Abstract
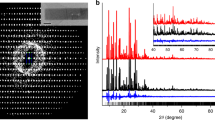
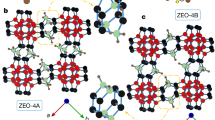
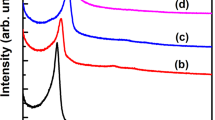
Zeolites are a family of crystalline aluminosilicate materials widely used as shape-selective catalysts, ion exchange materials, and adsorbents for organic compounds1,2. In the present work, zeolites were synthesized by adding a rationally designed amphiphilic organosilane surfactant to conventional alkaline zeolite synthesis mixtures. The zeolite products were characterized by a complementary combination of X-ray diffraction (XRD), nitrogen sorption, scanning electron microscopy (SEM) and transmission electron microscopy (TEM). The analyses show that the present method is suitable as a direct synthesis route to highly mesoporous zeolites. The mesopore diameters could be uniformly tailored, similar to ordered mesoporous silica with amorphous frameworks3. The mesoporous zeolite exhibited a narrow, small-angle XRD peak, which is characteristic of the short-range correlation between mesopores, similar to disordered wormhole-like mesoporous materials4,5. The XRD patterns and electron micrographs of the samples taken during crystallization clearly showed the evolution of the mesoporous structure concomitantly to the crystallization of zeolite frameworks. The synthesis of the crystalline aluminosilicate materials with tunable mesoporosity and strong acidity has potentially important technological implications for catalytic reactions of large molecules, whereas conventional mesoporous materials lack hydrothermal stability and acidity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Cundy, C. S. & Cox, P. A. The hydrothermal synthesis of zeolites: history and development from the earliest days to the present time. Chem. Rev. 103, 663–701 (2003).
Corma, A. From microporous to mesoporous molecular sieve materials and their use in catalysis. Chem. Rev. 97, 2373–2419 (1997).
Kresge, C. T. et al. Ordered mesoporous molecular-sieves synthesized by a liquid-crystal template mechanism. Nature 359, 710–712 (1992).
Tanev, P. T. & Pinnavaia, T. J. A neutral templating route to mesoporous molecular sieves. Science 267, 865–867 (1995).
Ryoo, R., Kim, J. M., Ko, C. H. & Shin, C. H. Disordered molecular sieve with branched mesoporous channel network. J. Phys. Chem. 100, 17718–17721 (1996).
Zhao, D. Y. et al. Triblock copolymer syntheses of mesoporous silica with periodic 50 to 300 angstrom pores. Science 279, 548–552 (1998).
Kim, J. M. & Ryoo, R. Disintegration of mesoporous structures of MCM-41 and MCM-48 in water. Bull. Korean Chem. Soc. 17, 66–68 (1996).
Cassiers, K. et al. A detailed study of thermal, hydrothermal, and mechanical stabilities of a wide range of surfactant assembled mesoporous silicas. Chem. Mater. 14, 2317–2324 (2002).
Zhao, D. et al. Comparison of disordered mesoporous aluminosilicates with highly ordered Al-MCM-41 on stability, acidity and catalytic activity. Catal. Today 68, 11–20 (2001).
Beck, J. S. et al. Molecular or supramolecular templating: defining the role of surfactant chemistry in the formation of microporous and mesoporous molecular sieves. Chem. Mater. 6, 1816–1821 (1994).
Karlsson, A., Stocker, M. & Schmidt, R. Composites of micro- and mesoporous materials: Simultaneous syntheses of MFI/MCM-41 like phases by a mixed template approach. Micropor. Mesopor. Mater. 27, 181–192 (1999).
Petkov, N., Holzl, M., Metzger, T. H., Mintova, S. & Bein, T. Ordered micro/mesoporous composite prepared as thin films. J. Phys. Chem. B 109, 4485–4491 (2005).
Christiansen, S. C. et al. Molecularly ordered inorganic frameworks in layered silicate surfactant mesophases. J. Am. Chem. Soc. 123, 4519–4529 (2001).
Hedin, N. et al. Structure of a surfactant-templated silicate framework in the absence of 3D crystallinity. J. Am. Chem. Soc. 126, 9425–9432 (2004).
Liu, Y., Zhang, W. Z. & Pinnavaia, T. J. Steam-stable aluminosilicate mesostructures assembled from zeolite type Y seeds. J. Am. Chem. Soc. 122, 8791–8792 (2000).
Liu, Y., Zhang, W. Z. & Pinnavaia, T. J. Steam-stable MSU-S aluminosilicate mesostructures assembled from zeolite ZSM-5 and zeolite beta seeds. Angew. Chem. Int. Edn 40, 1255–1258 (2001).
Zhang, Z. T. et al. Mesoporous aluminosilicates with ordered hexagonal structure, strong acidity, and extraordinary hydrothermal stability at high temperatures. J. Am. Chem. Soc. 123, 5014–5021 (2001).
Liu, Y. & Pinnavaia, T. J. Assembly of hydrothermally stable aluminosilicate foams and large-pore hexagonal mesostructures from zeolite seeds under strongly acidic conditions. Chem. Mater. 14, 3–5 (2002).
On, D. T. & Kaliaguine, S. Zeolite-coated mesostructured cellular silica foams. J. Am. Chem. Soc. 125, 618–619 (2003).
Do, T. O. et al. Zeolite nanoclusters coated onto the mesopore walls of SBA-15. J. Am. Chem. Soc. 126, 14324–14325 (2004).
Xia, Y. D. & Mokaya, R. Are mesoporous silicas and aluminosilicas assembled from zeolite seeds inherently hydrothermally stable? Comparative evaluation of MCM-48 materials assembled from zeolite seeds. J. Mater. Chem. 14, 3427–3435 (2004).
Jacobsen, C. J. H. et al. Mesoporous zeolite single crystals. J. Am. Chem. Soc. 122, 7116–7117 (2000).
Tao, Y. S., Kanoh, H. & Kaneko, K. ZSM-5 monolith of uniform mesoporous channels. J. Am. Chem. Soc. 125, 6044–6045 (2003).
Schmidt, I. et al. Carbon nanotube templated growth of mesoporous zeolite single crystals. Chem. Mater. 13, 4416–4418 (2001).
Sakthivel, A. et al. Replication of mesoporous aluminosilicate molecular sieves (RMMs) with zeolite framework from mesoporous carbons (CMKs). Chem. Mater. 16, 3168–3175 (2004).
Holland, B. T., Abrams, L. & Stein, A. Dual templaing of macroporous silicates with zeolitic microporous frameworks. J. Am. Chem. Soc. 121, 4308–4309 (1999).
Yang, X. Y. et al. Design and size control of uniform zeolite nanocrystals synthesized in adjustable confined voids formed by recyclable monodisperse polymer spheres. Angew. Chem. Int. Edn 44, 2563–2568 (2005).
Kröger, N., Deutzmann, R. & Sumper, M. Polycationic peptides from diatom biosilica that direct silica nanosphere formation. Science 286, 1129–1132 (1999).
Qiu, S. R. et al. Modulation of calcium oxalate monohydrate crystallization by citrate through selective binding to atomic steps. J. Am. Chem. Soc. 127, 9036–9044 (2005).
Hűttinger, K. J. & Jung, M. F. Kinetik der synthese von trialkyl[-3-(trimethoxysilyl)-propyl]-ammoniumchloriden und deren antimikrobielle wirkung als fixierte biozide. Chem. Ing. Tech. 61, 258–259 (1989).
Jun, S. & Ryoo, R. Aluminum impregnation into mesoporous silica molecular sieves for catalytic application to Friedel–Crafts alkylation. J. Catal. 195, 237–243 (2000).
Che, S. et al. Direct observation of 3D mesoporous structure by scanning electron microscopy (SEM): SBA-15 silica and CMK-5 carbon. Angew. Chem. Int. Edn 42, 2182–2185 (2003).
Barrett, E. P., Joyner, L. G. & Halenda, P. P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 73, 373–380 (1951).
Acknowledgements
This work was supported by the Ministry of Science and Technology through the Creative Research Initiative Program, and by the School of Molecular Science through the Brain Korea 21Project. Synchrotron radiation XRD was supported by Pohang Light Source. The authors are grateful to S. Bai, and I. Lee for helpful discussions on electron microscopy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary figures S1-S5 (PDF 1961 kb)
Rights and permissions
About this article
Cite this article
Choi, M., Cho, H., Srivastava, R. et al. Amphiphilic organosilane-directed synthesis of crystalline zeolite with tunable mesoporosity. Nature Mater 5, 718–723 (2006). https://doi.org/10.1038/nmat1705
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat1705
This article is cited by
-
Liquid-phase benzylation of toluene by benzyl alcohol over micro-mesoporous hierarchical mordenite zeolite
Environmental Science and Pollution Research (2023)
-
Synthesis strategies and design principles for nanosized and hierarchical zeolites
Nature Synthesis (2022)
-
Effective and Stable Zeolite Imidazole Framework-Supported Copper Nanoparticles (Cu/ZIF-8) for Glycerol to Lactic Acid
Catalysis Letters (2022)
-
Monosodium glutamate-mediated hierarchical porous formation in LTA zeolite to enhance CO2 adsorption performance
Journal of Sol-Gel Science and Technology (2021)
-
Direct synthesis of hierarchical zeolites through mesoporous 3D wormhole framework using single-head quaternary ammonium salt as structure-directing agent
Journal of Materials Science (2021)