered graphitic carbon, pyrolytic carbon or inert metals have been obtained15,18,19, but not in the single-shelled, discrete, chemically functionalized and water-soluble forms desired for biological applications. Here, we present a scalable chemical vapour deposition method to synthesize FeCo/single-graphitic-shell nanocrystals that are soluble and stable in water solutions. We explore the multiple functionalities of these core–shell materials by characterizing the magnetic properties of the FeCo core and near-infrared optical absorbance of the single-layered graphitic shell. The nanocrystals exhibit ultra-high saturation magnetization, r1 and r2 relaxivities and high optical absorbance in the near-infrared region. Mesenchymal stem cells are able to internalize these nanoparticles, showing high negative-contrast enhancement in magnetic-resonance imaging (MRI). Preliminary in vivo experiments achieve long-lasting positive-contrast enhancement for vascular MRI in rabbits. These results point to the potential of using these nanocrystals for integrated diagnosis and therapeutic (photothermal-ablation) applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lewin, M. et al. Tat peptide-derivatized magnetic nanoparticles allow in vivo tracking and recovery of progenitor cells. Nature Biotechnol. 18, 410–414 (2000).
Alivisatos, P. The use of nanocrystals in biological detection. Nature Biotechnol. 22, 47–52 (2004).
Kim, S. et al. Near-infrared fluorescent type II quantum dots for sentinel lymph node mapping. Nature Biotechnol. 22, 93–97 (2004).
Hirsch, L. R. et al. Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc. Natl Acad. Sci. USA 100, 13549–13554 (2003).
Liao, H. W. & Hafner, J. H. Gold nanorod bioconjugates. Chem. Mater. 17, 4636–4641 (2005).
Huang, X., El-Sayed, I. H., Qian, W. & El-Sayed, M. A. Cancer cell imaging and photothermal therapy in the near-infrared region by using gold nanorods. J. Am. Chem. Soc. 128, 2115–2120 (2006).
Chen, C.-C. et al. DNA-gold nanorod conjugates for remote control of localized gene expression by near infrared irradiation. J. Am. Chem. Soc. 128, 3709–3715 (2006).
Kopelman, R. et al. Multifunctional nanoparticle platforms for in vivo MRI enhancement and photodynamic therapy of a rat brain cancer. J. Magn. Magn. Mater. 293, 404–410 (2005).
McCarthy, J. R., Jaffer, F. A. & Weissleder, R. A macrophage-targeted theranostic nanoparticle for biomedical applications. Small 2, 983–987 (2006).
Hütten, A. et al. New magnetic nanoparticles for biotechnology. J. Biotechnol. 112, 47–63 (2004).
Dinega, D. P. & Bawendi, M. G. A solution-phase chemical approach to a new crystal structure of cobalt. Angew. Chem. Int. Edn. 38, 1788–1791 (1999).
Puntes, V. F., Zanchet, D., Erdonmez, C. K. & Alivisatos, A. P. Synthesis of hcp-Co nanodisks. J. Am. Chem. Soc. 124, 12874–12880 (2002).
Sun, S., Murray, C. B., Weller, D., Folks, L. & Moser, A. Monodisperse FePt nanoparticles and ferromagnetic FePt nanocrystal superlattices. Science 287, 1989–1992 (2000).
Park, J. et al. Ultra-large-scale syntheses of monodisperse nanocrystals. Nature Mater. 3, 891–895 (2004).
Desvaux, C. et al. Multimillimetre-large superlattices of air-stable iron–cobalt nanoparticles. Nature Mater. 4, 750–753 (2005).
Bardos, D. I. Mean magnetic moments in bcc Fe–Co alloys. J. Appl. Phys. 40, 1371–1372 (1969).
Reiss, G. & Hütten, A. Magnetic nanoparticles: applications beyond data storage. Nature Mater. 4, 725–726 (2005).
Turgut, Z., Scott, J. H., Huang, M. Q., Majetich, S. A. & McHenry, M. E. Magnetic properties and ordering in C-coated FexCo1−x alloy nanocrystals. J. Appl. Phys. 83, 6468–6470 (1998).
Bai, J. & Wang, J.-P. High-magnetic-moment core-shell-type FeCo-Au/Ag nanoparticles. Appl. Phys. Lett. 87, 152502 (2005).
Tuinstra, F. & Koenig, J. L. Raman spectrum of graphite. J. Chem. Phys. 53, 1126–1130 (1970).
Lu, Y., Zhu, Z. & Liu, Z. Carbon-encapsulated Fe nanoparticles from detonation-induced pyrolysis of ferrocene. Carbon 43, 369–374 (2005).
Li, Y. et al. Growth of single-walled carbon nanotubes from discrete catalytic nanoparticles of various sizes. J. Phys. Chem. 105, 11424–11431 (2001).
Kam, N. W. S., Liu, Z. & Dai, H. Functionalization of carbon nanotubes via cleavable disulfide bonds for efficient intracellular delivery of siRNA and potent gene silencing. J. Am. Chem. Soc. 127, 12492–12493 (2005).
Kam, N. W. S., O’Connell, M., Wisdom, J. A. & Dai, H. Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc. Natl Acad. Sci. USA 102, 11600–11605 (2005).
Cataldo, F. An investigation on the optical properties of carbon black, fullerite, and other carbonaceous materials in relation to the spectrum of interstellar extinction of light. Full. Nanotubes Carb. Nanostruct. 10, 155–170 (2002).
Jung, C. W. & Jacobs, P. Physical and chemical properties of superparamagnetic iron oxide MR contrast agents: ferumoxides, ferumoxtran, ferumoxsil. Magn. Reson. Imag. 13, 661–674 (1995).
Kellar, K. E. et al. NC100150 injection, a preparation of optimized iron oxide nanoparticles for positive-contrast MR angiography. J. Magn. Reson. Imag. 11, 488–494 (2000).
Huang, D.-M. et al. Highly efficient cellular labeling of mesoporous nanoparticles in human mesenchymal stem cells: implication for stem cell tracking. FASEB J. 19, 2014–2016 (2005).
Kam, N. W. S. & Dai, H. Carbon nanotubes as intracellular protein transporters: Generality and biological functionality. J. Am. Chem. Soc. 127, 6021–6026 (2005).
Bianco, A., Kostarelos, K., Partidos, C. D. & Prato, M. Biomedical applications of functionalised carbon nanotubes. Chem. Commun. 571–577 (2005).
Acknowledgements
This work was supported in part by a Korea Research Foundation Grant (KRF-2005-214-C00074), a Ludwig Translational Research Grant at Stanford University, a NIH-NCI Cancer Center for Nanotechnology Excellence-Therapy Response (CCNE-TR) grant (# 1 U54 CA119367-01) at Stanford, a NIH RO1 grant (#HLO78678), and the National Center for Electron Microscopy, Lawrence Berkeley National Laboratory, which is supported by the US Department of Energy, supported this work in part. We thank H. Li for help with TEM.
Author information
Authors and Affiliations
Contributions
W.S.S., D.M. and H.D. carried out the design and experiments of synthesis, characterization and functionalization. X.M.S. and Z.L. carried out cytotoxicity assays. P.Y. and Y.S. participated in the stem cell experiment. M.T. and M.V.M carried out rabbit injections. J.H.L. and D.G.N. carried out MRI experiments. H.D., W.S.S. and J.H.L. designed the research and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary information and figures S1-S3 (PDF 63 kb)
Rights and permissions
About this article
Cite this article
Seo, W., Lee, J., Sun, X. et al. FeCo/graphitic-shell nanocrystals as advanced magnetic-resonance-imaging and near-infrared agents. Nature Mater 5, 971–976 (2006). https://doi.org/10.1038/nmat1775
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat1775
This article is cited by
-
Shape Memory Polymer-Based Nanocomposites Magnetically Enhanced with Fe3O4 Nanoparticles
Journal of Inorganic and Organometallic Polymers and Materials (2023)
-
Applications of Graphene-Based Nanomaterials as Contrast Agents in Biomedical Imaging Technologies: A Review
Biomedical Materials & Devices (2023)