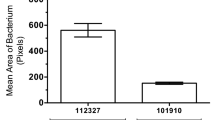
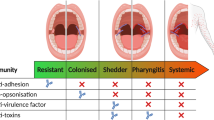
Abstract
The role of host genetic factors in conferring predisposition or protection in infectious diseases has become evident. Infection with group A streptococci causes a wide spectrum of disease ranging from pharyngitis to streptococcal toxic shock syndrome. The release of inflammatory cytokines triggered by streptococcal superantigens has a pivotal role in invasive streptococcal disease. However, individuals infected with the same strain can develop very different manifestations. We report here that the immunogenetics of the host influence the outcome of invasive streptococcal infection, and demonstrate the underlying mechanism for these genetic associations. Specific human leukocyte antigen class II haplotypes conferred strong protection from severe systemic disease, whereas others increased the risk of severe disease. Patients with the DRB1*1501/DQB1*0602 haplotype mounted significantly reduced responses and were less likely to develop severe systemic disease (P < 0.0001). We propose that human leukocyte antigen class II allelic variation contributes to differences in severity of invasive streptococcal infections through their ability to regulate cytokine responses triggered by streptococcal superantigens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Low, D.E., Schwartz, B. & McGeer, A. The re-emergance of severe group A streptococcal disease: an evolutionary perspective. in Emerging Pathogens Vol. 7 (ASM Press, Washington, DC, 1998).
Davies, H.D. et al. Invasive group A streptococcal infections in Ontario, Canada. Ontario Group A Streptococcal Study Group. N. Engl. J. Med. 335, 547–554 (1996).
Basma, H. et al. Risk factors in the pathogenesis of invasive group A streptococcal infections: role of protective humoral immunity. Infect. Immun. 67, 1871–1877 (1999).
Chatellier, S. et al. Genetic relatedness and superantigen expression of M type 1 group A streptococcal isolates from severe and nonsevere invasive disease. Infect. Immun. 68, 3523–3534 (2000).
Kotb, M. Bacterial exotoxins as superantigens. Clin. Microbiol. Rev. 8, 411–426 (1995).
Watanabe-Ohnishi, R. et al. Selective depletion of V_-bearing T cells in patients with severe invasive group A streptococcal infections and streptococcal toxic shock syndrome. J. Infect. Dis. 171, 74–84 (1995).
Norrby-Teglund, A. et al. Evidence for superantigen involvement in severe group A streptococcal tissue infections. J. Infect. Dis. 184, 853–860 (2001).
Norrby-Teglund, A. et al. Host variation in cytokine responses to superantigens determine the severity of invasive group A streptococcal infection. Eur. J. Immunol. 30, 3247–3255 (2000).
Hill, A.V. Genetics and genomics of infectious disease susceptibility. Br. Med. Bull. 55, 401–413 (1999).
Herman, A., Croteau, G., Sekaly, R.P., Kappler, J. & Marrack, P. HLA-DR alleles differ in their ability to present staphylococcal enterotoxins to T cells. J. Exp. Med. 172, 709–717 (1990).
Kaul, R., McGeer, A., Low, D.E., Green, K. & Schwartz, B. Population-based surveillance for group A streptococcal necrotizing fasciitis: Clinical features, prognostic indicators, and microbiologic analysis of seventy-seven cases. Ontario Group A Streptococcal Study. Am. J. Med. 103, 18–24 (1997).
Olerup, O. & Zetterquist, H. HLA-DR typing by PCR amplification with sequence-specific primers (PCR-SSP) in 2 hours: an alternative to serological DR typing in clinical practice including donor-recipient matching in cadaveric transplantation. Tissue Antigens 39, 2253 (1992).
Guedez, Y. et al. HLA class II associations with rheumatic heart disease are more evident and consistent among clinically homogeneous patients. Circulation 99, 2784–2790 (1999).
Wade, J., NG, EM, & Pellett, F. Caucasian Canadian normal. in HLA 1998 (ed. Gjertson, D.W. & Terasaki, P.I.) (American Society for Histocompatibility and Immunogenetics, Lenexa, 1998).
Kaul, R. et al. Intravenous immunoglobulin therapy for streptococcal toxic shock syndrome—a comparative observational study. The Canadian Streptococcal Study Group. Clin. Infect. Dis. 28, 800–807 (1999).
Dorak, M.T. et al. TNF microsatellites and streptococcal toxic shock syndrome. Hum. Immunol. 62 (Suppl. 1): S30 (2001).
Colman, G., Tanna, A., Efstratiou, A. & Gaworzewska, E.T. The serotypes of Streptococcus pyogenes present in Britain during 1980–1990 and their association with disease. J. Med. Microbiol. 39, 165–178 (1993).
Stromberg, A., Romanus, V. & Burman, L.G. Outbreak of group A streptococcal bacteremia in Sweden: an epidemiologic and clinical study. J. Infect. Dis. 164, 595–598 (1991).
Martin, P.R. & Hoiby, E.A. Streptococcal serogroup A epidemic in Norway 1987–1988. Scand. J. Infect. Dis. 22, 421–429 (1990).
Hoge, C.W. et al. The changing epidemiology of invasive group A streptococcal infections and the emergence of streptococcal toxic shock-like syndrome. A retrospective population–based study. JAMA 269, 384–389 (1993).
Norrby-Teglund, A., Lustig, R. & Kotb, M. Differential induction of Th1 versus Th2 cytokines by group A streptococcal toxic shock syndrome isolates. Infect. Immun. 65, 5209–5215 (1997).
Newton, D.W. et al. Mutations in the MHC class II binding domains of staphylococcal enterotoxin A differentially affect T cell receptor V_ specificity. J. Immunol. 157, 3988–3994 (1996).
Ohnishi, H. et al. CD28 cross-linking augments TCR-mediated signals and costimulates superantigen responses. J. Immunol. 154, 3180–3193 (1995).
Acknowledgements
We thank all infectious disease physicians in Canada who have contributed valuable samples and clinical information for this study; L. Trepski and the Ontario Streptococcal Study Group in Canada; and Y. Guédez of the Veterans Administration in Memphis. This work was supported by grants from the U.S. Veterans Administration Merit Award (M.K.), the National Institutes of Health (AI40198, M.K.) and the Swedish Medical Research Council (A.N.-T). The Ontario Streptococcal Study Group was originally started by funds from the Center for Disease Control.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Kotb, M., Norrby-Teglund, A., McGeer, A. et al. An immunogenetic and molecular basis for differences in outcomes of invasive group A streptococcal infections. Nat Med 8, 1398–1404 (2002). https://doi.org/10.1038/nm1202-800
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1202-800
This article is cited by
-
Novel insights into the immune response to bacterial T cell superantigens
Nature Reviews Immunology (2024)
-
A decisive bridge between innate immunity and the pathognomonic morphological characteristics of type 1 diabetes demonstrated by instillation of heat-inactivated bacteria in the pancreatic duct of rats
Acta Diabetologica (2022)
-
Streptolysin O concentration and activity is central to in vivo phenotype and disease outcome in Group A Streptococcus infection
Scientific Reports (2021)
-
Extracellular bacterial lymphatic metastasis drives Streptococcus pyogenes systemic infection
Nature Communications (2020)
-
Elevated risk of invasive group A streptococcal disease and host genetic variation in the human leucocyte antigen locus
Genes & Immunity (2020)