Abstract
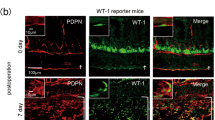
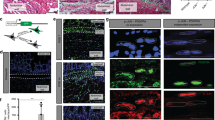
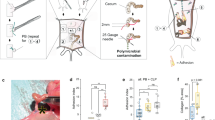
Intestinal adhesions are bands of fibrous tissue that connect the loops of the intestine to each other, to other abdominal organs or to the abdominal wall1,2,3. Fibrous tissue formation is regulated by the balance between plasminogen activator inhibitor type 1 (PAI-1) and tissue-type plasminogen activator (tPA), which reciprocally regulate fibrin deposition. Several components of the inflammatory system, including cytokines4, chemokines, cell adhesion molecules and neuropeptide substance P, have been reported to participate in adhesion formation4,5,6,7. We have used cecal cauterization to develop a unique experimental mouse model of intestinal adhesion. Mice developed severe intestinal adhesion after this treatment. Adhesion development depended upon the interferon-γ (IFN-γ) and signal transducer and activator of transcription-1 (STAT1) system. Natural killer T (NKT) cell–deficient mice developed adhesion poorly, whereas they developed severe adhesion after reconstitution with NKT cells from wild-type mice, suggesting that NKT cell IFN-γ production is indispensable for adhesion formation. This response does not depend on STAT4, STAT6, interleukin-12 (IL-12), IL-18, tumor necrosis factor-α, Toll-like receptor 4 or myeloid differentiation factor-88–mediated signals. Wild-type mice increased the ratio of PAI-1 to tPA after cecal cauterization, whereas Ifng−/− or Stat1−/− mice did not, suggesting that IFN-γ has a crucial role in the differential regulation of PAI-1 and tPA. Additionally, hepatocyte growth factor, a potent mitogenic factor for hepatocytes8,9, strongly inhibited intestinal adhesion by diminishing IFN-γ production, providing a potential new way to prevent postoperative adhesions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Holmdahl, L. The role of fibrinolysis in adhesion formation. Eur. J. Surg. 163 (Suppl. 577), 24–31 (1997).
Raftery, A.T. Regeneration of peritoneum: a fibrinolytic study. J. Anat. 129, 659–664 (1979).
Sulaiman, H., Dawson, L., Laurent, G.J., Bellingan, G.J. & Herrick, S.E. Role of plasminogen activators in peritoneal adhesion formation. Biochem. Soc. Trans. 30, 126–131 (2002).
Holmdahl, L. & Ivarsson, M.L. The role of cytokines, coagulation, and fibrinolysis in peritoneal tissue repair. Eur. J. Surg. 165, 1012–1109 (1999).
Fanciullacci, M., Fedi, S., Alessandri, M. & Pietrini, U. Substance P–induced fibrinolysis in the forearm of healthy humans. Experientia 49, 242–244 (1993).
Reed, K.L. et al. Neurokinin-1 receptor and substance P messenger RNA levels increase during intraabdominal adhesion formation. J. Surg. Res. 108, 165–172 (2002).
Reed, K.L. et al. A neurokinin 1 receptor antagonist decreases postoperative peritoneal adhesion formation and increases peritoneal fibrinolytic activity. Proc. Natl. Acad. Sci. USA 101, 9115–9120 (2004).
Miyazawa, K. et al. Molecular cloning and sequence analysis of cDNA for human hepatocyte growth factor. Biochem. Biophys. Res. Commun. 163, 967–973 (1989).
Nakamura, T. et al. Molecular cloning and expression of human hepatocyte growth factor. Nature 342, 440–443 (1989).
Ellis, H. The clinical significance of adhesions: focus on intestinal obstruction. Eur. J. Surg. 163 (Suppl. 577), 5–9 (1997).
Ellis, H. et al. Adhesion-related hospital readmissions after abdominal and pelvic surgery: a retrospective cohort study. Lancet 353, 1476–1480 (1999).
Menzies, D. & Ellis, H. Intestinal obstruction from adhesions–how big is the problem? Ann. R. Coll. Surg. Engl. 72, 60–63 (1990).
Parker, M.C. et al. Colorectal surgery: the risk and burden of adhesion-related complications. Colorectal Dis. 6, 506–511 (2004).
Ghellai, A.M. et al. Role of transforming growth factor-β1 in peritonitis-induced adhesions. J. Gastrointest. Surg. 4, 316–323 (2000).
Chung, D.R. et al. CD4+ T cells regulate surgical and postinfectious adhesion formation. J. Exp. Med. 195, 1471–1478 (2002).
Bendelac, A., Savage, P.B. & Teyton, L. The biology of NKT cells. Annu. Rev. Immunol. 25, 297–336 (2007).
Taniguchi, M., Harada, M., Kojo, S., Nakayama, T. & Wakao, H. The regulatory role of Vα14 NKT cells in innate and acquired immune response. Annu. Rev. Immunol. 21, 483–513 (2003).
Yoshimoto, T. & Paul, W.E. CD4pos, NK1.1pos T cells promptly produce interleukin 4 in response to in vivo challenge with anti-CD3. J. Exp. Med. 179, 1285–1295 (1994).
Cui, J. et al. Requirement for Vα14 NKT cells in IL-12–mediated rejection of tumors. Science 278, 1623–1626 (1997).
Meraz, M.A. et al. Targeted disruption of the Stat1 gene in mice reveals unexpected physiologic specificity in the JAK-STAT signaling pathway. Cell 84, 431–442 (1996).
Kawano, T. et al. Natural killer–like nonspecific tumor cell lysis mediated by specific ligand-activated Vα14 NKT cells. Proc. Natl. Acad. Sci. USA 95, 5690–5693 (1998).
Kaplan, M.H., Sun, Y.L., Hoey, T. & Grusby, M.J. Impaired IL-12 responses and enhanced development of TH2 cells in Stat4-deficient mice. Nature 382, 174–177 (1996).
Takeda, K. et al. Essential role of Stat6 in IL-4 signalling. Nature 380, 627–630 (1996).
Katayama, I. & Nishioka, K. Substance P augments fibrogenic cytokine-induced fibroblast proliferation: possible involvement of neuropeptide in tissue fibrosis. J. Dermatol. Sci. 15, 201–206 (1997).
Zimmer, A. et al. Hypoalgesia in mice with a targeted deletion of the tachykinin 1 gene. Proc. Natl. Acad. Sci. USA 95, 2630–2635 (1998).
Holmdahl, L., Eriksson, E., al-Jabreen, M. & Risberg, B. Fibrinolysis in human peritoneum during operation. Surgery 119, 701–705 (1996).
Kuroiwa, T. et al. Hepatocyte growth factor ameliorates acute graft-versus-host disease and promotes hematopoietic function. J. Clin. Invest. 107, 1365–1373 (2001).
Ido, A., Numata, M., Kodama, M. & Tsubouchi, H. Mucosal repair and growth factors: recombinant human hepatocyte growth factor as an innovative therapy for inflammatory bowel disease. J. Gastroenterol. 40, 925–931 (2005).
Kennedy, R., Costain, D.J., McAlister, V.C. & Lee, T.D. Prevention of experimental postoperative peritoneal adhesions by N,O-carboxymethyl chitosan. Surgery 120, 866–870 (1996).
Kocak, I., Unlu, C., Akcan, Y. & Yakin, K. Reduction of adhesion formation with cross-linked hyaluronic acid after peritoneal surgery in rats. Fertil. Steril. 72, 873–878 (1999).
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research on Priority Areas and a Hitech Research Center grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan and a Grant-in-Aid for Scientific Research (B; No. 19390342) from the Japan Society for Promotion of Science.
Author information
Authors and Affiliations
Contributions
Tomohiro Yoshimoto and K.N. formulated the hypothesis and initiated and organized the study; H.K. and Tomohiro Yoshimoto performed the main experimental work and analyzed the data; Takayuki Yoshimoto helped with some experimental procedures; Tomohiro Yoshimoto and K.N. oversaw the experiments, analyzed the data and provided the main funding for the research; J.F. contributed to data discussion; Tomohiro Yoshimoto drafted the manuscript and K.N. prepared the final manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–3, Supplementary Tables 1 and 2, and Supplementary Methods (PDF 301 kb)
Rights and permissions
About this article
Cite this article
Kosaka, H., Yoshimoto, T., Yoshimoto, T. et al. Interferon-γ is a therapeutic target molecule for prevention of postoperative adhesion formation. Nat Med 14, 437–441 (2008). https://doi.org/10.1038/nm1733
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1733
This article is cited by
-
Modified pull-through coloanal anastomosis to avoid permanent stomas and reduce postoperative complications for lower rectal tumors
Surgical Endoscopy (2023)
-
High visceral to subcutaneous fat area ratio predicts early postoperative small bowel obstruction after surgery for colorectal cancer
Langenbeck's Archives of Surgery (2022)
-
Study on the correlation between preoperative inflammatory indexes and adhesional perinephric fat before laparoscopic partial nephrectomy
BMC Urology (2021)
-
Cell barrier function of resident peritoneal macrophages in post-operative adhesions
Nature Communications (2021)
-
Is There a Genetic Predisposition to Postoperative Adhesion Development?
Reproductive Sciences (2021)