Abstract
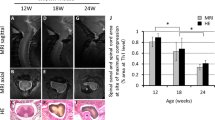
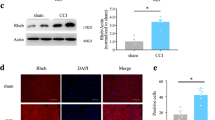
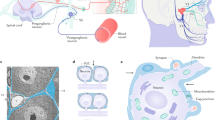
Treatment of neuropathic pain, triggered by multiple insults to the nervous system, is a clinical challenge because the underlying mechanisms of neuropathic pain development remain poorly understood1,2,3,4. Most treatments do not differentiate between different phases of neuropathic pain pathophysiology and simply focus on blocking neurotransmission, producing transient pain relief. Here, we report that early- and late-phase neuropathic pain development in rats and mice after nerve injury require different matrix metalloproteinases (MMPs). After spinal nerve ligation, MMP-9 shows a rapid and transient upregulation in injured dorsal root ganglion (DRG) primary sensory neurons consistent with an early phase of neuropathic pain, whereas MMP-2 shows a delayed response in DRG satellite cells and spinal astrocytes consistent with a late phase of neuropathic pain. Local inhibition of MMP-9 by an intrathecal route inhibits the early phase of neuropathic pain, whereas inhibition of MMP-2 suppresses the late phase of neuropathic pain. Further, intrathecal administration of MMP-9 or MMP-2 is sufficient to produce neuropathic pain symptoms. After nerve injury, MMP-9 induces neuropathic pain through interleukin-1β cleavage and microglial activation at early times, whereas MMP-2 maintains neuropathic pain through interleukin-1β cleavage and astrocyte activation at later times. Inhibition of MMP may provide a novel therapeutic approach for the treatment of neuropathic pain at different phases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ji, R.R. & Strichartz, G. Cell signaling and the genesis of neuropathic pain. Sci. STKE 2004, reE14 (2004).
Tsuda, M., Inoue, K. & Salter, M.W. Neuropathic pain and spinal microglia: a big problem from molecules in “small” glia. Trends Neurosci. 28, 101–107 (2005).
Woolf, C.J. & Mannion, R.J. Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet 353, 1959–1964 (1999).
Kehlet, H., Jensen, T.S. & Woolf, C.J. Persistent postsurgical pain: risk factors and prevention. Lancet 367, 1618–1625 (2006).
Parks, W.C., Wilson, C.L. & Lopez-Boado, Y.S. Matrix metalloproteinases as modulators of inflammation and innate immunity. Nat. Rev. Immunol. 4, 617–629 (2004).
Chattopadhyay, S., Myers, R.R., Janes, J. & Shubayev, V. Cytokine regulation of MMP-9 in peripheral glia: Implications for pathological processes and pain in injured nerve. Brain Behav. Immun. 21, 561–568 (2007).
Rosenberg, G.A. Matrix metalloproteinases in neuroinflammation. Glia 39, 279–291 (2002).
Wang, X. et al. Effects of matrix metalloproteinase-9 gene knock-out on morphological and motor outcomes after traumatic brain injury. J. Neurosci. 20, 7037–7042 (2000).
Yong, V.W. Metalloproteinases: mediators of pathology and regeneration in the CNS. Nat. Rev. Neurosci. 6, 931–944 (2005).
Zhao, B.Q. et al. Role of matrix metalloproteinases in delayed cortical responses after stroke. Nat. Med. 12, 441–445 (2006).
Kim, S.H. & Chung, J.M. An experimental model for peripheral neuropathy produced by segmental spinal nerve ligation in the rat. Pain 50, 355–363 (1992).
Zhuang, Z.Y. et al. A peptide c-Jun N-terminal kinase (JNK) inhibitor blocks mechanical allodynia after spinal nerve ligation: respective roles of JNK activation in primary sensory neurons and spinal astrocytes for neuropathic pain development and maintenance. J. Neurosci. 26, 3551–3560 (2006).
Levin, J.I. et al. The discovery of anthranilic acid-based MMP inhibitors. Part 3: incorporation of basic amines. Bioorg. Med. Chem. Lett. 11, 2975–2978 (2001).
Murphy, G. & Willenbrock, F. Tissue inhibitors of matrix metalloendopeptidases. Methods Enzymol. 248, 496–510 (1995).
Coull, J.A. et al. BDNF from microglia causes the shift in neuronal anion gradient underlying neuropathic pain. Nature 438, 1017–1021 (2005).
Raghavendra, V., Tanga, F. & DeLeo, J.A. Inhibition of microglial activation attenuates the development but not existing hypersensitivity in a rat model of neuropathy. J. Pharmacol. Exp. Ther. 306, 624–630 (2003).
Tsuda, M. et al. P2X4 receptors induced in spinal microglia gate tactile allodynia after nerve injury. Nature 424, 778–783 (2003).
Watkins, L.R., Milligan, E.D. & Maier, S.F. Glial activation: a driving force for pathological pain. Trends Neurosci. 24, 450–455 (2001).
Jin, S.X., Zhuang, Z.Y., Woolf, C.J. & Ji, R.R. p38 mitogen-activated protein kinase is activated after a spinal nerve ligation in spinal cord microglia and dorsal root ganglion neurons and contributes to the generation of neuropathic pain. J. Neurosci. 23, 4017–4022 (2003).
Schonbeck, U., Mach, F. & Libby, P. Generation of biologically active IL-1β by matrix metalloproteinases: a novel caspase-1-independent pathway of IL-1β processing. J. Immunol. 161, 3340–3346 (1998).
Samad, T.A. et al. Interleukin-1β-mediated induction of Cox-2 in the CNS contributes to inflammatory pain hypersensitivity. Nature 410, 471–475 (2001).
Sweitzer, S., Martin, D. & DeLeo, J.A. Intrathecal interleukin-1 receptor antagonist in combination with soluble tumor necrosis factor receptor exhibits an anti-allodynic action in a rat model of neuropathic pain. Neuroscience 103, 529–539 (2001).
Sung, C.-S. et al. Inhibition of p38 mitogen-activated protein kinase attenuates interleukin-1β-induced thermal hyperalgesia and inducible nitric oxide synthase expression in the spinal cord. J. Neurochem. 94, 742–752 (2005).
Clark, A.K. et al. Rapid co-release of interleukin 1β and caspase 1 in spinal cord inflammation. J. Neurochem. 99, 868–880 (2006).
Inoue, M. et al. Initiation of neuropathic pain requires lysophosphatidic acid receptor signaling. Nat. Med. 10, 712–718 (2004).
Zhuang, Z.Y., Gerner, P., Woolf, C.J. & Ji, R.R. ERK is sequentially activated in neurons, microglia, and astrocytes by spinal nerve ligation and contributes to mechanical allodynia in this neuropathic pain model. Pain 114, 149–159 (2005).
Dworkin, R.H. et al. Advances in neuropathic pain: diagnosis, mechanisms, and treatment recommendations. Arch. Neurol. 60, 1524–1534 (2003).
Tan, P.H., Yang, L.C., Shih, H.C., Lan, K.C. & Cheng, J.T. Gene knockdown with intrathecal siRNA of NMDA receptor NR2B subunit reduces formalin-induced nociception in the rat. Gene Ther. 12, 59–66 (2005).
Chen, S. et al. Disruption of ErbB receptor signaling in adult non-myelinating Schwann cells causes progressive sensory loss. Nat. Neurosci. 6, 1186–1193 (2003).
Acknowledgements
The work was supported in part by US National Institutes of Health grants R01-DE17794, R01-NS54362 and TW7180 to R.-R.J. and R01-NS37074, R01-NS48422, R01-NS56458, P01-NS55104 and P50-NS10828 to E.H.L. P.-H.T. was supported by a grant from E-DA Hospital/I-Shou University, Taiwan. We thank T. Kohno (Niigata University, Japan) for providing FR167653 compound and Q. Ma (Harvard Medical School) for critical reading of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.K. performed behavioral and immunohistochemical experiments; Z.-Z.X. conducted western blotting, DRG culture and zymography studies; X.W. performed zymography studies and contributed to project development; J.Y.P. conducted behavioral studies; Z.-Y.Z. conducted initial behavioral and histochemical studies; P.-H.T. designed siRNAs and initially tested MMP-9 siRNA; Y.-J.G. conducted ELISA studies and prepared astrocyte cultures; K.R. and G.C. contributed to electron microscopy studies; E.H.L. developed the project and provided critical review and comments on the manuscript. R.-R.J. developed and supervised the project, designed all the experiments, conducted some data analysis and prepared the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Methods, Supplementary Results, Supplementary Discussion (PDF 964 kb)
Rights and permissions
About this article
Cite this article
Kawasaki, Y., Xu, ZZ., Wang, X. et al. Distinct roles of matrix metalloproteases in the early- and late-phase development of neuropathic pain. Nat Med 14, 331–336 (2008). https://doi.org/10.1038/nm1723
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1723
This article is cited by
-
Hydrogen attenuates postoperative pain through Trx1/ASK1/MMP9 signaling pathway
Journal of Neuroinflammation (2023)
-
CaMKII and CaV3.2 T-type calcium channel mediate Connexin-43-dependent inflammation by activating astrocytes in vincristine-induced neuropathic pain
Cell Biology and Toxicology (2023)
-
Epigenetic Up-Regulation of ADAMTS4 in Sympathetic Ganglia is Involved in the Maintenance of Neuropathic Pain Following Nerve Injury
Neurochemical Research (2023)
-
Drug repositioning: diacerein as a new therapeutic approach in a mice model of sciatic nerve injury
Pharmacological Reports (2023)
-
Chondroitin and glucosamine sulphate reduced proinflammatory molecules in the DRG and improved axonal function of injured sciatic nerve of rats
Scientific Reports (2022)