Abstract
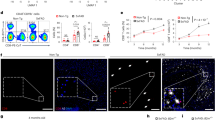
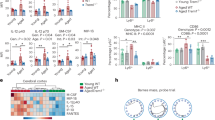
Alzheimer's disease is the most common dementia and is pathologically characterized by deposition of amyloid-β peptide (Aβ) into β-amyloid plaques, neuronal injury and low-level, chronic activation of brain immunity1. Transforming growth factor-βs (TGF-βs) are pleiotropic cytokines that have key roles in immune cell activation, inflammation and repair after injury2. We genetically interrupted TGF-β and downstream Smad2/3 signaling (TGF-β–Smad2/3) in innate immune cells by inducing expression of CD11c promoter–driven dominant-negative TGF-β receptor type II in C57BL/6 mice (CD11c-DNR)3, crossed these mice with mice overexpressing mutant human amyloid precursor protein, the Tg2576 Alzheimer's disease mouse model4, and evaluated Alzheimer's disease-like pathology. Aged double-transgenic mice showed complete mitigation of Tg2576-associated hyperactivity and partial mitigation of defective spatial working memory. Brain parenchymal and cerebrovascular β-amyloid deposits and Aβ abundance were markedly (up to 90%) attenuated in Tg2576–CD11c-DNR mice. This was associated with increased infiltration of Aβ-containing peripheral macrophages around cerebral vessels and β-amyloid plaques. In vitro, cultures of peripheral macrophages, but not microglia, from CD11c-DNR mice showed blockade of classical TGF-β–activated Smad2/3 but also showed hyperactivation of alternative bone morphogenic protein–activated Smad1/5/8 signaling and increased Aβ phagocytosis. Similar effects were noted after pharmacological inhibition of activin-like kinase-5, a type I TGF-β receptor. Taken together, our results suggest that blockade of TGF-β–Smad2/3 signaling in peripheral macrophages represents a new therapeutic target for Alzheimer's disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Selkoe, D.J. Alzheimer's disease: genes, proteins, and therapy. Physiol. Rev. 81, 741–766 (2001).
Li, M.O., Wan, Y.Y., Sanjabi, S., Robertson, A.K. & Flavell, R.A. Transforming growth factor-β regulation of immune responses. Annu. Rev. Immunol. 24, 99–146 (2006).
Laouar, Y., Sutterwala, F.S., Gorelik, L. & Flavell, R.A. Transforming growth factor-β controls T helper type 1 cell development through regulation of natural killer cell interferon-gamma. Nat. Immunol. 6, 600–607 (2005).
Hsiao, K. et al. Correlative memory deficits, Aβ elevation and amyloid plaques in transgenic mice. Science 274, 99–102 (1996).
Akiyama, H. et al. Inflammation and Alzheimer's disease. Neurobiol. Aging 21, 383–421 (2000).
Wisniewski, H.M., Wegiel, J., Wang, K.C., Kujawa, M. & Lach, B. Ultrastructural studies of the cells forming amyloid fibers in classical plaques. Can. J. Neurol. Sci. 16, 535–542 (1989).
Brionne, T.C., Tesseur, I., Masliah, E. & Wyss-Coray, T. Loss of TGF-β1 leads to increased neuronal cell death and microgliosis in mouse brain. Neuron 40, 1133–1145 (2003).
Wyss-Coray, T., Lin, C., Sanan, D.A., Mucke, L. & Masliah, E. Chronic overproduction of transforming growth factor-β1 by astrocytes promotes Alzheimer's disease–like microvascular degeneration in transgenic mice. Am. J. Pathol. 156, 139–150 (2000).
Wyss-Coray, T. et al. Amyloidogenic role of cytokine TGF-β1 in transgenic mice and in Alzheimer's disease. Nature 389, 603–606 (1997).
Wyss-Coray, T. et al. TGF-β1 promotes microglial amyloid-β clearance and reduces plaque burden in transgenic mice. Nat. Med. 7, 612–618 (2001).
Tesseur, I. et al. Deficiency in neuronal TGF-β signaling promotes neurodegeneration and Alzheimer's pathology. J. Clin. Invest. 116, 3060–3069 (2006).
Holcomb, L. et al. Accelerated Alzheimer-type phenotype in transgenic mice carrying both mutant amyloid precursor protein and presenilin 1 transgenes. Nat. Med. 4, 97–100 (1998).
King, D.L. et al. Progressive and gender-dependent cognitive impairment in the APP(SW) transgenic mouse model for Alzheimer's disease. Behav. Brain Res. 103, 145–162 (1999).
Irizarry, M.C., McNamara, M., Fedorchak, K., Hsiao, K. & Hyman, B.T. APPSw transgenic mice develop age-related Aβ deposits and neuropil abnormalities, but no neuronal loss in CA1. J. Neuropathol. Exp. Neurol. 56, 965–973 (1997).
Callahan, M.J. et al. Augmented senile plaque load in aged female β-amyloid precursor protein–transgenic mice. Am. J. Pathol. 158, 1173–1177 (2001).
Ellis, R.J. et al. Cerebral amyloid angiopathy in the brains of patients with Alzheimer's disease: the CERAD experience, Part XV. Neurology 46, 1592–1596 (1996).
Robbins, E.M. et al. Kinetics of cerebral amyloid angiopathy progression in a transgenic mouse model of Alzheimer disease. J. Neurosci. 26, 365–371 (2006).
DeMattos, R.B., Bales, K.R., Cummins, D.J., Paul, S.M. & Holtzman, D.M. Brain to plasma amyloid-β efflux: a measure of brain amyloid burden in a mouse model of Alzheimer's disease. Science 295, 2264–2267 (2002).
Lemere, C.A. et al. Nasal Aβ treatment induces anti-Aβ antibody production and decreases cerebral amyloid burden in PD-APP mice. Ann. NY Acad. Sci. 920, 328–331 (2000).
Jankowsky, J.L. et al. Co-expression of multiple transgenes in mouse CNS: a comparison of strategies. Biomol. Eng. 17, 157–165 (2001).
Geissmann, F., Jung, S. & Littman, D.R. Blood monocytes consist of two principal subsets with distinct migratory properties. Immunity 19, 71–82 (2003).
Westerman, M.A. et al. The relationship between Aβ and memory in the Tg2576 mouse model of Alzheimer's disease. J. Neurosci. 22, 1858–1867 (2002).
Nicoll, J.A. et al. Neuropathology of human Alzheimer disease after immunization with amyloid-β peptide: a case report. Nat. Med. 9, 448–452 (2003).
Town, T., Nikolic, V. & Tan, J. The microglial “activation” continuum: from innate to adaptive responses. J. Neuroinflammation 2, 24 (2005).
Stalder, A.K. et al. Invasion of hematopoietic cells into the brain of amyloid precursor protein transgenic mice. J. Neurosci. 25, 11125–11132 (2005).
Simard, A.R., Soulet, D., Gowing, G., Julien, J.P. & Rivest, S. Bone marrow–derived microglia play a critical role in restricting senile plaque formation in Alzheimer's disease. Neuron 49, 489–502 (2006).
El Khoury, J. et al. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nat. Med. 13, 432–438 (2007).
Ajami, B., Bennett, J.L., Krieger, C., Tetzlaff, W. & Rossi, F.M. Local self-renewal can sustain CNS microglia maintenance and function throughout adult life. Nat. Neurosci. 10, 1538–1543 (2007).
Mildner, A. et al. Microglia in the adult brain arise from Ly-6ChiCCR2+ monocytes only under defined host conditions. Nat. Neurosci. 10, 1544–1553 (2007).
Tan, J. et al. Role of CD40 ligand in amyloidosis in transgenic Alzheimer's mice. Nat. Neurosci. 5, 1288–1293 (2002).
Acknowledgements
We thank F. Manzo for help with preparing this manuscript and A. Ferrandino and S. Sojitra for expert technical assistance. We are grateful to Y. Wan, P. Rakic and J.J. Breunig for helpful discussion and to P. Rakic and J.J. Breunig for assistance with confocal microscopy. We also thank N. Ruddle for providing the brain FACS protocol. This work was supported by an Alzheimer's Association Grant (to R.A.F. and T.T.), and a US National Institutes of Health Pathway to Independence award (1K99AG029726 and 4R00AG029726 to T.T.). R.A.F. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
T.T., Y.L., C.P., J.T., R.S.D. and R.A.F. designed all experiments; T.T. carried out all experiments except behavioral analysis (C.P.), brain FACS assay (Y.L.) and β-amyloid plaque morphometric analysis (T.M.); T.T. and C.A.S. conducted statistical analysis of the data; T.T., R.S.D. and R.A.F. supervised the project; T.T. wrote the manuscript and T.T., C.P., J.T., R.S.D. and R.A.F. edited the manuscript.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–5, Supplementary Tables 1 and 2, and Supplementary Methods (PDF 2108 kb)
Rights and permissions
About this article
Cite this article
Town, T., Laouar, Y., Pittenger, C. et al. Blocking TGF-β–Smad2/3 innate immune signaling mitigates Alzheimer-like pathology. Nat Med 14, 681–687 (2008). https://doi.org/10.1038/nm1781
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1781
This article is cited by
-
Peripheral blood amyloid-β involved in the pathogenesis of Alzheimer’s disease via impacting on peripheral innate immune cells
Journal of Neuroinflammation (2024)
-
Exercise suppresses neuroinflammation for alleviating Alzheimer’s disease
Journal of Neuroinflammation (2023)
-
The neuroimmune axis of Alzheimer’s disease
Genome Medicine (2023)
-
Integrative Bioinformatics Analysis of mRNA Expression Profiles of Mice to Explore the Key Genes Involved in Crim1 Mutation-Induced Congenital Cataracts
Biochemical Genetics (2023)
-
Phenazopyridine promotes RPS23RG1/Rps23rg1 transcription and ameliorates Alzheimer-associated phenotypes in mice
Neuropsychopharmacology (2022)